Example solution to the exercise 1
... e4 The lifespan of the classical atom can be calculated when the above differential is integrated from t = 0 to tend when the electron collapses into the nucleus. During this time the radius changes from r = a0 , when t = 0 to r = 0 when t = tend . For the final calculation one requires the natural ...
... e4 The lifespan of the classical atom can be calculated when the above differential is integrated from t = 0 to tend when the electron collapses into the nucleus. During this time the radius changes from r = a0 , when t = 0 to r = 0 when t = tend . For the final calculation one requires the natural ...
Adobe Acrobat file () - Wayne State University Physics and
... In an analysis relating Bohr's theory to the de Broglie wavelength of electrons, when an electron moves from the n = 1 level to the n = 3 level, the circumference of its orbit becomes 9 times greater. This occurs because (a) there are 3 times as many wavelengths in the new orbit, (b) there are 3 tim ...
... In an analysis relating Bohr's theory to the de Broglie wavelength of electrons, when an electron moves from the n = 1 level to the n = 3 level, the circumference of its orbit becomes 9 times greater. This occurs because (a) there are 3 times as many wavelengths in the new orbit, (b) there are 3 tim ...
Lecture 22: Simon`s Problem and towards Shor 1 Overview 2
... The techniques used in the quantum solution to Simon’s problem is essential for Shor’s algorithms for integer factoring and the discrete logarithm problem. Shor’s algorithm is a quantum algorithm for order finding (given a numbers a and n, find ordn (a)). But the following lemma would show that this ...
... The techniques used in the quantum solution to Simon’s problem is essential for Shor’s algorithms for integer factoring and the discrete logarithm problem. Shor’s algorithm is a quantum algorithm for order finding (given a numbers a and n, find ordn (a)). But the following lemma would show that this ...
1991B2. In region I shown above, there is a potential difference V
... 1991B2. In region I shown above, there is a potential difference V between two large, parallel plates separated by a distance d. In region II, to the right of plate D, there is a uniform magnetic field B pointing perpendicularly out of the paper. An electron, charge –e and mass m, is released from r ...
... 1991B2. In region I shown above, there is a potential difference V between two large, parallel plates separated by a distance d. In region II, to the right of plate D, there is a uniform magnetic field B pointing perpendicularly out of the paper. An electron, charge –e and mass m, is released from r ...
Quantum Memories at Room-Temperature Supervisors: Dr Dylan
... For the Master’s project, we are proposing an investigation into a new noise-suppression technique in our lambda Raman quantum memory. This will be demonstration of a new protocol: a quantum Zeno noise suppression technique to kill a noise-process prohibits quantum operation, a process known as four ...
... For the Master’s project, we are proposing an investigation into a new noise-suppression technique in our lambda Raman quantum memory. This will be demonstration of a new protocol: a quantum Zeno noise suppression technique to kill a noise-process prohibits quantum operation, a process known as four ...
Atomic Structure
... Formulated an equation that would describe the behaviour and energies of submicroscopic particles in general. Used this equation to find the probability of locating an electron in a given volume. This led to quantum mechanics or wave mechanics model. Describe the distribution of electron in an atom ...
... Formulated an equation that would describe the behaviour and energies of submicroscopic particles in general. Used this equation to find the probability of locating an electron in a given volume. This led to quantum mechanics or wave mechanics model. Describe the distribution of electron in an atom ...
1 ψ ω ω ω ψ ψ ψ
... If the potential energy of the system does not vary with time, the time and spatial dependences of the wave function can be separated; and the time dependence can be represented simply by e − iωt as in this case, so we will concentrate only on the space part: ψ(x) Since the wave function is often co ...
... If the potential energy of the system does not vary with time, the time and spatial dependences of the wave function can be separated; and the time dependence can be represented simply by e − iωt as in this case, so we will concentrate only on the space part: ψ(x) Since the wave function is often co ...
Quantum Mechanics
... theory Niels Bohr (1913) proposed model of atom with electron orbits based on quantized energy states Difference between energy states always some multiple of Planck’s constant ...
... theory Niels Bohr (1913) proposed model of atom with electron orbits based on quantized energy states Difference between energy states always some multiple of Planck’s constant ...
Activity 2 - hrsbstaff.ednet.ns.ca
... 4. The experiment demonstrating interference of buckminsterfullerene, C60, had the molecules moving at 210 m/s. Each molecule has an atomic mass of 720 atomic units and a diameter of 1 nm. The molecules passed through the slits with widths of 50 nm and separations of 100 nm. After the slits, the mo ...
... 4. The experiment demonstrating interference of buckminsterfullerene, C60, had the molecules moving at 210 m/s. Each molecule has an atomic mass of 720 atomic units and a diameter of 1 nm. The molecules passed through the slits with widths of 50 nm and separations of 100 nm. After the slits, the mo ...
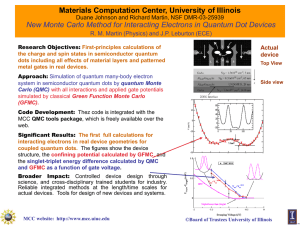
Materials Computation Center R.M. Martin and J.P. Leburton
... interacting electrons in real device geometries for coupled quantum dots. The figures show the device structure, the confining potential calculated by GFMC, and the singlet-triplet energy difference calculated by QMC and GFMC as a function of gate voltage. Broader Impact: Controlled device design th ...
... interacting electrons in real device geometries for coupled quantum dots. The figures show the device structure, the confining potential calculated by GFMC, and the singlet-triplet energy difference calculated by QMC and GFMC as a function of gate voltage. Broader Impact: Controlled device design th ...
Bohr Model, Quantum Mechanical Model
... electron of the hydrogen atom could occupy only certain energy states, stationary states. This explained the line spectra. 2. He explained that each line corresponded to a circular, fixed energy orbit around the nucleus. ...
... electron of the hydrogen atom could occupy only certain energy states, stationary states. This explained the line spectra. 2. He explained that each line corresponded to a circular, fixed energy orbit around the nucleus. ...
Solutions to the exam itself are now available.
... (c) You now know n, but not l or m for this atom. Calculate the maximum average radius this atom could have, and in doing so, tell me what l and m values lead to this maximum value. (If you didn’t get a value for n in part (b), assume n = 100, which isn’t the answer to part (b).) If n is this huge n ...
... (c) You now know n, but not l or m for this atom. Calculate the maximum average radius this atom could have, and in doing so, tell me what l and m values lead to this maximum value. (If you didn’t get a value for n in part (b), assume n = 100, which isn’t the answer to part (b).) If n is this huge n ...
1) Which of the following concepts was discussed in Chapter 1
... Q13) The figure shows three infinite potential wells of widths L, 2L, and 3L; each contains an electron in the state for which n = 10. Rank the wells according to (a) the number of maxima for the probability density of the electron and (b) the energy of the electron, greatest first. 1) (a) all tie ...
... Q13) The figure shows three infinite potential wells of widths L, 2L, and 3L; each contains an electron in the state for which n = 10. Rank the wells according to (a) the number of maxima for the probability density of the electron and (b) the energy of the electron, greatest first. 1) (a) all tie ...
preprint
... If you want to understand the nature of time, you might want to look at our most fundamental physical theories. There are two such: general relativity, and quantum field theory. Any accurate description of the universe — and in particular, of the nature of time — must somehow combine the insights of ...
... If you want to understand the nature of time, you might want to look at our most fundamental physical theories. There are two such: general relativity, and quantum field theory. Any accurate description of the universe — and in particular, of the nature of time — must somehow combine the insights of ...
photoelectric effect
... Note from Eq. (3) that h/e has the dimensions of volt-sec (V-s) and W0/e has the dimensions of volts (V). From these results you can directly express h in terms of eV-s and W0 in terms of eV, where 1eV = 1 electron – volt = (charge of electrons) (1 volt). If you had some independent determination of ...
... Note from Eq. (3) that h/e has the dimensions of volt-sec (V-s) and W0/e has the dimensions of volts (V). From these results you can directly express h in terms of eV-s and W0 in terms of eV, where 1eV = 1 electron – volt = (charge of electrons) (1 volt). If you had some independent determination of ...
Quantum electrodynamics

In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and special relativity is achieved. QED mathematically describes all phenomena involving electrically charged particles interacting by means of exchange of photons and represents the quantum counterpart of classical electromagnetism giving a complete account of matter and light interaction.In technical terms, QED can be described as a perturbation theory of the electromagnetic quantum vacuum. Richard Feynman called it ""the jewel of physics"" for its extremely accurate predictions of quantities like the anomalous magnetic moment of the electron and the Lamb shift of the energy levels of hydrogen.