WEEK 2: 4 S
... absorb a photon? (How do you know?) A. n = 1 to n = 3 B. n = 4 to n = 3 C. n = 3 to n = 2 D. n = 3 to n = 1 E. n = 2 to n = 3 5B. Which of the above electron transitions in a hydrogen atom will result in emission of light with the longest wavelength? ------------------------------------------------- ...
... absorb a photon? (How do you know?) A. n = 1 to n = 3 B. n = 4 to n = 3 C. n = 3 to n = 2 D. n = 3 to n = 1 E. n = 2 to n = 3 5B. Which of the above electron transitions in a hydrogen atom will result in emission of light with the longest wavelength? ------------------------------------------------- ...
N 2
... into an upper level E4 by some mechanism (for example, a collision with another atom or absorption of high-energy radiation). It then decays to E3, then to E2, and finally to the ground state E1. Let us assume that the time it takes to decay from E4 to E3 is much longer than the time it takes to dec ...
... into an upper level E4 by some mechanism (for example, a collision with another atom or absorption of high-energy radiation). It then decays to E3, then to E2, and finally to the ground state E1. Let us assume that the time it takes to decay from E4 to E3 is much longer than the time it takes to dec ...
Example 1:
... There are seven identically-shaped lollipops in a closed paper bag, of varying flavors: four grape, two cherry, and one blueberry. You are allowed to randomly draw two lollipops to keep for yourself. a) What is the probability of drawing a cherry lollipop in your 1st draw? ________________ b) Say yo ...
... There are seven identically-shaped lollipops in a closed paper bag, of varying flavors: four grape, two cherry, and one blueberry. You are allowed to randomly draw two lollipops to keep for yourself. a) What is the probability of drawing a cherry lollipop in your 1st draw? ________________ b) Say yo ...
poster
... • Individual quanta are detected as localized particles on the screen, yet an interference pattern still develops over a period of time. ...
... • Individual quanta are detected as localized particles on the screen, yet an interference pattern still develops over a period of time. ...
lecture 19 (zipped power point) (update: 13Jan 04)
... Due to the probabilistic interpretation of the matter wave, the notion of “existence” of a physical entity, at its most fundamental level, begins to deviate from our conventional wisdom The existence of an entity is now no more be deterministic notion (e.g. it either exist or not at all) but only ...
... Due to the probabilistic interpretation of the matter wave, the notion of “existence” of a physical entity, at its most fundamental level, begins to deviate from our conventional wisdom The existence of an entity is now no more be deterministic notion (e.g. it either exist or not at all) but only ...
Lecture Notes, Feb 24, 2016
... What happens when we have more than one electron... We will discuss it later as it turns out that each “state” can accommodate only two electrons (Pauli Exclusion Principle) ...
... What happens when we have more than one electron... We will discuss it later as it turns out that each “state” can accommodate only two electrons (Pauli Exclusion Principle) ...
Manifestation of classical phase in a single spontaneously emitted
... wave in a multimode quantized field. We study spontaneous emission, scattering, and re-absorption by two-level atoms in a one-dimensional optical cavity, and observe classical phase information in the complex quantum amplitude for re-absorption of the scattered field. The concept of phase for a quan ...
... wave in a multimode quantized field. We study spontaneous emission, scattering, and re-absorption by two-level atoms in a one-dimensional optical cavity, and observe classical phase information in the complex quantum amplitude for re-absorption of the scattered field. The concept of phase for a quan ...
Physics 120 Homework Set #1 (due Sunday
... frequency of the oscillator via Planck’s constant: E = h. This solves the ultraviolet catastrophe: at high frequency (short wavelength) the energy associated with each quanta is very large and only a few oscillators will have this much energy at any finite temperature. 2) a) What is particle-wav ...
... frequency of the oscillator via Planck’s constant: E = h. This solves the ultraviolet catastrophe: at high frequency (short wavelength) the energy associated with each quanta is very large and only a few oscillators will have this much energy at any finite temperature. 2) a) What is particle-wav ...
pdf
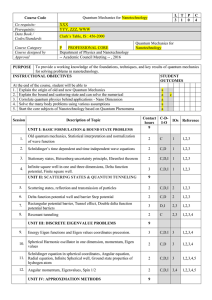
... To provide a working knowledge of the foundations, techniques, and key results of quantum mechanics for solving problems in nanotechnology. INSTRUCTIONAL OBJECTIVES STUDENT OUTCOMES At the end of the course, student will be able to 1. Explain the origin of old and new Quantum Mechanics a 2. Explain ...
... To provide a working knowledge of the foundations, techniques, and key results of quantum mechanics for solving problems in nanotechnology. INSTRUCTIONAL OBJECTIVES STUDENT OUTCOMES At the end of the course, student will be able to 1. Explain the origin of old and new Quantum Mechanics a 2. Explain ...
SAMPLE ABSTRACT
... We demonstrate the narrow switching distribution of an underdamped Josephson junction from the zero to the finite voltage state at millikelvin temperatures. The width of the switching distribution at a nominal temperature of about 20mK was 4.5 nA, which corresponds to an effective noise temperature ...
... We demonstrate the narrow switching distribution of an underdamped Josephson junction from the zero to the finite voltage state at millikelvin temperatures. The width of the switching distribution at a nominal temperature of about 20mK was 4.5 nA, which corresponds to an effective noise temperature ...
Atomic and Molecular Physics for Physicists Ben-Gurion University of the Negev
... angular momentum J, and as JZ=LZ+SZ, and as, when calculating the distances and therefore the forces one has to take into account that g for the orbital motion is gL=1 while for the spin is gS=2, we will have the following forces acting on the atoms: F(LZ=+1, SZ=+1/2), F(LZ=+0, SZ=+1/2), F(LZ=-1, SZ ...
... angular momentum J, and as JZ=LZ+SZ, and as, when calculating the distances and therefore the forces one has to take into account that g for the orbital motion is gL=1 while for the spin is gS=2, we will have the following forces acting on the atoms: F(LZ=+1, SZ=+1/2), F(LZ=+0, SZ=+1/2), F(LZ=-1, SZ ...
Brief introduction to quantum mechanics
... A very elementary approach to Quantum mechanics „There was a time when newspapers said that only twelve men understood the theory of relativity. I do not believe that there ever was such a time... On the other hand, I think it is safe to say that no one understands quantum mechanics“ R.P. Feynman Th ...
... A very elementary approach to Quantum mechanics „There was a time when newspapers said that only twelve men understood the theory of relativity. I do not believe that there ever was such a time... On the other hand, I think it is safe to say that no one understands quantum mechanics“ R.P. Feynman Th ...
Quantum electrodynamics

In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and special relativity is achieved. QED mathematically describes all phenomena involving electrically charged particles interacting by means of exchange of photons and represents the quantum counterpart of classical electromagnetism giving a complete account of matter and light interaction.In technical terms, QED can be described as a perturbation theory of the electromagnetic quantum vacuum. Richard Feynman called it ""the jewel of physics"" for its extremely accurate predictions of quantities like the anomalous magnetic moment of the electron and the Lamb shift of the energy levels of hydrogen.