Periodic Trends Notes 14-15
... • Describe the arrangement of elements in the periodic table in order of increasing atomic number. • Distinguish between the terms group and period. • Apply the relationship between the electron arrangement of elements and their position in the periodic table. • Apply the relationship between the nu ...
... • Describe the arrangement of elements in the periodic table in order of increasing atomic number. • Distinguish between the terms group and period. • Apply the relationship between the electron arrangement of elements and their position in the periodic table. • Apply the relationship between the nu ...
Review of Periodic Trends
... upper left hand corner of the periodic table lower left hand corner of the periodic table lower right hand corner of the periodic table upper right hand corner of the periodic table ...
... upper left hand corner of the periodic table lower left hand corner of the periodic table lower right hand corner of the periodic table upper right hand corner of the periodic table ...
Chapter TWO
... G.R for : It is incorrect to define the atomic radius as the distance from the nucleus to the farthest electron *Because it is impossible to determine the exact position of any electron around the nucleus . Example : If the bond length in the chlorine molecule Cl—Cl is 1.98 Ao and the bond length be ...
... G.R for : It is incorrect to define the atomic radius as the distance from the nucleus to the farthest electron *Because it is impossible to determine the exact position of any electron around the nucleus . Example : If the bond length in the chlorine molecule Cl—Cl is 1.98 Ao and the bond length be ...
Safety First I can… o Follow safe laboratory practices o Identify lab
... use Zeff , shielding, and electron-electron repulsion to explain the trends discuss the size of an atom in terms of the size of the electron cloud. Discuss the role played by electron-electron repulsions and electron-proton attractions on the size of the electron cloud. define and give an example of ...
... use Zeff , shielding, and electron-electron repulsion to explain the trends discuss the size of an atom in terms of the size of the electron cloud. Discuss the role played by electron-electron repulsions and electron-proton attractions on the size of the electron cloud. define and give an example of ...
Lecture 3 - TCD Chemistry
... Is the energy that is necessary to remove an electron from an atom or a molecule. The Ionisation Energy is a measure for the force by which an electron is bound in the atom. The Ionisation Energy is a function of the radius and the charge of an atom: ...
... Is the energy that is necessary to remove an electron from an atom or a molecule. The Ionisation Energy is a measure for the force by which an electron is bound in the atom. The Ionisation Energy is a function of the radius and the charge of an atom: ...
Ch. 5.1 History of the periodic table ppt.
... • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. – By 1886, these elements (scandium, gallium, and germanium) were disc ...
... • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. – By 1886, these elements (scandium, gallium, and germanium) were disc ...
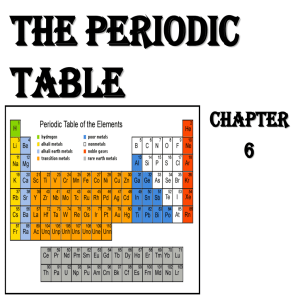
Chemistry 1 Notes #10 Chapter 6 Modern Periodic Table
... • Atoms tend to be smaller the farther to the right they are found across a period. • The trend to smaller atoms across a period is caused by the increasing positive charge of the nucleus, which attracts electrons toward the nucleus. • Atoms tend to be larger the farther down in a group they are fou ...
... • Atoms tend to be smaller the farther to the right they are found across a period. • The trend to smaller atoms across a period is caused by the increasing positive charge of the nucleus, which attracts electrons toward the nucleus. • Atoms tend to be larger the farther down in a group they are fou ...
PERIODIC TABLE
... 4. Noble gases have full orbitals in the highest energy level, called an ____________________. 5. From this low reactivity we can infer that the noble gas electron configuration is very ____________________. 6. Atoms from the other 17 groups either gain or lose electrons to form compounds. In doing ...
... 4. Noble gases have full orbitals in the highest energy level, called an ____________________. 5. From this low reactivity we can infer that the noble gas electron configuration is very ____________________. 6. Atoms from the other 17 groups either gain or lose electrons to form compounds. In doing ...
Chapter 5 The Periodic Table Section 1 Organizing the Elements
... – noble gas: one of the elements of Group 18 of the periodic table – halogen: one of the elements of Group 17 of the periodic table • The noble gases are relatively inert. • The halogens combine easily with metals to form salts. • Nonmetals and their compounds are plentiful on Earth. ...
... – noble gas: one of the elements of Group 18 of the periodic table – halogen: one of the elements of Group 17 of the periodic table • The noble gases are relatively inert. • The halogens combine easily with metals to form salts. • Nonmetals and their compounds are plentiful on Earth. ...
1 CHAPTER 5 – THE PERIODIC LAW What types of useful
... A. Before the Periodic Table was invented, about 63 elements were known. However, they were not organized and only random properties were known about each of the elements. Scientist (who are always looking for patterns) wanted to organize these. B. Dmitri Mendeleev – he made cards for all 63 known e ...
... A. Before the Periodic Table was invented, about 63 elements were known. However, they were not organized and only random properties were known about each of the elements. Scientist (who are always looking for patterns) wanted to organize these. B. Dmitri Mendeleev – he made cards for all 63 known e ...
1 CHAPTER 5 – THE PERIODIC LAW What types of useful
... A. Before the Periodic Table was invented, about 63 elements were known. However, they were not organized and only random properties were known about each of the elements. Scientist (who are always looking for patterns) wanted to organize these. B. Dmitri Mendeleev – he made cards for all 63 known e ...
... A. Before the Periodic Table was invented, about 63 elements were known. However, they were not organized and only random properties were known about each of the elements. Scientist (who are always looking for patterns) wanted to organize these. B. Dmitri Mendeleev – he made cards for all 63 known e ...
Introduction to Mendeleev*s Periodic Table of Elements
... • Ca, Sr, Ba react easily with cold water. • Which way along the group does reactivity increase? ...
... • Ca, Sr, Ba react easily with cold water. • Which way along the group does reactivity increase? ...
The Periodic Table - Anderson High School
... number of valence electrons. • They will form the same kinds of ions. ...
... number of valence electrons. • They will form the same kinds of ions. ...
Atomic Structure and the Periodic Table
... their chemical and physical properties – Known as a group – Labeled by a number at the top of the column – Sometimes a group is called a family of elements because they seem to be related – Ex: Group 17 – “Halogen” group • Tend to combine easily with other elements and compounds, especially elements ...
... their chemical and physical properties – Known as a group – Labeled by a number at the top of the column – Sometimes a group is called a family of elements because they seem to be related – Ex: Group 17 – “Halogen” group • Tend to combine easily with other elements and compounds, especially elements ...
Alkaline Earth Metals
... • Mendeleev - The properties of the elements are a periodic function of their atomic masses. • Moseley - The properties of the elements are a periodic function of their atomic numbers. • Modern Periodic Law states that many of the physical and chemical properties of the elements tend to recur in a s ...
... • Mendeleev - The properties of the elements are a periodic function of their atomic masses. • Moseley - The properties of the elements are a periodic function of their atomic numbers. • Modern Periodic Law states that many of the physical and chemical properties of the elements tend to recur in a s ...
The Periodic Table - Clydebank High School
... • Every element has a unique number called Atomic Number. • The Atomic Number is the number of Protons ( this is equal to the number of electrons in a neutral atom) • The Mass Number is a total of the particles with mass i.e. protons and neutrons. ...
... • Every element has a unique number called Atomic Number. • The Atomic Number is the number of Protons ( this is equal to the number of electrons in a neutral atom) • The Mass Number is a total of the particles with mass i.e. protons and neutrons. ...
The Periodic Table
... Throw it off the highest building, and I'll not break. But put me in the ocean, and I will. What am I? What can run but never walks, has a mouth but never talks, has a head but never weeps, has a bed but ...
... Throw it off the highest building, and I'll not break. But put me in the ocean, and I will. What am I? What can run but never walks, has a mouth but never talks, has a head but never weeps, has a bed but ...
Daily Inquiry: 10-31-2011
... • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. • By 1886, these elements (scandium, gallium, and germanium) were disc ...
... • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. • By 1886, these elements (scandium, gallium, and germanium) were disc ...
Periodic properties
... are highly electropositive giving +1 ions. Because of the very high second ionisation energies of these elements, their oxidation state in compounds never exceeds +1. On the other hand , alkaline earth metals are in general less reactive than alkali metals. This is because of their relatively high i ...
... are highly electropositive giving +1 ions. Because of the very high second ionisation energies of these elements, their oxidation state in compounds never exceeds +1. On the other hand , alkaline earth metals are in general less reactive than alkali metals. This is because of their relatively high i ...
Matching - hrsbstaff.ednet.ns.ca
... ____ 32. What is the energy required to remove an electron from an atom in the gaseous state called? a. nuclear energy c. shielding energy b. ionization energy d. electronegative energy ____ 33. For Group 2 (2A) metals, which electron is the most difficult to remove? a. the first b. the second c. th ...
... ____ 32. What is the energy required to remove an electron from an atom in the gaseous state called? a. nuclear energy c. shielding energy b. ionization energy d. electronegative energy ____ 33. For Group 2 (2A) metals, which electron is the most difficult to remove? a. the first b. the second c. th ...
Are there atoms in the air? Why or why not?
... How do we know where to put the elements? The elements are arranged based on their atomic number. What is that again? The atomic number is the number of protons in the ...
... How do we know where to put the elements? The elements are arranged based on their atomic number. What is that again? The atomic number is the number of protons in the ...
Groups and Families
... reactive metals, some Lanthanides andused to make steel Actinies: 2 rows Electrons in the Outer Shellat (Valence Electrons): 1 or 2 bottom of periodic Actinides: radioactive table Less to keep table (unstable) Elements after Reactivity: reactive than from being toooccur wide. plutonium do not in alk ...
... reactive metals, some Lanthanides andused to make steel Actinies: 2 rows Electrons in the Outer Shellat (Valence Electrons): 1 or 2 bottom of periodic Actinides: radioactive table Less to keep table (unstable) Elements after Reactivity: reactive than from being toooccur wide. plutonium do not in alk ...
The Periodic Table
... are very reactive and have a low melting point. Shade the alkali metals blue. 5. The nobel gases are found in group 18. Their outer energy level is filled, and they are very nonreactive, colorless gases. Shade the nobel gases red. 6. Halogens have seven electrons in the outer shell. They are located ...
... are very reactive and have a low melting point. Shade the alkali metals blue. 5. The nobel gases are found in group 18. Their outer energy level is filled, and they are very nonreactive, colorless gases. Shade the nobel gases red. 6. Halogens have seven electrons in the outer shell. They are located ...
Groups in a Periodic Table
... This is a shorter way to write the shorthand notation for electron configurations. There are four easy steps: 1.) Locate the element in the periodic table. 2.) Find the noble gas that precedes it. 3.) Place the symbol for this gas in brackets ([ ]). 4.) Write the remaining electron configuration. Wr ...
... This is a shorter way to write the shorthand notation for electron configurations. There are four easy steps: 1.) Locate the element in the periodic table. 2.) Find the noble gas that precedes it. 3.) Place the symbol for this gas in brackets ([ ]). 4.) Write the remaining electron configuration. Wr ...