6-1-Periodic Law
... known elements – 23 1864 John Newlands – 1st proposed organizational scheme for elements ...
... known elements – 23 1864 John Newlands – 1st proposed organizational scheme for elements ...
Ch. 5.1 History of the periodic table ppt.
... Mendeleev’s Periodic Table • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. – By 1886, these elements (scandium, galli ...
... Mendeleev’s Periodic Table • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. – By 1886, these elements (scandium, galli ...
The periodic table as we have it today has not always
... (Ca, Ba, Sr) were very similar. He also recognized that the atomic mass of Sr was about midway between that of Ca and Ba. He grouped these three elements together and called them a triad. ...
... (Ca, Ba, Sr) were very similar. He also recognized that the atomic mass of Sr was about midway between that of Ca and Ba. He grouped these three elements together and called them a triad. ...
The Periodic Table
... These are at the bottom of the table Lanthanides – shiny and reactive Actinides – radioactive and unstable ...
... These are at the bottom of the table Lanthanides – shiny and reactive Actinides – radioactive and unstable ...
Daily Inquiry: 10-31-2011
... Mendeleev’s Periodic Table • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. • By 1886, these elements (scandium, galli ...
... Mendeleev’s Periodic Table • In order for similar elements to line up, Mendeleev left gaps in his chart. • Mendeleev stated these were undiscovered elements. He made predictions about these undiscovered elements based on the other elements in the same row. • By 1886, these elements (scandium, galli ...
2.2 Periodic Chart
... The Alkaline Earth Family The alkaline earth metal family is less reactive than the alkali metals but alkaline earth metals burn brightly in air, some very colourful (strontium is used in red fireworks). Alkaline earth elements also react with water and are very common elements in earth rocks (calc ...
... The Alkaline Earth Family The alkaline earth metal family is less reactive than the alkali metals but alkaline earth metals burn brightly in air, some very colourful (strontium is used in red fireworks). Alkaline earth elements also react with water and are very common elements in earth rocks (calc ...
History of the Atom and Periodic Table
... Henry Moseley created the Modern Periodic Table of Elements that we use today. It is arranged by atomic number rather than mass. ...
... Henry Moseley created the Modern Periodic Table of Elements that we use today. It is arranged by atomic number rather than mass. ...
Lauren Gill O`Brien Grade 9 Science 4.1 Organizing the Elements
... Mendeleev – organized the known 64 elements into a periodic table which made the study of chemistry more manageable Mendeleev’s periodic law- if the elements are arranged according to their atomic mass, a pattern can be seen in which similar properties occur regularly ...
... Mendeleev – organized the known 64 elements into a periodic table which made the study of chemistry more manageable Mendeleev’s periodic law- if the elements are arranged according to their atomic mass, a pattern can be seen in which similar properties occur regularly ...
THE MINISTRY OF HIGHER AND SECONDARY SPECIAL
... characteristics of the elements began to repeat. The success of Mendeleev's table came from two decisions he made: The first was to leave gaps in the table when it seemed that the corresponding element had not yet been discovered. Mendeleev was not the first chemist to do so, but he was the first to ...
... characteristics of the elements began to repeat. The success of Mendeleev's table came from two decisions he made: The first was to leave gaps in the table when it seemed that the corresponding element had not yet been discovered. Mendeleev was not the first chemist to do so, but he was the first to ...
Periodic Table
... Properties are repeated in each period or row of the table. Elements are arranged by increasing atomic number – not mass as Mendeleev had previously thought. ...
... Properties are repeated in each period or row of the table. Elements are arranged by increasing atomic number – not mass as Mendeleev had previously thought. ...
Chapter Twelve: Atoms and the Periodic Table
... • Mendeleev arranged the elements in order of increasing mass so that elements with similar properties were in the same column. • Mendeleev used the properties of existing elements to predict properties of undiscovered elements. • The close match between Mendeleev’s predictions and the actual prope ...
... • Mendeleev arranged the elements in order of increasing mass so that elements with similar properties were in the same column. • Mendeleev used the properties of existing elements to predict properties of undiscovered elements. • The close match between Mendeleev’s predictions and the actual prope ...
Intro to Periodic Table and Lewis Structures
... • The modern PT is still organized in this way! ...
... • The modern PT is still organized in this way! ...
MENDELEEV`S PERIODIC TABLE
... grand plan called the periodic table. Mendeleev discovered (with a few exceptions) that arranging elements in order of increasing atomic mass across rows and similar characteric properties down the columns would result in elements obeying the periodic law (they repeated their characteristic proper ...
... grand plan called the periodic table. Mendeleev discovered (with a few exceptions) that arranging elements in order of increasing atomic mass across rows and similar characteric properties down the columns would result in elements obeying the periodic law (they repeated their characteristic proper ...
mendeleev*s periodic table
... grand plan called the periodic table. Mendeleev discovered (with a few exceptions) that arranging elements in order of increasing atomic mass across rows and similar characteric properties down the columns would result in elements obeying the periodic law (they repeated their characteristic proper ...
... grand plan called the periodic table. Mendeleev discovered (with a few exceptions) that arranging elements in order of increasing atomic mass across rows and similar characteric properties down the columns would result in elements obeying the periodic law (they repeated their characteristic proper ...
9The-Periodic-table1 (3).pptx
... ! Dmitri Mendeleev was a Russian chemist that classified elements by their properties ...
... ! Dmitri Mendeleev was a Russian chemist that classified elements by their properties ...
Dmitri MendeleevанааA Russian chemist, noticed a repeating
... Dmitri Mendeleev A Russian chemist, noticed a repeating pattern of chemical properties in the elements that were known at the time. Mendeleev arranged the elements in the order of increasing atomic mass to form something close to the modern day periodic table. The pattern of repeating ord ...
... Dmitri Mendeleev A Russian chemist, noticed a repeating pattern of chemical properties in the elements that were known at the time. Mendeleev arranged the elements in the order of increasing atomic mass to form something close to the modern day periodic table. The pattern of repeating ord ...
Periodic Table Notes Odysseyware Vocabulary the average relative
... In the late ______________the sixty-three known elements had been classified according to their properties, but they hadn’t been organized. Then, he (Mendeleev) began to lay the cards out according to increasing ________________ ____________________. The Modern Periodic Table Notice that the element ...
... In the late ______________the sixty-three known elements had been classified according to their properties, but they hadn’t been organized. Then, he (Mendeleev) began to lay the cards out according to increasing ________________ ____________________. The Modern Periodic Table Notice that the element ...
periodic table - rosedalegrade9chemistry
... known elements in the early 1800’s. Could the elements be organized based on properties like colour, smell or taste? Not really, because the characteristics or properties were not unique. Early scientists found a property unique to each element, atomic mass. ...
... known elements in the early 1800’s. Could the elements be organized based on properties like colour, smell or taste? Not really, because the characteristics or properties were not unique. Early scientists found a property unique to each element, atomic mass. ...
The Modern Periodic Table
... b. Mendeleev’s Periodic Table 1. Mendeleev’s Proposal Mendeleev arranged the elements into rows in order of increasing mass so that elements with similar properties were in the same column. 2. Mendeleev’s Prediction Mendeleev could not make a complete table of the elements because many elements had ...
... b. Mendeleev’s Periodic Table 1. Mendeleev’s Proposal Mendeleev arranged the elements into rows in order of increasing mass so that elements with similar properties were in the same column. 2. Mendeleev’s Prediction Mendeleev could not make a complete table of the elements because many elements had ...
Reading the Periodic table
... ▪ He was very accurate in his predictions, which led the world to accept his ideas about periodicity and a logical periodic table. ▪ Mendeleev wasn’t the first to arrange elements in a table, but he was the first to publish and make predictions about undiscovered elements ...
... ▪ He was very accurate in his predictions, which led the world to accept his ideas about periodicity and a logical periodic table. ▪ Mendeleev wasn’t the first to arrange elements in a table, but he was the first to publish and make predictions about undiscovered elements ...
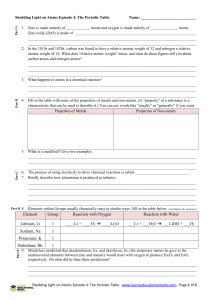
Element Group Reaction with Oxygen Reaction with Water Lithium
... Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2. In the 1810s and 1820s, carbon w ...
... Zinc oxide (ZnO) is made of ______________________________________________________________ ______________________________________________________________________________________ ______________________________________________________________________________________ 2. In the 1810s and 1820s, carbon w ...
Textbook Questions - Teach-n-Learn-Chem
... 36. The most electronegative element is ____________, with a value of ____. The least electronegative element is __________, with a value of ____. 37. How do we explain the trends that exist among elements’ properties? ...
... 36. The most electronegative element is ____________, with a value of ____. The least electronegative element is __________, with a value of ____. 37. How do we explain the trends that exist among elements’ properties? ...
1. In what order did Mendeleev arrange the elements in his periodic
... c) increasing number of neutrons d) increasing atomic weight 2. What family of elements was unknown when Mendeleev created the periodic table? a) noble gases b) alkali metals c) alkaline earth metals d) halogens 3. Mendeleev predicted the existence of which then unknown element? a) zinc b) silicon c ...
... c) increasing number of neutrons d) increasing atomic weight 2. What family of elements was unknown when Mendeleev created the periodic table? a) noble gases b) alkali metals c) alkaline earth metals d) halogens 3. Mendeleev predicted the existence of which then unknown element? a) zinc b) silicon c ...
Periodic Table cloze activity.
... metals, nonmetals, periodic, properties, symbol All matter is composed of various elements. An _________________ is a form of matter that is composed of a single type of _________________. In 1869, Dmitri _________________ created the _________________ table to group the elements. Periodic tables us ...
... metals, nonmetals, periodic, properties, symbol All matter is composed of various elements. An _________________ is a form of matter that is composed of a single type of _________________. In 1869, Dmitri _________________ created the _________________ table to group the elements. Periodic tables us ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.