Heavy Metals - High specific-gravity elements, such as arsenic
... and Lead (Pb), apparently because the relatively high density that they have in common, causes them to be toxic or poisonous at even low concentrations. This high density translates scientifically into a high specific-gravity (usually meaning, greater than 4.0). Sadly, it is this group of elements t ...
... and Lead (Pb), apparently because the relatively high density that they have in common, causes them to be toxic or poisonous at even low concentrations. This high density translates scientifically into a high specific-gravity (usually meaning, greater than 4.0). Sadly, it is this group of elements t ...
View PDF - CiteSeerX
... not be surprised that neither Mayer, Kelvin nor Thomson pointed out the similarity between the patterns of magnetised needles and the structure of the periodic system. Explicit references to the periodic system were rare in the 1870s, even in the chemistry literature, and only by the late 1880s had ...
... not be surprised that neither Mayer, Kelvin nor Thomson pointed out the similarity between the patterns of magnetised needles and the structure of the periodic system. Explicit references to the periodic system were rare in the 1870s, even in the chemistry literature, and only by the late 1880s had ...
Catalyst – September, 7(1+1) 2009 - stroh
... Key Point #5: Electronegativity DECREASES as you go DOWN the periodic table and INCREASES as you go LEFT TO RIGHT across the periodic table. ...
... Key Point #5: Electronegativity DECREASES as you go DOWN the periodic table and INCREASES as you go LEFT TO RIGHT across the periodic table. ...
classification of elements and periodicity in properties
... CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPER TIES ...
... CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPER TIES ...
Elements of the Periodic Table
... powerful refrigerant.On top of this neon is a rare non-toxic gas that appears in 18 parts per million of air. Xenon gives off a bright white light that is often used in commercial lighting applications including spotlights and movie projectors. Xenon can be used as a powerful anesthetic when inhaled ...
... powerful refrigerant.On top of this neon is a rare non-toxic gas that appears in 18 parts per million of air. Xenon gives off a bright white light that is often used in commercial lighting applications including spotlights and movie projectors. Xenon can be used as a powerful anesthetic when inhaled ...
Chapter 4 Elements and the Periodic Table The Periodic Table
... How did Mendeleev discover the pattern that led to the periodic table? ...
... How did Mendeleev discover the pattern that led to the periodic table? ...
Unit 3.pmd
... However, oxygen has lower ionisation enthalpy than nitrogen. Explain. 33. First member of each group of representative elements (i.e., s and p-block elements) shows anomalous behaviour. Illustrate with two examples. 34. p-Block elements form acidic, basic and amphoteric oxides. Explain each property ...
... However, oxygen has lower ionisation enthalpy than nitrogen. Explain. 33. First member of each group of representative elements (i.e., s and p-block elements) shows anomalous behaviour. Illustrate with two examples. 34. p-Block elements form acidic, basic and amphoteric oxides. Explain each property ...
NC SCS Chemistry
... The Organizing the Elements activity allows students to EXPLORE the arrangement of elements on the periodic table. It will provide students with the background on which they can better understand that elements are arranged in groups according to properties and atomic mass (Mendeleev) or atomic numbe ...
... The Organizing the Elements activity allows students to EXPLORE the arrangement of elements on the periodic table. It will provide students with the background on which they can better understand that elements are arranged in groups according to properties and atomic mass (Mendeleev) or atomic numbe ...
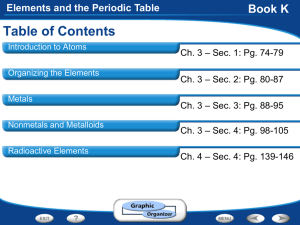
Elements and the Periodic Table
... Properties of Metals • The chemical property, reactivity, is the ease and speed with which an element combines or reacts with other elements or compounds. • The gradual wearing away of a metal element due to a chemical ...
... Properties of Metals • The chemical property, reactivity, is the ease and speed with which an element combines or reacts with other elements or compounds. • The gradual wearing away of a metal element due to a chemical ...
Electrons
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
Chapter 2 - Department of Chemistry and Physics
... All atoms of a given element have identical properties that differ from those of other elements. Atoms cannot be created, destroyed, or transformed into atoms of another element. Atoms of 2 or more elements can combine to form compounds. These combine in fixed proportions Chemical Reactions rearrang ...
... All atoms of a given element have identical properties that differ from those of other elements. Atoms cannot be created, destroyed, or transformed into atoms of another element. Atoms of 2 or more elements can combine to form compounds. These combine in fixed proportions Chemical Reactions rearrang ...
Slide 1
... radius increases as well. These trends hold for both positive and negative ions. Metals tend to lose one or more electrons and form a positive ion. As you move across a period, the ionic radii of metal cations tend to decrease because of the increasing nuclear charge. As you come to the nonmetal ele ...
... radius increases as well. These trends hold for both positive and negative ions. Metals tend to lose one or more electrons and form a positive ion. As you move across a period, the ionic radii of metal cations tend to decrease because of the increasing nuclear charge. As you come to the nonmetal ele ...
Periodicity Group Project
... The worksheet and or presentation should include questions that compare the following: H and Cs; S and F; Li. Si, and Te; Na and Cl. Worksheet should include drawings showing relative atomic radius and require students to write the Noble gas configurations. ...
... The worksheet and or presentation should include questions that compare the following: H and Cs; S and F; Li. Si, and Te; Na and Cl. Worksheet should include drawings showing relative atomic radius and require students to write the Noble gas configurations. ...
The Periodic Table
... The Periodic Table: Periods The periodic table provides information about the locations of electrons in an atomic of an element based on the period in which the element appears. There are seven periods in the periodic table. The two rows at the bottom of the table are actually parts of Period ...
... The Periodic Table: Periods The periodic table provides information about the locations of electrons in an atomic of an element based on the period in which the element appears. There are seven periods in the periodic table. The two rows at the bottom of the table are actually parts of Period ...
(Periodic Trends) - stroh
... T or F? Atomic size decreases as you move right across the periodic table. T or F? As you move down the Periodic Table, atoms get smaller. Rank the following sets of elements in order of increasing atomic size (small big). Set A: Bh, Mn, Re, Tc Set C: Y, Ti, Sg, Ta ...
... T or F? Atomic size decreases as you move right across the periodic table. T or F? As you move down the Periodic Table, atoms get smaller. Rank the following sets of elements in order of increasing atomic size (small big). Set A: Bh, Mn, Re, Tc Set C: Y, Ti, Sg, Ta ...
Electrons in Atoms - Effingham County Schools
... Name the block and group in which each of the following elements is located in the periodic table. Then, use the periodic table to name each element. Identify each element as a metal, nonmetal, or metalloid. Finally, describe whether each element has high reactivity or low reactivity ...
... Name the block and group in which each of the following elements is located in the periodic table. Then, use the periodic table to name each element. Identify each element as a metal, nonmetal, or metalloid. Finally, describe whether each element has high reactivity or low reactivity ...
Vol. 34. Main group chemist
... carbonyls, metal-to-metal bonds, metal-atom clusters, transition-metal compounds with bonds to hydrogen and carbon, and biological systems, are described in detail. There are some inconsistencies of presentation in the three volumes, and abbreviation of the ligands is not necessarily consistent with ...
... carbonyls, metal-to-metal bonds, metal-atom clusters, transition-metal compounds with bonds to hydrogen and carbon, and biological systems, are described in detail. There are some inconsistencies of presentation in the three volumes, and abbreviation of the ligands is not necessarily consistent with ...
Chapter 8. Chemistry of Main Group Elements
... Chapter 8. Chemistry of Main Group Elements 8.4.2 chemical Properties - generally, tend to loose two e- → noble gas electron config. good reducing agent - react with acid Mg + 2H+ → Mg2+ + H2 - react with water (Ca and heavier) Ca + 2H2O → Ca(OH)2 + H2 - Be: BeX2 has various form depend on the cond ...
... Chapter 8. Chemistry of Main Group Elements 8.4.2 chemical Properties - generally, tend to loose two e- → noble gas electron config. good reducing agent - react with acid Mg + 2H+ → Mg2+ + H2 - react with water (Ca and heavier) Ca + 2H2O → Ca(OH)2 + H2 - Be: BeX2 has various form depend on the cond ...
Unit 05 - Periodicity - Lincoln Park High School
... all elements had been discovered which would cause “holes” in any arrangement system. To see how this may have caused problems arrange the following eight symbols in the 3x3 grid provided. You must write the symbol and mass in each square. You should put the symbols in their appropriate boxes by con ...
... all elements had been discovered which would cause “holes” in any arrangement system. To see how this may have caused problems arrange the following eight symbols in the 3x3 grid provided. You must write the symbol and mass in each square. You should put the symbols in their appropriate boxes by con ...
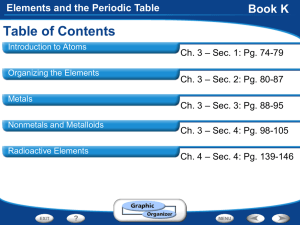
Elements and the Periodic Table
... Properties of Metals • The chemical property, reactivity, is the ease and speed with which an element combines or reacts with other elements or compounds. • The gradual wearing away of a metal element due to a chemical ...
... Properties of Metals • The chemical property, reactivity, is the ease and speed with which an element combines or reacts with other elements or compounds. • The gradual wearing away of a metal element due to a chemical ...
Periodic Classification of Elements
... If the elements are arranged sequentially in the increasing order of their atomic weights, every eighth element is having the similar properties to that of the first element like in the octave of music. This is called "Newland's Concept of Octaves". Eg: 1 ...
... If the elements are arranged sequentially in the increasing order of their atomic weights, every eighth element is having the similar properties to that of the first element like in the octave of music. This is called "Newland's Concept of Octaves". Eg: 1 ...
GCSE Chemistry Specimen Question Paper Higher Specimen
... The student did a test to find which metal ion was in the anti-caking compound. The student had not seen any red colour in the flame while doing the flame test. The student added water to make a solution of Low Sodium Salt. The student then added sodium hydroxide solution. A white precipitate formed ...
... The student did a test to find which metal ion was in the anti-caking compound. The student had not seen any red colour in the flame while doing the flame test. The student added water to make a solution of Low Sodium Salt. The student then added sodium hydroxide solution. A white precipitate formed ...
Addrienne`s Element Lesson Plan
... according to the same rules they used. The rows of the periodic table are called “periods”, and the columns are called “groups”. Since elements in the same group have the same number of valence electrons, they react in similar ways. This is why elements in the same groups have similar properties. As ...
... according to the same rules they used. The rows of the periodic table are called “periods”, and the columns are called “groups”. Since elements in the same group have the same number of valence electrons, they react in similar ways. This is why elements in the same groups have similar properties. As ...
Dmitri Mendeleev

Dmitri Ivanovich Mendeleev (/ˌmɛndəlˈeɪəf/; Russian: Дми́трий Ива́нович Менделе́ев; IPA: [ˈdmʲitrʲɪj ɪˈvanəvʲɪtɕ mʲɪndʲɪˈlʲejɪf]; 8 February 1834 – 2 February 1907 O.S. 27 January 1834 – 20 January 1907) was a Russian chemist and inventor. He formulated the Periodic Law, created his own version of the periodic table of elements, and used it to correct the properties of some already discovered elements and also to predict the properties of eight elements yet to be discovered.