Chem 200 Dr. Saidane
... a) The Law of Conservation of Mass, which states that mass is neither destroyed nor created during ordinary chemical reactions. b) The Law of Definite Proportions, which states that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the s ...
... a) The Law of Conservation of Mass, which states that mass is neither destroyed nor created during ordinary chemical reactions. b) The Law of Definite Proportions, which states that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the s ...
Chapter 10 Test A
... 1. This scientist published a detailed atomic theory in 1808 based on evidence he gathered through experiments with gases. His atomic theory laid the groundwork for later atomic models. a. James Chadwick b. John Dalton c. Ernest Rutherford d. J. J. Thomson 2. One kind of particle that makes up the a ...
... 1. This scientist published a detailed atomic theory in 1808 based on evidence he gathered through experiments with gases. His atomic theory laid the groundwork for later atomic models. a. James Chadwick b. John Dalton c. Ernest Rutherford d. J. J. Thomson 2. One kind of particle that makes up the a ...
Particular particle knowledge
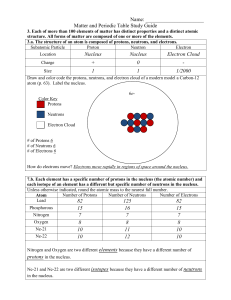
... 13. These atoms are called ......................................................................... 14. The nuclei of six unidentified elements are shown below. Circle the ones which are isotopes of each other. ...
... 13. These atoms are called ......................................................................... 14. The nuclei of six unidentified elements are shown below. Circle the ones which are isotopes of each other. ...
and the atomic
... • this is NOT IB material until Rutherford • it is very interesting from a geeky-science stand point • it will help you understand and appreciate the structure of the atom • you are not responsible for knowing the information from all thescientists ...
... • this is NOT IB material until Rutherford • it is very interesting from a geeky-science stand point • it will help you understand and appreciate the structure of the atom • you are not responsible for knowing the information from all thescientists ...
Chapter 3
... 3. Atoms cannot be created, destroyed, or transformed into atoms of another element. 4. Compounds are formed when atoms of different elements combine with one another in small wholenumber ratios. 5. The relative numbers and kinds of atoms are constant in a given compound. ...
... 3. Atoms cannot be created, destroyed, or transformed into atoms of another element. 4. Compounds are formed when atoms of different elements combine with one another in small wholenumber ratios. 5. The relative numbers and kinds of atoms are constant in a given compound. ...
are made up of
... Severalscientists,including Newlands, Meyer, and Mendeleevworked on classificationsystems that grouped elements accordingto their properties. They found that these properties repeated in a regular or periodic manner. This fact was used to predict properties of undiscovered elements. Reviewelectron a ...
... Severalscientists,including Newlands, Meyer, and Mendeleevworked on classificationsystems that grouped elements accordingto their properties. They found that these properties repeated in a regular or periodic manner. This fact was used to predict properties of undiscovered elements. Reviewelectron a ...
Atoms and Their Electrons
... number of protons and the neutrons of an atom together. An element must have a certain number of protons but it can have a range of numbers of neutrons i.e. hydrogen can have 0, 1, or 2 neutrons to go with its 1 proton. These are called isotopes of hydrogen In real life there may be a number of diff ...
... number of protons and the neutrons of an atom together. An element must have a certain number of protons but it can have a range of numbers of neutrons i.e. hydrogen can have 0, 1, or 2 neutrons to go with its 1 proton. These are called isotopes of hydrogen In real life there may be a number of diff ...
Atomic Theory
... was continuous-did not believe in atoms • His opinion was accepted for nearly 2000 years ...
... was continuous-did not believe in atoms • His opinion was accepted for nearly 2000 years ...
14_1_atoms and isotopes FPS3
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
Matter and the Periodic Table Study Guide Answer Key
... Semimetals/Metalloids have properties of both metals and non-metals. 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound ...
... Semimetals/Metalloids have properties of both metals and non-metals. 3.b. Compounds are formed by combining two or more different elements and compounds have properties that are different from their constituent elements. 3.f. Use the periodic table to identify elements in simple compounds. Compound ...
Lecture 2: Atoms - U of L Class Index
... Mass number (A) = # protons + # neutrons Atomic number (Z) = # protons ...
... Mass number (A) = # protons + # neutrons Atomic number (Z) = # protons ...
nuclear chemistry - Wood County Schools
... • Usually accomplished by bombarding a nucleus with a neutron ...
... • Usually accomplished by bombarding a nucleus with a neutron ...
electrons
... particles, not light. He envisioned an atom as a sphere of positive charge with negatively charged electrons spread out evenly within. This is sometimes called The ...
... particles, not light. He envisioned an atom as a sphere of positive charge with negatively charged electrons spread out evenly within. This is sometimes called The ...
12.1 Atoms and Isotopes
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
CHAPTER 4 EXAM: THE NATURE OF THE ATOM (modified)
... throughout he called the Plum Pudding Model? a. Thomson c. Bohr b. Chadwick 4. Which scientist proved the existence of the neutron? a. Thomson c. Chadwick b. Bohr 5. Which is the number of neutrons in ...
... throughout he called the Plum Pudding Model? a. Thomson c. Bohr b. Chadwick 4. Which scientist proved the existence of the neutron? a. Thomson c. Chadwick b. Bohr 5. Which is the number of neutrons in ...
atomic number
... • The mass number of an atom is the sum of the number of protons and neutrons in an atom. Mass number = number of protons + number of neutrons ...
... • The mass number of an atom is the sum of the number of protons and neutrons in an atom. Mass number = number of protons + number of neutrons ...
What does an elements atomic mass tell us about the element?
... Potassium - K Atomic # = 19 Mass # = 39 K nucleus contains 19 protons 39 – 19 = 20 neutrons How many electrons? Same as # Protons (19) ...
... Potassium - K Atomic # = 19 Mass # = 39 K nucleus contains 19 protons 39 – 19 = 20 neutrons How many electrons? Same as # Protons (19) ...
1.1 Guided Reading
... 9. The symbol for lead is ______, and the symbol for iron is ________. The symbols of these elements come from _______________names. Each element is made of a different atom. (page 11) 10. What 3 ideas did 1800s British scientist John Dalton propose? A. B. C. 11. Which part (see #10) of Dalton’s the ...
... 9. The symbol for lead is ______, and the symbol for iron is ________. The symbols of these elements come from _______________names. Each element is made of a different atom. (page 11) 10. What 3 ideas did 1800s British scientist John Dalton propose? A. B. C. 11. Which part (see #10) of Dalton’s the ...
Ch. 6 Vocabulary
... • atoms of the same element that have the same number of protons but a different number of neutrons ...
... • atoms of the same element that have the same number of protons but a different number of neutrons ...
Element: a pure, simple substance that can`t be broken down into
... What is the smallest unit of matter that we can find everywhere, even in tuna fish? What charge do electrons have? What are elements? Who organized the atomic elements? What do we call a horizontal row on the periodic table? What do we call the vertical columns on the periodic table? The number of p ...
... What is the smallest unit of matter that we can find everywhere, even in tuna fish? What charge do electrons have? What are elements? Who organized the atomic elements? What do we call a horizontal row on the periodic table? What do we call the vertical columns on the periodic table? The number of p ...
800 - Paint Valley Local Schools
... considered radioactive because of its large, unstable nucleus. It was one of the fuels used to construct the early atomic bombs in the WWII era. ...
... considered radioactive because of its large, unstable nucleus. It was one of the fuels used to construct the early atomic bombs in the WWII era. ...
Chapter 7
... Rutherford model of the atom The most direct way to probe the structure of a fruitcake would be to stick a finger in it as a probe. In 1911 a New Zealand scientist named Ernest Rutherford proposed an experiment to probe the structure of an atom. A few years earlier it was discovered that some minera ...
... Rutherford model of the atom The most direct way to probe the structure of a fruitcake would be to stick a finger in it as a probe. In 1911 a New Zealand scientist named Ernest Rutherford proposed an experiment to probe the structure of an atom. A few years earlier it was discovered that some minera ...
10B Atoms and Isotopes
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
... You have learned that atoms contain three smaller particles called protons, neutrons, and electrons, and that the number of protons determines the type of atom. How can you figure out how many neutrons an atom contains, and whether it is neutral or has a charge? Once you know how many protons and ne ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.
![Atomic Structure [PowerPoint]](http://s1.studyres.com/store/data/000122096_1-1d100da6540d2f26db122fc51f672fe5-300x300.png)