Structure of the Atom
... experiment is carried out using a foil of a metal other than gold? Answer: If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin f ...
... experiment is carried out using a foil of a metal other than gold? Answer: If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin f ...
Atomic Structure - Monona Grove School District
... each of these domains can be predicted from day to day. Most biorhythm models use three cycles: a 23-day "physical" cycle, a 28-day "emotional" cycle, and a 33-day "intellectual" cycle. Although the 28-day cycle is the same length as the average woman's menstrual cycle and was originally described a ...
... each of these domains can be predicted from day to day. Most biorhythm models use three cycles: a 23-day "physical" cycle, a 28-day "emotional" cycle, and a 33-day "intellectual" cycle. Although the 28-day cycle is the same length as the average woman's menstrual cycle and was originally described a ...
Atoms and - 4LTR Press
... Scientists now have experimental evidence for the existence of more than 60 subatomic particles. However, only three are important to our understanding of the chemical view of matter: protons and neutrons, found in the nucleus of the atom, and electrons, found outside the nucleus (Table 3.1). The ma ...
... Scientists now have experimental evidence for the existence of more than 60 subatomic particles. However, only three are important to our understanding of the chemical view of matter: protons and neutrons, found in the nucleus of the atom, and electrons, found outside the nucleus (Table 3.1). The ma ...
Elements and the Periodic Table
... • Elements with atomic numbers higher than 92 are sometimes described as synthetic elements because they are not found naturally on Earth. • Instead, elements that follow uranium are made – or synthesized – when nuclear particles are forced to crash into one another. • To make even heavier elements ...
... • Elements with atomic numbers higher than 92 are sometimes described as synthetic elements because they are not found naturally on Earth. • Instead, elements that follow uranium are made – or synthesized – when nuclear particles are forced to crash into one another. • To make even heavier elements ...
EARLY ATOMIC THEORY AND STRUCTURE
... 1. The formula for water is H2O. There is one atom of oxygen for every two atoms of hydrogen. The molar mass of oxygen is 16.00 g and the molar mass of hydrogen is 1.008 g. For H2O the mass of two hydrogen atoms is 2.016 g and the mass of one oxygen atom is 16.00 g. The ratio of hydrogen to oxygen i ...
... 1. The formula for water is H2O. There is one atom of oxygen for every two atoms of hydrogen. The molar mass of oxygen is 16.00 g and the molar mass of hydrogen is 1.008 g. For H2O the mass of two hydrogen atoms is 2.016 g and the mass of one oxygen atom is 16.00 g. The ratio of hydrogen to oxygen i ...
Chemistry 139
... (2 pts) One of the main features of Dalton's atomic theory which is no longer considered valid is: All atoms of each element are identical in every respect. Which of the following is the best explanation of why this feature is no longer considered valid? ...
... (2 pts) One of the main features of Dalton's atomic theory which is no longer considered valid is: All atoms of each element are identical in every respect. Which of the following is the best explanation of why this feature is no longer considered valid? ...
Unit 2 Atomic Theories and Structures Packet
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
Unit 2 - Solon City Schools
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
... 14)______________________ A positively charged particle. 15)______________________ Chemical compounds have the same elements in exactly the same ratios. 16)______________________ This English scientist is credited with discovering the neutron. 17)______________________ It represents the number of pr ...
Redox Reactions - Hillsborough County Public Schools
... oxygen normally has an oxidation number of -2 and there are two oxygen atoms. 2(-2) = -4. The total number of negative charges is 4 negatives. The only other atom that is present is nitrogen. That means the nitrogen is responsible for all for the positive charge. X + -4 = 0. X = +4. Therefore, the o ...
... oxygen normally has an oxidation number of -2 and there are two oxygen atoms. 2(-2) = -4. The total number of negative charges is 4 negatives. The only other atom that is present is nitrogen. That means the nitrogen is responsible for all for the positive charge. X + -4 = 0. X = +4. Therefore, the o ...
EARLY ATOMIC THEORY AND STRUCTURE
... Atoms may combine in different ratios to form more than one compound. ...
... Atoms may combine in different ratios to form more than one compound. ...
Atomic Structure Practice Test
... 7. Because most particles fired at metal foil passed straight through, Rutherford concluded that a. atoms were mostly empty space. c. electrons formed the nucleus. b. atoms contained no charged particles. d. atoms were indivisible. ...
... 7. Because most particles fired at metal foil passed straight through, Rutherford concluded that a. atoms were mostly empty space. c. electrons formed the nucleus. b. atoms contained no charged particles. d. atoms were indivisible. ...
Atomic Structure Practice Test
... 7. Because most particles fired at metal foil passed straight through, Rutherford concluded that a. atoms were mostly empty space. c. electrons formed the nucleus. b. atoms contained no charged particles. d. atoms were indivisible. ...
... 7. Because most particles fired at metal foil passed straight through, Rutherford concluded that a. atoms were mostly empty space. c. electrons formed the nucleus. b. atoms contained no charged particles. d. atoms were indivisible. ...
atom
... *Electrons are negatively charged subatomic particles. *They were primarily the interest of electricians rather than chemist. *These scientists (i.e. electricians) studied the flow of gases in an enclosed tube…at low pressure. The tube had two metal plates at each end. One of the plates held a posit ...
... *Electrons are negatively charged subatomic particles. *They were primarily the interest of electricians rather than chemist. *These scientists (i.e. electricians) studied the flow of gases in an enclosed tube…at low pressure. The tube had two metal plates at each end. One of the plates held a posit ...
Atoms and the Periodic Table
... *1. MASS of an ELECTRON is 1,860 times LESS than a PROTON or NEUTRON *2. (e.g.) lithium [Li]: protons = 3+; electrons = 3-; neutrons = 4 = 7 u (or 7 amu) ...
... *1. MASS of an ELECTRON is 1,860 times LESS than a PROTON or NEUTRON *2. (e.g.) lithium [Li]: protons = 3+; electrons = 3-; neutrons = 4 = 7 u (or 7 amu) ...
The Structure of the Atom 4
... identical in size, mass, and chemical properties. Atoms of a specific element are different from atoms of another element. Different atoms combine in simple whole-number ratios to form compound. In a chemical reaction, atoms are ...
... identical in size, mass, and chemical properties. Atoms of a specific element are different from atoms of another element. Different atoms combine in simple whole-number ratios to form compound. In a chemical reaction, atoms are ...
Atoms and Molecules
... properties. Mendeleyev was able to organize the table in its present form even though many of the elements hadn’t been discovered yet. Although Mendeleev is credited with developing the Periodic Table, many scientists contributed to its development. The organization was done in such a way that as ne ...
... properties. Mendeleyev was able to organize the table in its present form even though many of the elements hadn’t been discovered yet. Although Mendeleev is credited with developing the Periodic Table, many scientists contributed to its development. The organization was done in such a way that as ne ...
study of the contribution of nobel prize winners to the development
... radioactive elements (1903). In accordance with this theory, some of the heaviest elements acquire resistance, throwing small but sufficiently scattered units of mass, charge and energy out of their nuclei in the form of alpha-, beta- and gamma-radiation. The other elements are formed in the process ...
... radioactive elements (1903). In accordance with this theory, some of the heaviest elements acquire resistance, throwing small but sufficiently scattered units of mass, charge and energy out of their nuclei in the form of alpha-, beta- and gamma-radiation. The other elements are formed in the process ...
Atoms and Molecules
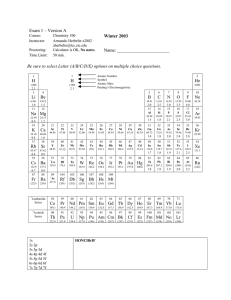
... Q: How many elements are there? A: There are 117 known elements. 90 of them are naturally occurring elements, and scientists have been able to create 27 more in the laboratory. Note: In 1869, Dmitri Mendeleyev was credited with putting together the Periodic Table of Elements. He listed all of the kn ...
... Q: How many elements are there? A: There are 117 known elements. 90 of them are naturally occurring elements, and scientists have been able to create 27 more in the laboratory. Note: In 1869, Dmitri Mendeleyev was credited with putting together the Periodic Table of Elements. He listed all of the kn ...
Class IX Chapter 4 – Structure of the Atom Science
... If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin foil of gold can be easily made. It is difficult to make such foils from oth ...
... If the α-scattering experiment is carried out using a foil of a metal rather than gold, there would be no change in the observation. In the α-scattering experiment, a gold foil was taken because gold is malleable and a thin foil of gold can be easily made. It is difficult to make such foils from oth ...
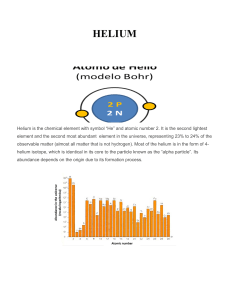
HELIUM - IDC
... Helium is believed to be formed during primordial nucleosynthesis, at 3 minutes and 45 seconds after the start of the universe when the universe expansion cooled enough to 1 billion of ° K and finally the 2hydrogen isotopes could be formed initiating a rapid sequence of nuclear reactions into 3-Heli ...
... Helium is believed to be formed during primordial nucleosynthesis, at 3 minutes and 45 seconds after the start of the universe when the universe expansion cooled enough to 1 billion of ° K and finally the 2hydrogen isotopes could be formed initiating a rapid sequence of nuclear reactions into 3-Heli ...
DEFINING THE ATOM - BradyMathScience
... ________ 14. The number of neutrons in the nucleus of an atom can be calculated by a. adding together the numbers of electrons and protons. b. subtracting the number of protons from the number of electrons. c. subtracting the number of protons from the mass number. d. adding the mass number to the n ...
... ________ 14. The number of neutrons in the nucleus of an atom can be calculated by a. adding together the numbers of electrons and protons. b. subtracting the number of protons from the number of electrons. c. subtracting the number of protons from the mass number. d. adding the mass number to the n ...
FREE Sample Here
... Use the periodic table to find the element corresponding to each symbol. a. K2Cr2O7 is made up of 2 atoms of K (potassium), 2 atoms of Cr (chromium) and 7 atoms of O (oxygen). b. C5H8NNaO4 is made up of 5 atoms of C (carbon), 8 atoms of H (hydrogen), 1 atom of N (nitrogen), 1 atom of Na (sodium) and ...
... Use the periodic table to find the element corresponding to each symbol. a. K2Cr2O7 is made up of 2 atoms of K (potassium), 2 atoms of Cr (chromium) and 7 atoms of O (oxygen). b. C5H8NNaO4 is made up of 5 atoms of C (carbon), 8 atoms of H (hydrogen), 1 atom of N (nitrogen), 1 atom of Na (sodium) and ...
Preview Sample 1
... Use the periodic table to find the element corresponding to each symbol. a. K2Cr2O7 is made up of 2 atoms of K (potassium), 2 atoms of Cr (chromium) and 7 atoms of O (oxygen). b. C5H8NNaO4 is made up of 5 atoms of C (carbon), 8 atoms of H (hydrogen), 1 atom of N (nitrogen), 1 atom of Na (sodium) and ...
... Use the periodic table to find the element corresponding to each symbol. a. K2Cr2O7 is made up of 2 atoms of K (potassium), 2 atoms of Cr (chromium) and 7 atoms of O (oxygen). b. C5H8NNaO4 is made up of 5 atoms of C (carbon), 8 atoms of H (hydrogen), 1 atom of N (nitrogen), 1 atom of Na (sodium) and ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.