Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
Chapter 2: Atoms, Ions, and the Periodic Table
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
... A) Pure water is composed of the elements oxygen and hydrogen in a mass ratio of 8 to 1. B) Any sample of a given compound always contains the same proportions by mass of the component elements. C) The mass of the products of a chemical reaction is equal to the mass of the starting materials of the ...
Incorrect…try again
... • 8 is the number of neutrons. This does not indicate element name. You need to look at the protons to determine the element name. • Mass # is found by adding protons and neutrons • 12 is only the number of neutrons. You must add the protons to this number. ...
... • 8 is the number of neutrons. This does not indicate element name. You need to look at the protons to determine the element name. • Mass # is found by adding protons and neutrons • 12 is only the number of neutrons. You must add the protons to this number. ...
Atomic Theory and Periodic Table Review Multiple Choice Identify
... a. Protons, neutrons, and electrons all have about the same mass. b. Unlike protons or neutrons, electrons have no mass. c. Neutrons have no charge and no mass. d. An electron has far less mass than either a proton or neutron. 10. Which of the following is unique for any given element? a. the number ...
... a. Protons, neutrons, and electrons all have about the same mass. b. Unlike protons or neutrons, electrons have no mass. c. Neutrons have no charge and no mass. d. An electron has far less mass than either a proton or neutron. 10. Which of the following is unique for any given element? a. the number ...
Electron - CoolHub
... Tell students that this is the periodic table. Explain that each box contains information about a different atom. The periodic table shows all the atoms that everything in the known universe is made from. It’s kind of like the alphabet in which only 26 letters, in different combinations, make up man ...
... Tell students that this is the periodic table. Explain that each box contains information about a different atom. The periodic table shows all the atoms that everything in the known universe is made from. It’s kind of like the alphabet in which only 26 letters, in different combinations, make up man ...
The Masses of Atoms
... I ask the indulgence of chemists and physicist for the approach I have taken with this book. The simplifications and short cuts that I have taken with their subjects, the anecdotal nature of this book as well as its cartoons, all have one sole aim: to allow a reader, who has not done much chemistry, ...
... I ask the indulgence of chemists and physicist for the approach I have taken with this book. The simplifications and short cuts that I have taken with their subjects, the anecdotal nature of this book as well as its cartoons, all have one sole aim: to allow a reader, who has not done much chemistry, ...
Elements and the Periodic Table
... Rutherford not only discovered that the nucleus of an atom occupies a very tiny volume at the centre of the atom. He also discovered many important characteristics of the components that make up the nucleus. The results of his research are discussed below. Protons and Neutrons After Thomson discover ...
... Rutherford not only discovered that the nucleus of an atom occupies a very tiny volume at the centre of the atom. He also discovered many important characteristics of the components that make up the nucleus. The results of his research are discussed below. Protons and Neutrons After Thomson discover ...
Chapter 4 Elements and the Periodic Table
... compounds by sharing or gaining one electron when reacting with atoms of other elements. ...
... compounds by sharing or gaining one electron when reacting with atoms of other elements. ...
Chapter 4 Elements and the Periodic Table
... compounds by sharing or gaining one electron when reacting with atoms of other elements. ...
... compounds by sharing or gaining one electron when reacting with atoms of other elements. ...
Chromatographic Enrichment of Lithium Isotopes by Hydrous
... lithium, 7Li could be used as a pH control agent (7LiOH) of the coolant in nuclear fission reactors. And the lithium compounds rich in 6Li will be required for the tritium breeder blanket in deuterium-tritium fusion power reactors in the future.7 Because of the large cross section for thermal neutro ...
... lithium, 7Li could be used as a pH control agent (7LiOH) of the coolant in nuclear fission reactors. And the lithium compounds rich in 6Li will be required for the tritium breeder blanket in deuterium-tritium fusion power reactors in the future.7 Because of the large cross section for thermal neutro ...
atoms - WordPress.com
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
Nucleon number
... (a) Proton number = 8, so there are 8 protons. Nucleon number = 17, so the number of neutrons is 17-8 = 9. The number of electrons is the same as the number of protons, that is 8. (b) Proton numbers = 80, so there are 80 protons. Nucleon number = 199, so the number of neutrons is 199-80 = 119. The n ...
... (a) Proton number = 8, so there are 8 protons. Nucleon number = 17, so the number of neutrons is 17-8 = 9. The number of electrons is the same as the number of protons, that is 8. (b) Proton numbers = 80, so there are 80 protons. Nucleon number = 199, so the number of neutrons is 199-80 = 119. The n ...
FREE Sample Here
... 1) According to history, the concept that all matter is composed of atoms was first proposed by A) the Greek philosopher Democritus, but not widely accepted until modern times. B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einste ...
... 1) According to history, the concept that all matter is composed of atoms was first proposed by A) the Greek philosopher Democritus, but not widely accepted until modern times. B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einste ...
General Chemistry: Atoms First (McMurry/Fay/Pribush)
... Chapter 2 The Structure and Stability of Atoms 2.1 Multiple Choice Questions 1) According to history, the concept that all matter is composed of atoms was first proposed by A) the Greek philosopher Democritus, but not widely accepted until modern times. B) Dalton, but not widely accepted until the w ...
... Chapter 2 The Structure and Stability of Atoms 2.1 Multiple Choice Questions 1) According to history, the concept that all matter is composed of atoms was first proposed by A) the Greek philosopher Democritus, but not widely accepted until modern times. B) Dalton, but not widely accepted until the w ...
Chemical Foundations: Elements, Atoms, and Ions
... Fortunately for those trying to understand and systematize it, nature often uses a relatively small number of fundamental units to assemble even extremely complex materials. For example, proteins, a group of substances that serve the human body in almost uncountable ways, are all made by linking tog ...
... Fortunately for those trying to understand and systematize it, nature often uses a relatively small number of fundamental units to assemble even extremely complex materials. For example, proteins, a group of substances that serve the human body in almost uncountable ways, are all made by linking tog ...
The d- and f- Block Element Block Elements The d- and f
... The d orbitals of the transition elements project to the periphery of an atom more than the other orbitals (i.e., s and p), hence, they are more influenced by the surroundings as well as affecting the atoms or molecules n surrounding them. In some respects, ions of a given d configuration (n = 1 – 9 ...
... The d orbitals of the transition elements project to the periphery of an atom more than the other orbitals (i.e., s and p), hence, they are more influenced by the surroundings as well as affecting the atoms or molecules n surrounding them. In some respects, ions of a given d configuration (n = 1 – 9 ...
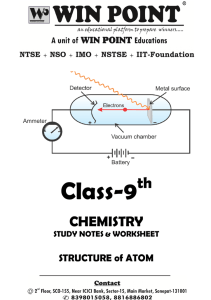
Chapter 4: The Structure of the Atom
... began to make connections between matter and electric charge. For instance, has your hair ever clung to your comb? To explore the connection, some scientists wondered how electricity might behave in the absence of matter. With the help of the newly invented vacuum pump, they passed electricity throu ...
... began to make connections between matter and electric charge. For instance, has your hair ever clung to your comb? To explore the connection, some scientists wondered how electricity might behave in the absence of matter. With the help of the newly invented vacuum pump, they passed electricity throu ...
Atomic Structure
... observing the averaged properties of all the different isotopes in the sample. Luckily, aside from having different masses, most other properties of different isotopes are similar. Knowing about the different isotopes is important when it comes to calculating atomic mass. The atomic mass (sometimes ...
... observing the averaged properties of all the different isotopes in the sample. Luckily, aside from having different masses, most other properties of different isotopes are similar. Knowing about the different isotopes is important when it comes to calculating atomic mass. The atomic mass (sometimes ...
NUCL 1 Early life of Albert Ghiorso: Preparation for future role as
... Although young Albert was a good student in classical subjects, he early on showed his mechanical ability. His family lived close to the Oakland Airport and Albert set his sights on becoming an aeronautical engineer. When he graduated from high school in 1932 in the depth of the Great Depression he ...
... Although young Albert was a good student in classical subjects, he early on showed his mechanical ability. His family lived close to the Oakland Airport and Albert set his sights on becoming an aeronautical engineer. When he graduated from high school in 1932 in the depth of the Great Depression he ...
Chapter 4: The Structure of the Atom
... research, he proposed his atomic theory in 1803. In many ways Democritus’s and Dalton’s theories are similar. What similarities and differences can you find between the two theories? Recall from Chapter 3 that the law of conservation of mass states that mass is conserved in any process, such as a ch ...
... research, he proposed his atomic theory in 1803. In many ways Democritus’s and Dalton’s theories are similar. What similarities and differences can you find between the two theories? Recall from Chapter 3 that the law of conservation of mass states that mass is conserved in any process, such as a ch ...
File
... In 1830, Michael Faraday showed that if electricity is passed through a solution of an electrolyte, chemical reactions occurred at the electrodes, which resulted in the liberation and deposition of matter at the electrodes. In mid 1850s many scientists mainly Faraday began to study electrical discha ...
... In 1830, Michael Faraday showed that if electricity is passed through a solution of an electrolyte, chemical reactions occurred at the electrodes, which resulted in the liberation and deposition of matter at the electrodes. In mid 1850s many scientists mainly Faraday began to study electrical discha ...
Acquiring the Foundation: The Periodic Table for Middle
... animated periodic table of the first eighteen elements on the periodic table of elements. The animated periodic table of elements will be organized according to the same principles Mendeleyev used when he placed elements in the original periodic table. This unit will help students build a strong fou ...
... animated periodic table of the first eighteen elements on the periodic table of elements. The animated periodic table of elements will be organized according to the same principles Mendeleyev used when he placed elements in the original periodic table. This unit will help students build a strong fou ...
Chapter 2 "Elements, Atoms, and the Periodic Table"
... 2.3 The Structure of Atoms LEARNING OBJECTIVES 1. Describe the three main subatomic particles. 2. State how the subatomic particles are arranged in atoms. ...
... 2.3 The Structure of Atoms LEARNING OBJECTIVES 1. Describe the three main subatomic particles. 2. State how the subatomic particles are arranged in atoms. ...
Mendeleev`s Periodic Table
... The Figure above also shows that there are distinct peaks in the total electron density at particular distances and that these peaks occur at different distances from the nucleus for each element. Each peak in a given plot corresponds to the electron density in a given principal shell. Because heli ...
... The Figure above also shows that there are distinct peaks in the total electron density at particular distances and that these peaks occur at different distances from the nucleus for each element. Each peak in a given plot corresponds to the electron density in a given principal shell. Because heli ...
Neptunium
.png?width=300)
Neptunium is a chemical element with symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, pyrophoric, and can accumulate in bones, which makes the handling of neptunium dangerous.Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear reactors. The vast majority is generated as a by-product in conventional nuclear power reactors. While neptunium itself has no commercial uses at present, it is widely used as a precursor for the formation of plutonium-238, used in radioisotope thermal generators. Neptunium has also been used in detectors of high-energy neutrons.The most stable isotope of neptunium, neptunium-237, is a by-product of nuclear reactors and plutonium production. It, and the isotope neptunium-239, are also found in trace amounts in uranium ores due to neutron capture reactions and beta decay.