Acids and bases
... much larger than the size of the molecules themselves. 2. Particles in a gas move in straight line paths and random directions. 3. Particles in a gas collide frequently with the sides of the container and less frequently with each other. All collisions are elastic (no energy is gained or lost as a r ...
... much larger than the size of the molecules themselves. 2. Particles in a gas move in straight line paths and random directions. 3. Particles in a gas collide frequently with the sides of the container and less frequently with each other. All collisions are elastic (no energy is gained or lost as a r ...
Name
... The exchange of nitrogen between the Earth and the atmosphere. Nitrogen is essential to all living systems. To become a part of an organism, nitrogen must first be combined or fixed with oxygen or hydrogen. Nitrogen is removed from the atmosphere by nitrogen fixing bacteria located in the roots of ...
... The exchange of nitrogen between the Earth and the atmosphere. Nitrogen is essential to all living systems. To become a part of an organism, nitrogen must first be combined or fixed with oxygen or hydrogen. Nitrogen is removed from the atmosphere by nitrogen fixing bacteria located in the roots of ...
Honors Chemistry II Review 1. Express the following in scientific
... Which reactant is limiting if there are 80kg of water to be removed and 65kg of Li2O available? How many kg of the excess reactant remain? 20. After lithium hydroxide is produced aboard the space shuttle, it is used to remove exhaled carbon dioxide from the air supply according to the following equa ...
... Which reactant is limiting if there are 80kg of water to be removed and 65kg of Li2O available? How many kg of the excess reactant remain? 20. After lithium hydroxide is produced aboard the space shuttle, it is used to remove exhaled carbon dioxide from the air supply according to the following equa ...
1. Global Warming terms: Greenhouse effect: the process by which
... and other fuels and from certain industrial processes. When these chemicals are released from power plants and other sources, winds blow them far from their source. If the acid chemicals in the air are blown into areas where the weather is wet, the acids can fall to earth in rain, snow, fog, mist. I ...
... and other fuels and from certain industrial processes. When these chemicals are released from power plants and other sources, winds blow them far from their source. If the acid chemicals in the air are blown into areas where the weather is wet, the acids can fall to earth in rain, snow, fog, mist. I ...
What are reactions? - UTLNET Secure Site
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
... If they are formed in a reaction you will see __________. This can be a sign that a chemical __________ has happened. 2. Other signs of a chemical reaction might be an increase in temperature if _____ is released or a change in ________. 3. Physical changes like _________ do not make new materials a ...
Unit_Chemistry_1a_Oil
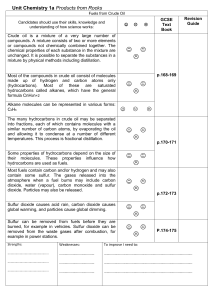
... The many hydrocarbons in crude oil may be separated into fractions, each of which contains molecules with a similar number of carbon atoms, by evaporating the oil and allowing it to condense at a number of different temperatures. This process is fractional distillation. Some properties of hydrocarbo ...
... The many hydrocarbons in crude oil may be separated into fractions, each of which contains molecules with a similar number of carbon atoms, by evaporating the oil and allowing it to condense at a number of different temperatures. This process is fractional distillation. Some properties of hydrocarbo ...
Acid rain
Acid rain is a rain or any other form of precipitation that is unusually acidic, meaning that it possesses elevated levels of hydrogen ions (low pH). It can have harmful effects on plants, aquatic animals and infrastructure. Acid rain is caused by emissions of sulfur dioxide and nitrogen oxide, which react with the water molecules in the atmosphere to produce acids. Governments have made efforts since the 1970s to reduce the release of sulfur dioxide into the atmosphere with positive results. Nitrogen oxides can also be produced naturally by lightning strikes and sulfur dioxide is produced by volcanic eruptions. The chemicals in acid rain can cause paint to peel, corrosion of steel structures such as bridges, and erosion of stone statues.