Precision, accuracy and significant figures
... remaining gases, when bubbled through a NaOH solution, increased its mass by 27.83 g. In a separate experiment, a 6.21 g sample of the compound was vaporised. The vapour occupied 2.17 L at 200°C and 1.25 × 105 Pa. Calculate the molecular formula of the compound. ...
... remaining gases, when bubbled through a NaOH solution, increased its mass by 27.83 g. In a separate experiment, a 6.21 g sample of the compound was vaporised. The vapour occupied 2.17 L at 200°C and 1.25 × 105 Pa. Calculate the molecular formula of the compound. ...
Stoichiometry of Ozonation of Environmentally
... Because of ozone’s high reactivity, it is difficult to attain its true solubility equilibrium (20). Under the steady flow conditions used in this work, Henry’s law is not expected to be obeyed because the decomposition of ozone in reaction 3 is significant on the time scales of headspace gas-exchan ...
... Because of ozone’s high reactivity, it is difficult to attain its true solubility equilibrium (20). Under the steady flow conditions used in this work, Henry’s law is not expected to be obeyed because the decomposition of ozone in reaction 3 is significant on the time scales of headspace gas-exchan ...
NO - Blue Devil Chem
... gas causing the column of carbon puff up, just like gases during cooking cause a cake to rise. The name carbohydrate derives from the formula of sugars such as sucrose, C12H22O11 (notice the 2:1 H:O ratio C12(H2O)11) in which the formula appears to be a hydrate of carbon. C12H22O11(s) → 11H2O(g) + 1 ...
... gas causing the column of carbon puff up, just like gases during cooking cause a cake to rise. The name carbohydrate derives from the formula of sugars such as sucrose, C12H22O11 (notice the 2:1 H:O ratio C12(H2O)11) in which the formula appears to be a hydrate of carbon. C12H22O11(s) → 11H2O(g) + 1 ...
LABORATORY MANUAL CHEMISTRY 121
... reaction can be followed visually because the products are red while the reactant is green. The purpose of this exercise is to determine the rate law for the hydrolysis of trans-[Co(en)2Cl2]+ and to determine the activation energy for the hydrolysis reaction by carrying out the reaction at several d ...
... reaction can be followed visually because the products are red while the reactant is green. The purpose of this exercise is to determine the rate law for the hydrolysis of trans-[Co(en)2Cl2]+ and to determine the activation energy for the hydrolysis reaction by carrying out the reaction at several d ...
OCR Gateway Science
... The equation for the complete combustion of methane is shown below. CH4 + 2O2 → CO2 + 2H2O When 80 g of methane is completely combusted, 220 g of carbon dioxide and 180 g of water are formed. (a) Why is the mass of carbon dioxide formed greater than the mass of methane burnt? (b) Calculate the mass ...
... The equation for the complete combustion of methane is shown below. CH4 + 2O2 → CO2 + 2H2O When 80 g of methane is completely combusted, 220 g of carbon dioxide and 180 g of water are formed. (a) Why is the mass of carbon dioxide formed greater than the mass of methane burnt? (b) Calculate the mass ...
Document
... macrocycles onto silica and have used these materials in the analysis of a wide variety of enantiomeric and diastereomeric guests. Recent work in our laboratory has shown that the intercalation of chiral cationic host molecules into R-zirconium phosphate, a lamellar cation exchanger, provides a usef ...
... macrocycles onto silica and have used these materials in the analysis of a wide variety of enantiomeric and diastereomeric guests. Recent work in our laboratory has shown that the intercalation of chiral cationic host molecules into R-zirconium phosphate, a lamellar cation exchanger, provides a usef ...
CHM203 - National Open University of Nigeria
... orderly and rigid fashion. When such as solid is heated, the thermal energy of the molecules increases. This finally leads to the disintegration of the crystal structure and at the melting point a disorderly and random arrangement of particles, characteristic of a liquid, is obtained. Since the elec ...
... orderly and rigid fashion. When such as solid is heated, the thermal energy of the molecules increases. This finally leads to the disintegration of the crystal structure and at the melting point a disorderly and random arrangement of particles, characteristic of a liquid, is obtained. Since the elec ...
Coordination and Chemistry of Stable Cu (II) Complexes in the Gas
... which is influenced by a number of common factors. First, complexes containing just one or two ligands are frequently either absent or have very low intensities because they are unstable with respect to charge transfer. The ionization energy of Cu+ is 20.29 eV, compared with that of most ligands at ...
... which is influenced by a number of common factors. First, complexes containing just one or two ligands are frequently either absent or have very low intensities because they are unstable with respect to charge transfer. The ionization energy of Cu+ is 20.29 eV, compared with that of most ligands at ...
organic problems - St. Olaf College
... A) carbon forms strong covalent bonds to itself, allowing chains and rings to be made. B) carbon expands its valence shell to accomodate more than eight electrons and thus forms double and triple bonds. C) carbon forms strong covalent bonds to elements like nitrogen and oxygen because it does not ha ...
... A) carbon forms strong covalent bonds to itself, allowing chains and rings to be made. B) carbon expands its valence shell to accomodate more than eight electrons and thus forms double and triple bonds. C) carbon forms strong covalent bonds to elements like nitrogen and oxygen because it does not ha ...
CHAPTER 15 ACIDS AND BASES
... At this point, we can make an assumption that x is very small compared to 0.60. Hence, 0.60 − x ≈ 0.60 Oftentimes, assumptions such as these are valid if K is very small. A very small value of K means that a very small amount of reactants go to products. Hence, x is small. If we did not make this as ...
... At this point, we can make an assumption that x is very small compared to 0.60. Hence, 0.60 − x ≈ 0.60 Oftentimes, assumptions such as these are valid if K is very small. A very small value of K means that a very small amount of reactants go to products. Hence, x is small. If we did not make this as ...
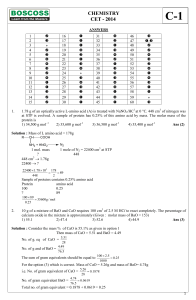
CHEMISTRY CET
... An incorrect statement with respect to SN1 and SN2 mechanisms for alkyl halide is (1) Competing reaction for an SN2 reaction is rearrangement. (2) A weak nucleophile and a protic solvent increases the rate or favours SN1 reaction. (3) A strong nucleophile in an aprotic solvent increases the rate or ...
... An incorrect statement with respect to SN1 and SN2 mechanisms for alkyl halide is (1) Competing reaction for an SN2 reaction is rearrangement. (2) A weak nucleophile and a protic solvent increases the rate or favours SN1 reaction. (3) A strong nucleophile in an aprotic solvent increases the rate or ...
Liquid–liquid extraction

Liquid–liquid extraction (LLE) consists in transferring one (or more) solute(s) contained in a feed solution to another immiscible liquid (solvent). The solvent that is enriched in solute(s) is called extract. The feed solution that is depleted in solute(s) is called raffinate.Liquid–liquid extraction also known as solvent extraction and partitioning, is a method to separate compounds based on their relative solubilities in two different immiscible liquids, usually water and an organic solvent. It is an extraction of a substance from one liquid into another liquid phase. Liquid–liquid extraction is a basic technique in chemical laboratories, where it is performed using a variety of apparatus, from separatory funnels to countercurrent distribution equipment. This type of process is commonly performed after a chemical reaction as part of the work-up.The term partitioning is commonly used to refer to the underlying chemical and physical processes involved in liquid–liquid extraction, but on another reading may be fully synonymous with it. The term solvent extraction can also refer to the separation of a substance from a mixture by preferentially dissolving that substance in a suitable solvent. In that case, a soluble compound is separated from an insoluble compound or a complex matrix.Solvent extraction is used in nuclear reprocessing, ore processing, the production of fine organic compounds, the processing of perfumes, the production of vegetable oils and biodiesel, and other industries.Liquid–liquid extraction is possible in non-aqueous systems: In a system consisting of a molten metal in contact with molten salts, metals can be extracted from one phase to the other. This is related to a mercury electrode where a metal can be reduced, the metal will often then dissolve in the mercury to form an amalgam that modifies its electrochemistry greatly. For example, it is possible for sodium cations to be reduced at a mercury cathode to form sodium amalgam, while at an inert electrode (such as platinum) the sodium cations are not reduced. Instead, water is reduced to hydrogen. A detergent or fine solid can be used to stabilize an emulsion, or third phase.