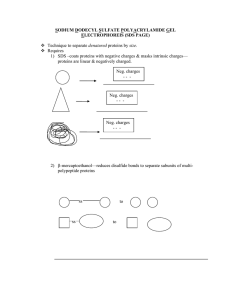
SDS Electrophoresis
... But then separated subunits/polypeptides will be linear & negative due to SDS treatment 3) Heat—to further denature proteins 4) polyacrylamide—gel matrix that acts as size sorter 5) electrophoresis, using electric field with positive anode and negative cathode, all proteins are attracted to bottom ...
... But then separated subunits/polypeptides will be linear & negative due to SDS treatment 3) Heat—to further denature proteins 4) polyacrylamide—gel matrix that acts as size sorter 5) electrophoresis, using electric field with positive anode and negative cathode, all proteins are attracted to bottom ...
Protein Origami
... makes fireflies glow, and a lot more. Proteins also aid viruses in invading cells. AIDS can be tied to proteins that break through cellular defenses and replicate the HIV virus. Cancer is linked to damage in proteins that inhibit uncontrolled cell growth. Alzheimer’s disease is believed to be relate ...
... makes fireflies glow, and a lot more. Proteins also aid viruses in invading cells. AIDS can be tied to proteins that break through cellular defenses and replicate the HIV virus. Cancer is linked to damage in proteins that inhibit uncontrolled cell growth. Alzheimer’s disease is believed to be relate ...
投影片 1
... proteasomal degradation, most proteins are covalently modified with ubiquitin (Ub). Typically, three enzyme types are involved in this process — ubiquitin-activating (E1), ubiquitin-conjugating (E2) and ubiquitin ligase (E3) enzymes. Proteins tagged with chains of four or more ubiquitins are shuttle ...
... proteasomal degradation, most proteins are covalently modified with ubiquitin (Ub). Typically, three enzyme types are involved in this process — ubiquitin-activating (E1), ubiquitin-conjugating (E2) and ubiquitin ligase (E3) enzymes. Proteins tagged with chains of four or more ubiquitins are shuttle ...
protein - CSU, Chico
... The more restrictive the diet, the more potential for nutritional inadequacy Special attention to combining proteins: ...
... The more restrictive the diet, the more potential for nutritional inadequacy Special attention to combining proteins: ...
Protein Structures
... groups in amino acids are either hydrophobic or hydrophilic and will seek aquatic or non-aquatic environments accordingly, which determines their location within the protein. Hydrogen bonds facilitate stabilization within the proteins based on the shape established by the hydrophobic interactions. I ...
... groups in amino acids are either hydrophobic or hydrophilic and will seek aquatic or non-aquatic environments accordingly, which determines their location within the protein. Hydrogen bonds facilitate stabilization within the proteins based on the shape established by the hydrophobic interactions. I ...
Protein 101 A3 poster.indd
... Protein is an essential part of a balanced diet. Protein molecules consist of chains of amino acids. Different proteins are made up of different amino acids, nine of which are essential for humans, as they cannot be synthesized by the body. The nine essential amino acids are: lysine, histidine, isol ...
... Protein is an essential part of a balanced diet. Protein molecules consist of chains of amino acids. Different proteins are made up of different amino acids, nine of which are essential for humans, as they cannot be synthesized by the body. The nine essential amino acids are: lysine, histidine, isol ...
Protein PPT Editted
... Maintains acid-base balance in the blood Carries vital substances (combines with fat to form lipoproteins, transport of iron and other nutrients and oxygen in the blood Provides energy as a last resort if the body can’t get energy from carbs and fat or if there is too much protein in the diet Protei ...
... Maintains acid-base balance in the blood Carries vital substances (combines with fat to form lipoproteins, transport of iron and other nutrients and oxygen in the blood Provides energy as a last resort if the body can’t get energy from carbs and fat or if there is too much protein in the diet Protei ...
T-cell regulator RNF125/TRAC-1 belongs to a novel family of
... or E1, binds ubiquitin and transfers it to the active site of an ubiquitin-conjugating enzyme, or E2. E2 proteins interact with the ubiquitin ligases, or E3s, and promote the attachment of ubiquitin to substrates of these proteins. Ubiquitin is attached primarily to lysine residues on target protein ...
... or E1, binds ubiquitin and transfers it to the active site of an ubiquitin-conjugating enzyme, or E2. E2 proteins interact with the ubiquitin ligases, or E3s, and promote the attachment of ubiquitin to substrates of these proteins. Ubiquitin is attached primarily to lysine residues on target protein ...
L2 - Proteins
... 2. Salt bridges – ionic bonds between acidic and basic residues. 3. Hydrogen bonds – between polar residues 4. Hydrophobic interactions – between nonpolar residues. ...
... 2. Salt bridges – ionic bonds between acidic and basic residues. 3. Hydrogen bonds – between polar residues 4. Hydrophobic interactions – between nonpolar residues. ...
Fibrous proteins
... because free iron is toxic and oxidize cells (form reactive oxygen species). Ferritin is present in liver, spleen and bone marrow. The amount of ferritin stored reflects the amount of iron stored. Ferritin releases iron to areas where it is required. ...
... because free iron is toxic and oxidize cells (form reactive oxygen species). Ferritin is present in liver, spleen and bone marrow. The amount of ferritin stored reflects the amount of iron stored. Ferritin releases iron to areas where it is required. ...
SIP - Proteins from oil seedsremarks - 20150317
... contain valuable compounds like proteins. The quality of the proteins and lipids in these co-products is largely determined by the processing conditions. An important step in the processing of rapeseed is the inactivation of enzymes like myrosinase and lipoxygenase. Enzyme activity of both enzymes l ...
... contain valuable compounds like proteins. The quality of the proteins and lipids in these co-products is largely determined by the processing conditions. An important step in the processing of rapeseed is the inactivation of enzymes like myrosinase and lipoxygenase. Enzyme activity of both enzymes l ...
Proteomics – 2D gels - Department of Chemistry and Biochemistry
... Scanners and computer programs are used to read the gels. They are only as good as the scanners acuity and the programs ability to discern spots on the scan. ...
... Scanners and computer programs are used to read the gels. They are only as good as the scanners acuity and the programs ability to discern spots on the scan. ...
Chapter 5 Separations: I) Based on Charge or pI A) Electrophoresis
... 1→Must denature the protein first. 2→Then break the polypeptide chain into smaller fragments with: proteolytic enzymes (endopeptidases) Trypsin cleaves protein after R and K residues Chymotrypsin cleaves after aromatics F, Y, W Elastase cleaves after small hydrophobic residues Chemical cyanogens bro ...
... 1→Must denature the protein first. 2→Then break the polypeptide chain into smaller fragments with: proteolytic enzymes (endopeptidases) Trypsin cleaves protein after R and K residues Chymotrypsin cleaves after aromatics F, Y, W Elastase cleaves after small hydrophobic residues Chemical cyanogens bro ...
Fibrous and globular proteins Structure
... Function of cross-linking These cross-links stabilize the side-by-side packing of collagen molecules and generate a strong fibril If cross-linking is inhibited, the tensile strength of the fibrils is drastically reduced; collagenous tissues become fragile, and structures such as skin, tendons, and ...
... Function of cross-linking These cross-links stabilize the side-by-side packing of collagen molecules and generate a strong fibril If cross-linking is inhibited, the tensile strength of the fibrils is drastically reduced; collagenous tissues become fragile, and structures such as skin, tendons, and ...
Fibrous and globular proteins Structure
... Function of cross-linking These cross-links stabilize the side-by-side packing of collagen molecules and generate a strong fibril If cross-linking is inhibited, the tensile strength of the fibrils is drastically reduced; collagenous tissues become fragile, and structures such as skin, tendons, and ...
... Function of cross-linking These cross-links stabilize the side-by-side packing of collagen molecules and generate a strong fibril If cross-linking is inhibited, the tensile strength of the fibrils is drastically reduced; collagenous tissues become fragile, and structures such as skin, tendons, and ...
Group : Nanochemical Biology Project : Tyrosine cross
... Horseradish peroxidase (HRP) is used to activate tyrosine residues in proteins, converting them into tyrosine radicals, which then cross-react with other amino acid residues (mostly tyrosine). A major drawback of the HRP is its high reactivity, leading to dirty products that are very difficult to pu ...
... Horseradish peroxidase (HRP) is used to activate tyrosine residues in proteins, converting them into tyrosine radicals, which then cross-react with other amino acid residues (mostly tyrosine). A major drawback of the HRP is its high reactivity, leading to dirty products that are very difficult to pu ...
Document
... 1) Forms protein’s initial S-S bonds in similar way (protein –SH attacks PDI S-S bond to give mixed disulfide) 2) Protein SH attacks protein-PDI mixed S-S bond to give protein S-S bond 3) Continues until protein in native S-S configuration and PDI cannot bind to exposed hydrophobic patches on the pr ...
... 1) Forms protein’s initial S-S bonds in similar way (protein –SH attacks PDI S-S bond to give mixed disulfide) 2) Protein SH attacks protein-PDI mixed S-S bond to give protein S-S bond 3) Continues until protein in native S-S configuration and PDI cannot bind to exposed hydrophobic patches on the pr ...
In Silico Prediction of Peroxisomal Proteins in Mouse
... caused either by impairment of peroxisome biogenesis or by defects of individual peroxisomal enzymes. Despite the fact that peroxisomes were first discovered in the 1960s, the functions performed by these organelles are still not fully understood. Identification of all peroxisome components will aid ...
... caused either by impairment of peroxisome biogenesis or by defects of individual peroxisomal enzymes. Despite the fact that peroxisomes were first discovered in the 1960s, the functions performed by these organelles are still not fully understood. Identification of all peroxisome components will aid ...
The Scientist : Lab Tools: Close Encounters
... The problem: Y2H measures only binary interactions, not multiprotein complexes. But coIP /MS requires high-quality, specific antibodies, few of which are available. Superti-Furga needed a generic way to isolate intact complexes from cells. The Solution: Superti-Furga and his team adapted a process c ...
... The problem: Y2H measures only binary interactions, not multiprotein complexes. But coIP /MS requires high-quality, specific antibodies, few of which are available. Superti-Furga needed a generic way to isolate intact complexes from cells. The Solution: Superti-Furga and his team adapted a process c ...
Introduction to Proteins: Biotech 2
... Primary (the AA sequence of its polypeptide chain) Secondary (H bonding between peptide bonds) Tertiary (covalent, ionic, H bonding, hydrophobic) Quaternary (involves more than one subunit) ...
... Primary (the AA sequence of its polypeptide chain) Secondary (H bonding between peptide bonds) Tertiary (covalent, ionic, H bonding, hydrophobic) Quaternary (involves more than one subunit) ...
Proteins - Madison Public Schools
... groups, disulfide bridges between R groups containing sulfur, and weak forces of attraction called van der Waals forces all contribute to the folding of proteins. ...
... groups, disulfide bridges between R groups containing sulfur, and weak forces of attraction called van der Waals forces all contribute to the folding of proteins. ...
Enzymes and proteins - Hochschule Biberach
... of the side chain, acid-base behaviour of the side chain, polarity of the side chain, chemical differentiation • Structure systems in proteins (helix, folding pattern, reverse turn, domains), X-ray diffraction analysis, interaction between protein side chains • Non-protein structure components (glyc ...
... of the side chain, acid-base behaviour of the side chain, polarity of the side chain, chemical differentiation • Structure systems in proteins (helix, folding pattern, reverse turn, domains), X-ray diffraction analysis, interaction between protein side chains • Non-protein structure components (glyc ...
Nutritional Requirements of Non
... Other plant and animal proteins are often used in combination with SBM. • Fishmeal (60-70% CP) (Crude Protein) • Milk products, such as dried skimmed milk (33% CP) and dried whey (13.3% CP) • Meat and bone meal (50% CP) • Spray dried plasma protein and bloodmeal (86% CP) ...
... Other plant and animal proteins are often used in combination with SBM. • Fishmeal (60-70% CP) (Crude Protein) • Milk products, such as dried skimmed milk (33% CP) and dried whey (13.3% CP) • Meat and bone meal (50% CP) • Spray dried plasma protein and bloodmeal (86% CP) ...
Ubiquitin

Ubiquitin is a small (8.5 kDa) regulatory protein that has been found in almost all tissues (ubiquitously) of eukaryotic organisms. It was discovered in 1975 by Goldstein and further characterized throughout the 1970s and 1980s. There are four genes in the human genome that produce ubiquitin: UBB, UBC, UBA52 and RPS27A.The addition of ubiquitin to a substrate protein is called ubiquitination or ubiquitylation. Ubiquitination can affect proteins in many ways: It can signal for their degradation via the proteasome, alter their cellular location, affect their activity, and promote or prevent protein interactions. Ubiquitination is carried out in three main steps: activation, conjugation, and ligation, performed by ubiquitin-activating enzymes (E1s), ubiquitin-conjugating enzymes (E2s), and ubiquitin ligases (E3s), respectively. The result of this sequential cascade binds ubiquitin to lysine residues on the protein substrate via an isopeptide bond or to the amino group of the protein's N-terminus via a peptide bond.The protein modifications can be either a single ubiquitin protein (monoubiquitination) or a chain of ubiquitin (polyubiquitination). The ubiquitination bonds are always formed with one of the seven lysine residues from the ubiquitin molecule. These 'linking' lysines are represented by a ""K"" (which is the one-letter amino acid notation of lysine) and a number, referring to its position in the ubiquitin molecule. First, a ubiquitin molecule is bonded by its C-terminus to a specific lysine residue (e.g. K48, K29, K63,...) on the target protein. Poly-ubiquitination occurs when the C-terminus of another ubiquitin, will be linked again to a lysine residue (for example again K48 or K29) on the previously added ubiquitin molecule, forming a chain. This process repeats several times, leading to the addition of several ubiquitins. Only poly-ubiquitination on defined lysines, mostly on K48 and K29, is related to degradation with the proteasome (referred to as the ""molecular kiss of death""), while other polyubiquitinations (e.g. on K63, K11, K6) and monoubiquitinations may regulate processes such as endocytic trafficking, inflammation, translation and DNA repair.Lysine 48-linked chains have been much-studied. They are the forms of chains that signal proteins to the proteasome, which destroys and recycles proteins. This discovery won the Nobel Prize for chemistry in 2004.