Measuring Temperature
... When we use the earth's energy resources, we do not decrease the earth's total energy. This is just another way of stating the Law of Conservation of Energy. Instead, we are changing highly useful forms of energy into less useful forms. And this restates the 2nd Law of Thermodynamics. ...
... When we use the earth's energy resources, we do not decrease the earth's total energy. This is just another way of stating the Law of Conservation of Energy. Instead, we are changing highly useful forms of energy into less useful forms. And this restates the 2nd Law of Thermodynamics. ...
Lecture #3 09/02/04
... •Recall: At nonzero finite temperature, thermal energy can result in the population of multiple states inside an ensemble •What does this mean? •We have to consider the statistical weight of each possible orientation ...
... •Recall: At nonzero finite temperature, thermal energy can result in the population of multiple states inside an ensemble •What does this mean? •We have to consider the statistical weight of each possible orientation ...
Modeling of combined thermal and mechanical action in roller
... is carried out using a three-dimensional finite element method. In this work a numerical procedure for the simulation of construction process and service life of RCC dams is presented. It takes into account the more relevant features of the behavior of concrete such as hydration, ageing and creep. A ...
... is carried out using a three-dimensional finite element method. In this work a numerical procedure for the simulation of construction process and service life of RCC dams is presented. It takes into account the more relevant features of the behavior of concrete such as hydration, ageing and creep. A ...
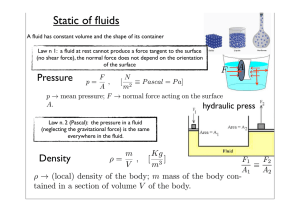
Static of fluids
... Equipartition of energy In the motion of molecules in a gas at a given temperature, to every independent component of the motion (degree of freedom) corresponds the same kinetic energy ...
... Equipartition of energy In the motion of molecules in a gas at a given temperature, to every independent component of the motion (degree of freedom) corresponds the same kinetic energy ...
Pyroelectric-Based Solar and Wind Energy Harvesting System
... involving the Olsen cycle [4], which is based on the thermodynamic Ericsson cycle, with two isothermal and two isoelectric field processes. This cycle has been realized for different materials like polymers, single crystals, and relaxor ferroelectrics and for different electrical fields and temperat ...
... involving the Olsen cycle [4], which is based on the thermodynamic Ericsson cycle, with two isothermal and two isoelectric field processes. This cycle has been realized for different materials like polymers, single crystals, and relaxor ferroelectrics and for different electrical fields and temperat ...
The Electronic Structures of Atoms Electromagnetic Radiation The
... In 1913 Neils Bohr incorporated Planck’s quantum theory into the hydrogen spectrum explanation. Here are the postulates of Bohr’s theory. Atom has a number of definite and discrete energy levels (orbits) in which an electron may exist without emitting or absorbing electromagnetic radiation. As the o ...
... In 1913 Neils Bohr incorporated Planck’s quantum theory into the hydrogen spectrum explanation. Here are the postulates of Bohr’s theory. Atom has a number of definite and discrete energy levels (orbits) in which an electron may exist without emitting or absorbing electromagnetic radiation. As the o ...
electrons - TAMU Chemistry
... Light of a characteristic wavelength (and frequency) is absorbed when electron jumps from lower E (orbit, n = 2) to higher E (orbit, n= 4) – This is the origin of absorption spectra. ...
... Light of a characteristic wavelength (and frequency) is absorbed when electron jumps from lower E (orbit, n = 2) to higher E (orbit, n= 4) – This is the origin of absorption spectra. ...
241 Lecture 11
... Zeroth Law of Thermodynamics • Remember with Thermal equilibrium • Two systems are said to be in thermal equilibrium if there is no net flow of heat between them when they are brought into thermal contact. • Temperature is the indicator of thermal equilibrium • Two systems individually in thermal e ...
... Zeroth Law of Thermodynamics • Remember with Thermal equilibrium • Two systems are said to be in thermal equilibrium if there is no net flow of heat between them when they are brought into thermal contact. • Temperature is the indicator of thermal equilibrium • Two systems individually in thermal e ...
2 - Mineola ISD
... and liquids. Heat convection does not occur in solids because solids are unable to — ¢A absorb heat by vibrating ¢B transfer heat by fluid motion ¢C emit radiation by reflecting light ¢D exchange heat by direct contact Solids do radiate heat to their surroundi ngs ...
... and liquids. Heat convection does not occur in solids because solids are unable to — ¢A absorb heat by vibrating ¢B transfer heat by fluid motion ¢C emit radiation by reflecting light ¢D exchange heat by direct contact Solids do radiate heat to their surroundi ngs ...
Introduction to Spectroscopic Methods ver.2
... interaction of various types of radiation with matter. Historically, the interactions of interest between electromagnetic radiation and matter, but now spectroscopy has been broadened to include interactions between matter and other forms of energy. Examples include acoustic waves and beams of parti ...
... interaction of various types of radiation with matter. Historically, the interactions of interest between electromagnetic radiation and matter, but now spectroscopy has been broadened to include interactions between matter and other forms of energy. Examples include acoustic waves and beams of parti ...
Thermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of charged particles in matter. An object with a temperature greater than absolute zero emits thermal radiation. When the temperature of the body is greater than absolute zero, interatomic collisions cause the kinetic energy of the atoms or molecules to change. This results in charge-acceleration and/or dipole oscillation which produces electromagnetic radiation, and the wide spectrum of radiation reflects the wide spectrum of energies and accelerations that occur even at a single temperature.Examples of thermal radiation include the visible light and infrared light emitted by an incandescent light bulb, the infrared radiation emitted by animals and detectable with an infrared camera, and the cosmic microwave background radiation. Thermal radiation is different from thermal convection and thermal conduction—a person near a raging bonfire feels radiant heating from the fire, even if the surrounding air is very cold.Sunlight is part of thermal radiation generated by the hot plasma of the Sun. The Earth also emits thermal radiation, but at a much lower intensity and different spectral distribution (infrared rather than visible) because it is cooler. The Earth's absorption of solar radiation, followed by its outgoing thermal radiation are the two most important processes that determine the temperature and climate of the Earth.If a radiation-emitting object meets the physical characteristics of a black body in thermodynamic equilibrium, the radiation is called blackbody radiation. Planck's law describes the spectrum of blackbody radiation, which depends only on the object's temperature. Wien's displacement law determines the most likely frequency of the emitted radiation, and the Stefan–Boltzmann law gives the radiant intensity.Thermal radiation is one of the fundamental mechanisms of heat transfer.