Chapter 2 What Is Matter
... in a liquid. Since there are three common phases of matter, there are nine possible types of solutions. Such as: Air is a gas dissolved in a gas. Soft drinks are a gas dissolved in a liquid. Humid air is a liquid dissolved in a gas. Antifreeze is a liquid dissolved in a liquid. Saltwater is a solid ...
... in a liquid. Since there are three common phases of matter, there are nine possible types of solutions. Such as: Air is a gas dissolved in a gas. Soft drinks are a gas dissolved in a liquid. Humid air is a liquid dissolved in a gas. Antifreeze is a liquid dissolved in a liquid. Saltwater is a solid ...
Chemistry- The Gas Phase
... is to react a strong acid with a metal: 2HCl(aq) + Zn(s) → H2(g) + ZnCl2(aq) If 512 g of HCl are reacted with excess Zn at T=24 oC and P=0.95 atm, what volume of H2 gas will be produced? ...
... is to react a strong acid with a metal: 2HCl(aq) + Zn(s) → H2(g) + ZnCl2(aq) If 512 g of HCl are reacted with excess Zn at T=24 oC and P=0.95 atm, what volume of H2 gas will be produced? ...
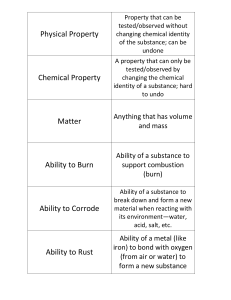
Physical Property
... A property that can only be tested/observed by changing the chemical identity of a substance; hard to undo ...
... A property that can only be tested/observed by changing the chemical identity of a substance; hard to undo ...
42.89 KB
... 18. At very high pressures (~ 1000 atm), the measured pressure exerted by real gases is greater than that predicted by the ideal gas equation. This is mainly because A) the volume occupied by the gas molecules themselves becomes significant. B) real gases will condense to form solids at 1000 atm pre ...
... 18. At very high pressures (~ 1000 atm), the measured pressure exerted by real gases is greater than that predicted by the ideal gas equation. This is mainly because A) the volume occupied by the gas molecules themselves becomes significant. B) real gases will condense to form solids at 1000 atm pre ...
Separation and Purification Methods
... The other methods fall under the category of LC (liquid chromatography), where the mobile phase is a solvent system, which can be used instead of temperature to leverage retention. Occasionally, this is a single solvent, but more often it is a binary mixture of solvents with different polarities. Th ...
... The other methods fall under the category of LC (liquid chromatography), where the mobile phase is a solvent system, which can be used instead of temperature to leverage retention. Occasionally, this is a single solvent, but more often it is a binary mixture of solvents with different polarities. Th ...
clear-traction-msds - Pioneer Research Corporation
... Eyes: Flush immediately with large volumes of water for at least 15 minutes. Take to physician for medical treatment Ingestion: Consult physician or Poison Control Center immediately Skin Contact: Wash well with soap and water ...
... Eyes: Flush immediately with large volumes of water for at least 15 minutes. Take to physician for medical treatment Ingestion: Consult physician or Poison Control Center immediately Skin Contact: Wash well with soap and water ...
Chem 480A
... you track the units carefully at the end of this page you will see that it is necessary to keep the 1 mol out of the logarithm for the units to make sense.) ...
... you track the units carefully at the end of this page you will see that it is necessary to keep the 1 mol out of the logarithm for the units to make sense.) ...
Scientific Notation, Measurements, and
... Temperature is a measurement of how hot or cold something is. It is a calculation of the average kinetic energy of all the particles in the substance. Scientists theorize that at absolute zero, or –273.15oC, particle motion stops and that no more energy can be removed from the object. So this is the ...
... Temperature is a measurement of how hot or cold something is. It is a calculation of the average kinetic energy of all the particles in the substance. Scientists theorize that at absolute zero, or –273.15oC, particle motion stops and that no more energy can be removed from the object. So this is the ...
Dielectric properties of critical conducting mixtures
... mixtures. In this lecture the dielectric properties - dielectric permittivity, conductivity and non-linear dielectric effect will be presented in pure binary mixtures and in mixtures doped by small amounts of ionic ingredients. Interesting properties related to the existence of ions capable to migra ...
... mixtures. In this lecture the dielectric properties - dielectric permittivity, conductivity and non-linear dielectric effect will be presented in pure binary mixtures and in mixtures doped by small amounts of ionic ingredients. Interesting properties related to the existence of ions capable to migra ...
AP Chemistry Chapter 11 Notes - Properties of Solutions In a , or
... Sample Exercise 11.8 on page 528: Calculate the Molar mass by Boiling Point Elevation: A solution was prepared by dissolving 18.00 g glucose in 150.0 g water. The resulting solution was found to have a boiling point of 100.34 ºC. Calculate the molar mass of glucose. Glucose is a molecular solid that ...
... Sample Exercise 11.8 on page 528: Calculate the Molar mass by Boiling Point Elevation: A solution was prepared by dissolving 18.00 g glucose in 150.0 g water. The resulting solution was found to have a boiling point of 100.34 ºC. Calculate the molar mass of glucose. Glucose is a molecular solid that ...
High pressure differential scanning calorimetry of the hydrothermal
... below the liquid 1-gas critical endpoint R.7 The schematic T–x projections of the two phase diagrams of interest (type 1d, 2d0 ) are shown in Fig. 1. As in naturally occurring salt solutions typically more than one salt is dissolved, higher order mixtures are also of interest. In the phase diagrams ...
... below the liquid 1-gas critical endpoint R.7 The schematic T–x projections of the two phase diagrams of interest (type 1d, 2d0 ) are shown in Fig. 1. As in naturally occurring salt solutions typically more than one salt is dissolved, higher order mixtures are also of interest. In the phase diagrams ...
Regents Chemistry
... o use solubility curves to predict how much water is required to dissolve a given amount of solute at a given temp or how much solute will dissolve in a given amount of water o be able to predict the amount of solute that will crystallize (precipitate) from solution when it is chilled o use Table G ...
... o use solubility curves to predict how much water is required to dissolve a given amount of solute at a given temp or how much solute will dissolve in a given amount of water o be able to predict the amount of solute that will crystallize (precipitate) from solution when it is chilled o use Table G ...
NAME: AP CHEMISTRY CHAPTER 8, #5 (Questions 1
... A solution is made by dissolving a nonvolatile solute in pure solvent. Compared to the pure solvent, the solution A. has a lower than normal boiling point B. has a higher vapor pressure C. has the same vapor pressure D. has a lower than normal freezing point E. is more nearly ideal ...
... A solution is made by dissolving a nonvolatile solute in pure solvent. Compared to the pure solvent, the solution A. has a lower than normal boiling point B. has a higher vapor pressure C. has the same vapor pressure D. has a lower than normal freezing point E. is more nearly ideal ...
Mole assignment
... represent the number of atoms or molecules. For instance, in about 18.02 g of water, there is one mole of water molecules (i.e. 6.02 x 1023). This is a very large number. Since atoms and molecules are very small in size, we must work with many of them for every day applications. To better understand ...
... represent the number of atoms or molecules. For instance, in about 18.02 g of water, there is one mole of water molecules (i.e. 6.02 x 1023). This is a very large number. Since atoms and molecules are very small in size, we must work with many of them for every day applications. To better understand ...
(p. 522)
... 3. Disulfide bridges. These form between the side chains of two cysteine residues, and may hold distant parts of the chain close together. 4. Salt links. These form between oppositely-charged side chains, the charges being due to the COO¯ and -NH3+ groups. 5. Dispersion forces. Nonpolar side chains ...
... 3. Disulfide bridges. These form between the side chains of two cysteine residues, and may hold distant parts of the chain close together. 4. Salt links. These form between oppositely-charged side chains, the charges being due to the COO¯ and -NH3+ groups. 5. Dispersion forces. Nonpolar side chains ...