L1 – CHEMISTRY FINAL REVIEW
... water); only substance in which the solid state is less dense than the liquid state; unusually high BP when compared to other molecular compounds with similar molecular weight. 34. How is a saturation point an equilibrium point? the rate at which the solute is dissolving is equal to the rate at whic ...
... water); only substance in which the solid state is less dense than the liquid state; unusually high BP when compared to other molecular compounds with similar molecular weight. 34. How is a saturation point an equilibrium point? the rate at which the solute is dissolving is equal to the rate at whic ...
Chemistry 12 Keq WORKSHEET #1
... 4. The equilibrium constant for the formation of ammonia by the reaction N2 (g) + 3 H2 (g) <===> 2 NH3 (g) is 2.0 at a certain temperature. If the equilibrium concentration of N2 in a mixture is 0.50 M and H2 is 2.0 M, determine the concentration of ammonia. 5. At 2000oK, a mixture of H2, S2, and H2 ...
... 4. The equilibrium constant for the formation of ammonia by the reaction N2 (g) + 3 H2 (g) <===> 2 NH3 (g) is 2.0 at a certain temperature. If the equilibrium concentration of N2 in a mixture is 0.50 M and H2 is 2.0 M, determine the concentration of ammonia. 5. At 2000oK, a mixture of H2, S2, and H2 ...
Chapter 13
... the reciprocal of that for the reaction written in reverse. When the equation for a reaction is multiplied by n, EEnew = (EEoriginal)n The units for K depend on the reaction being considered. K values are customarily written without units. ...
... the reciprocal of that for the reaction written in reverse. When the equation for a reaction is multiplied by n, EEnew = (EEoriginal)n The units for K depend on the reaction being considered. K values are customarily written without units. ...
Pervaporation overview
... 1. Feed concentration: Refers to the concentration of the preferentially permeating (usually minor) solution component, being depleted in the process. There are two aspects to be considered:the activity of the target component in the feed and the solubility of the target component in the membrane. • ...
... 1. Feed concentration: Refers to the concentration of the preferentially permeating (usually minor) solution component, being depleted in the process. There are two aspects to be considered:the activity of the target component in the feed and the solubility of the target component in the membrane. • ...
Chemistry 520 - Problem Set 2
... you will perform for this problem are the same as those needed to analyze one of the experiments in physical chemistry lab] (a) A student in a physical chemistry laboratory performed such an experiment. In the rst step, she burned a 0.825 g sample of benzoic acid (C6 H5COOH) for which the internal ...
... you will perform for this problem are the same as those needed to analyze one of the experiments in physical chemistry lab] (a) A student in a physical chemistry laboratory performed such an experiment. In the rst step, she burned a 0.825 g sample of benzoic acid (C6 H5COOH) for which the internal ...
Chapter 3: Mass Relationships in Chemical Reactions – Khan
... A The mole is a unit of measure that measures quantity. It is the same concept as a dozen. You can have a dozen doughnuts, oranges, golf balls, etc. and you always have a quantity of 12. The same goes for a mole, we can have a mole of doughnuts, oranges or golf balls and would always have 6.02 X 102 ...
... A The mole is a unit of measure that measures quantity. It is the same concept as a dozen. You can have a dozen doughnuts, oranges, golf balls, etc. and you always have a quantity of 12. The same goes for a mole, we can have a mole of doughnuts, oranges or golf balls and would always have 6.02 X 102 ...
Properties of Gases
... This law states that the volume (V) of a given mass of gas is directly proportional to its temperature (T) in Kelvin scale, at a constant Pressure (P). (As the Temperature increases Volume of the gas increases at constant P) V = (constant) x T ...
... This law states that the volume (V) of a given mass of gas is directly proportional to its temperature (T) in Kelvin scale, at a constant Pressure (P). (As the Temperature increases Volume of the gas increases at constant P) V = (constant) x T ...
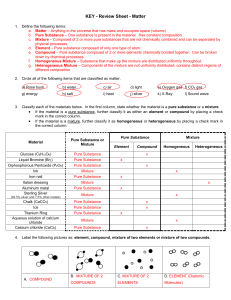
Chapter 2 Matter and Change
... Mixtures are a physical blend of at least two substances; have variable composition. They can be either: 1) Heterogeneous – the mixture is not uniform in composition • Chocolate chip cookie, gravel, soil. 2) Homogeneous - same composition throughout; called “solutions” • Kool-aid, air, salt water ...
... Mixtures are a physical blend of at least two substances; have variable composition. They can be either: 1) Heterogeneous – the mixture is not uniform in composition • Chocolate chip cookie, gravel, soil. 2) Homogeneous - same composition throughout; called “solutions” • Kool-aid, air, salt water ...
Solute
... PA A P A • PA = vapor pressure of sol’n • PA = vapor pressure of pure solvent • A = the mole fraction of solvent. ...
... PA A P A • PA = vapor pressure of sol’n • PA = vapor pressure of pure solvent • A = the mole fraction of solvent. ...
Honors Chemistry Final Essay Questions 2007
... 6. For the following reactions, write a balanced equation, net ionic equation (if one occurs), label the states of the reactants and products on both the balanced equation and the net ionic equation, and list the spectator ions and driving force for each of the reactions. a. silver nitrate solution ...
... 6. For the following reactions, write a balanced equation, net ionic equation (if one occurs), label the states of the reactants and products on both the balanced equation and the net ionic equation, and list the spectator ions and driving force for each of the reactions. a. silver nitrate solution ...
CHEM 101 Final (Term 141)
... substance B are lower than those for substance A. B) The pressure at the triple point, normal boiling and normal melting point for substance A are lower than those of substance B. C) The pressure at the triple point for substance A is higher than that of substance B, but the normal boiling and norma ...
... substance B are lower than those for substance A. B) The pressure at the triple point, normal boiling and normal melting point for substance A are lower than those of substance B. C) The pressure at the triple point for substance A is higher than that of substance B, but the normal boiling and norma ...
rocks and minerals quiz
... (6) From the Heat Capacities data table: H2O(liquid) = 1.000 cal/deg/gm (7) 100 g Water (1.000 cal/deg) ------------------- = 100 cal/deg 1g (8) 20oC (100 calories) ----------------- = 2000 calories 1oC (9) Total calories: 1940 calories + 7980 calories + 2000 calories (10) Total calories = 10,120 ca ...
... (6) From the Heat Capacities data table: H2O(liquid) = 1.000 cal/deg/gm (7) 100 g Water (1.000 cal/deg) ------------------- = 100 cal/deg 1g (8) 20oC (100 calories) ----------------- = 2000 calories 1oC (9) Total calories: 1940 calories + 7980 calories + 2000 calories (10) Total calories = 10,120 ca ...
Chapter one
... measurement of the amount of a solution of known concentration that is required to react completely with a measured amount of a solution of unknown concentration. ...
... measurement of the amount of a solution of known concentration that is required to react completely with a measured amount of a solution of unknown concentration. ...
rocks and minerals quiz
... (6) From the Heat Capacities data table: H2O(liquid) = 1.000 cal/deg/gm (7) 100 g Water (1.000 cal/deg) ------------------- = 100 cal/deg 1g (8) 20oC (100 calories) ----------------- = 2000 calories 1oC (9) Total calories: 1940 calories + 7980 calories + 2000 calories (10) Total calories = 11,920 ca ...
... (6) From the Heat Capacities data table: H2O(liquid) = 1.000 cal/deg/gm (7) 100 g Water (1.000 cal/deg) ------------------- = 100 cal/deg 1g (8) 20oC (100 calories) ----------------- = 2000 calories 1oC (9) Total calories: 1940 calories + 7980 calories + 2000 calories (10) Total calories = 11,920 ca ...
The Noble-Abel Stiffened-Gas equation of state
... energy and the heat bond of the corresponding phase. Parameters γ, P∞ , q and b are constant coefficients characteristic of the thermodynamic properties of the fluid. Among them the coefficient b represents the covolume of the fluid. This equation of state is a combination of the ‘Stiffened Gas’ EOS ...
... energy and the heat bond of the corresponding phase. Parameters γ, P∞ , q and b are constant coefficients characteristic of the thermodynamic properties of the fluid. Among them the coefficient b represents the covolume of the fluid. This equation of state is a combination of the ‘Stiffened Gas’ EOS ...
Name - cloudfront.net
... A 0.12 M solution of an acid that ionizes only slightly in solution would be termed ____. In the reaction: CO32- + H2O ↔ HCO31- + OH1-, the carbonate ion (CO32-) is acting as a(n) ____. What is transferred between a conjugate acid-base pair? What are the acids in the following: CN1- + H2O ↔ HCN + OH ...
... A 0.12 M solution of an acid that ionizes only slightly in solution would be termed ____. In the reaction: CO32- + H2O ↔ HCO31- + OH1-, the carbonate ion (CO32-) is acting as a(n) ____. What is transferred between a conjugate acid-base pair? What are the acids in the following: CN1- + H2O ↔ HCN + OH ...
Slide 1
... As the oxygen atoms are shared between adjacent tetrahedra, the chemical formula is SiO3 or Si2O6. MgSiO3 or Mg2Si2O6 (enstatite, as compared to forsterite there is ...
... As the oxygen atoms are shared between adjacent tetrahedra, the chemical formula is SiO3 or Si2O6. MgSiO3 or Mg2Si2O6 (enstatite, as compared to forsterite there is ...