16.050 Thermal Energy
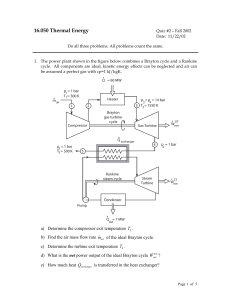
... a) Which way, if at all, does the piston move? Indicate by an arrow on a drawing in your exam book (A sentence or two is expected, perhaps bolstered by some equations). b) Is the final internal energy Ufinal of the system (both compartments together) lower, equal or higher than the initial internal ...
... a) Which way, if at all, does the piston move? Indicate by an arrow on a drawing in your exam book (A sentence or two is expected, perhaps bolstered by some equations). b) Is the final internal energy Ufinal of the system (both compartments together) lower, equal or higher than the initial internal ...
Change of state - Mrs. Coyle`s College Chemistry
... Molecules that have gone through vaporization, can return to the liquid state by colliding with the liquid surface At equilibrium, Rate of condensation = rate of evaporation There IS still condensation and evaporation going on the rates of both are equal ...
... Molecules that have gone through vaporization, can return to the liquid state by colliding with the liquid surface At equilibrium, Rate of condensation = rate of evaporation There IS still condensation and evaporation going on the rates of both are equal ...
Class notes
... New text books have elected to define one bar as the standard state for pressure ...
... New text books have elected to define one bar as the standard state for pressure ...
Ch.1 Section 1.9 Notes - Effingham County Schools
... visibly distinguishable parts consists of two or more regions called phases that differ in properties Examples: pizza, chicken noodle soup, ice cubes in water ...
... visibly distinguishable parts consists of two or more regions called phases that differ in properties Examples: pizza, chicken noodle soup, ice cubes in water ...
CHEMISTRY 313 PHYSICAL CHEMISTRY I Additional Problems for
... with its vapor in a closed vessel. (a) How many phases and components are present. (b) What is the variance of the system? Identify the independent variables. III.10. Suppose that the solution referred to in Problem III.9 is not saturated. (a) How many phases and components are present. (b) What is ...
... with its vapor in a closed vessel. (a) How many phases and components are present. (b) What is the variance of the system? Identify the independent variables. III.10. Suppose that the solution referred to in Problem III.9 is not saturated. (a) How many phases and components are present. (b) What is ...
Name:_____________ Chemistry 114 Second Hour Exam
... intermolecular interactions and make it harder to go into the vapor phase, thus lowering its vapor pressure. NaCl has a lower melting point than MgS. Both are ionic and the ions are the same size, but MgS had +2 and -2 ions, making its lattice energy stronger so it will have stonger interaction in t ...
... intermolecular interactions and make it harder to go into the vapor phase, thus lowering its vapor pressure. NaCl has a lower melting point than MgS. Both are ionic and the ions are the same size, but MgS had +2 and -2 ions, making its lattice energy stronger so it will have stonger interaction in t ...
Chromatography
... Separatory Funnels: To separate liquid-liquid mixtures that have different densities. Typically, one of the phases will be aqueous, and the other a fatty, organic solvent Most organic solvents float on top of an aqueous phase, ...
... Separatory Funnels: To separate liquid-liquid mixtures that have different densities. Typically, one of the phases will be aqueous, and the other a fatty, organic solvent Most organic solvents float on top of an aqueous phase, ...
Fractional Distillation
... In practical terms, it is important to keep the fractionating column very hot. We can do this by wrapping the column in glass wool and by shinning a heat lamp on it. The efficiency of a fractionating column is typically reported in terms of its Hold-Up and its Number of Theoretical Plates. The Hold- ...
... In practical terms, it is important to keep the fractionating column very hot. We can do this by wrapping the column in glass wool and by shinning a heat lamp on it. The efficiency of a fractionating column is typically reported in terms of its Hold-Up and its Number of Theoretical Plates. The Hold- ...
Study Sheet
... Define molarity, molality, mole fraction, weight percent, ppm Convert one concentration into another Realize when density is needed for these calculations Define unsaturated, saturated, and supersaturated. Compare these terms with dilute and concentrated. (AgNO3 970 g/100g & AgCl .00127 g/100g) Soli ...
... Define molarity, molality, mole fraction, weight percent, ppm Convert one concentration into another Realize when density is needed for these calculations Define unsaturated, saturated, and supersaturated. Compare these terms with dilute and concentrated. (AgNO3 970 g/100g & AgCl .00127 g/100g) Soli ...
Predictions of binary mixtures of noble gases and n
... Transferrable force fields, based on the n-6 Lennard Jones potential, are presented for noble gases. By using tuning the repulsive exponent the presented two body potential can predict vapor pressures and saturated liquid densities with a high degree of accuracy [1-2] without the use of blending par ...
... Transferrable force fields, based on the n-6 Lennard Jones potential, are presented for noble gases. By using tuning the repulsive exponent the presented two body potential can predict vapor pressures and saturated liquid densities with a high degree of accuracy [1-2] without the use of blending par ...
review/theory of separation (mass transfer) unit-ops
... Purpose: Separate components based on volatility Method: Selective evaporation and condensation Types: Batch & continuous ...
... Purpose: Separate components based on volatility Method: Selective evaporation and condensation Types: Batch & continuous ...
Expt. 5: Binary Phase Diagram CHEM 366 V-1 Binary Solid
... may behave more or less independent of each other but merely diluted, i.e., an ideal solution or mixture, or there may be substantial chemical interaction or complex formation between the constituents. The study of such mixtures can lead to an understanding of the most fundamental intermolecular int ...
... may behave more or less independent of each other but merely diluted, i.e., an ideal solution or mixture, or there may be substantial chemical interaction or complex formation between the constituents. The study of such mixtures can lead to an understanding of the most fundamental intermolecular int ...
Find your NEW seats Bellringer: Please complete Ms - Parkway C-2
... Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in which molecules anywhere in the liquid escape, resulting in the formation of vapor bubbles within the liquid. • During evap ...
... Evaporation is a surface phenomenon in which molecules located near the liquid's edge, escape into the surroundings as vapor (a gas). On the other hand, boiling is a process in which molecules anywhere in the liquid escape, resulting in the formation of vapor bubbles within the liquid. • During evap ...
Summary of Gas Laws
... fraction of molecules that possess enough kinetic energy to break away into a gas phase This process is called evaporation Therefore, some part of a liquid substance is always present in form of vapor (gas phase) over the surface of the liquid Gas phase molecules can strike the liquid surface and be ...
... fraction of molecules that possess enough kinetic energy to break away into a gas phase This process is called evaporation Therefore, some part of a liquid substance is always present in form of vapor (gas phase) over the surface of the liquid Gas phase molecules can strike the liquid surface and be ...
Chapter 6 - Department of Chemical Engineering
... More than often, mixtures consisting of at least two components form an azeotrope, which occurs when liquid and vapor fractions cannot be changed during distillation [2]. An azeotrope can result in two conditions, where the mixture can boil at either higher or lower temperature than the boiling poin ...
... More than often, mixtures consisting of at least two components form an azeotrope, which occurs when liquid and vapor fractions cannot be changed during distillation [2]. An azeotrope can result in two conditions, where the mixture can boil at either higher or lower temperature than the boiling poin ...
Pre-AP Chemistry Kinetic Theory and Heat Quiz
... 1. According to the kinetic theory, the primary difference in the phases of matter (of the same substance) is the __speed_ of the particles. Thus, the two factors that influence which state of matter exists are __pressure_ and temperature. 2. The higher the energy of the particles, the faster they m ...
... 1. According to the kinetic theory, the primary difference in the phases of matter (of the same substance) is the __speed_ of the particles. Thus, the two factors that influence which state of matter exists are __pressure_ and temperature. 2. The higher the energy of the particles, the faster they m ...
Properties of pure substances: introduction, PV diagrams with phase
... • A property is characteristic of the system such as specific volume (v), temperature (T), pressure (P), (specific) internal energy (u). • A state is the condition of a system as determined by its properties. • A simple compressible system is a system whose only mode of performing quasi-equilibrium ...
... • A property is characteristic of the system such as specific volume (v), temperature (T), pressure (P), (specific) internal energy (u). • A state is the condition of a system as determined by its properties. • A simple compressible system is a system whose only mode of performing quasi-equilibrium ...
The Liquid-Vapor Phase Diagram for Ethanol
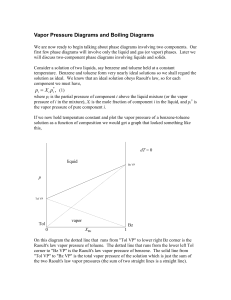
... Once A,az and B,az have been determined, the van Laar coefficients AL and BL can be calculated from equations (14) and (15). Now equations (12) and (13) can be used to calculate model values for A and B at any other system composition. These model coefficients can then be used to model the entir ...
... Once A,az and B,az have been determined, the van Laar coefficients AL and BL can be calculated from equations (14) and (15). Now equations (12) and (13) can be used to calculate model values for A and B at any other system composition. These model coefficients can then be used to model the entir ...