EFFECTS OF THE CHANGES IN SLOPE OCCURRING ON
... 1133° C. However, revised data for the whole system are not available, and it will be assumed in the following discussion that the system remains ternary. Since most of the compositions considered lie in the plagioclase field, departures from ternary behavior probably have little effect on the gener ...
... 1133° C. However, revised data for the whole system are not available, and it will be assumed in the following discussion that the system remains ternary. Since most of the compositions considered lie in the plagioclase field, departures from ternary behavior probably have little effect on the gener ...
Ch 12 Solutions
... nonpolar molecules. So, the two substances will remain separated. Entropy - Entropy is a measure of the natural tendency towards disorder. - The amount of overall entropy always increases as a result of spontaneous reactions. - Substances will mix and become disordered if no forces prevent them from ...
... nonpolar molecules. So, the two substances will remain separated. Entropy - Entropy is a measure of the natural tendency towards disorder. - The amount of overall entropy always increases as a result of spontaneous reactions. - Substances will mix and become disordered if no forces prevent them from ...
GUIDELINES FOR PUBLICATION OF EQUATIONS OF STATE
... that wide-range empirical equations which accurately represent all the critically assessed data will meet all criteria, whereas theoretical equations will usually trade compliance to some criteria for applicability to a wider range of substances. Since these criteria are of importance to many of the ...
... that wide-range empirical equations which accurately represent all the critically assessed data will meet all criteria, whereas theoretical equations will usually trade compliance to some criteria for applicability to a wider range of substances. Since these criteria are of importance to many of the ...
LECTURE_pptnotes Fipps Stochiometry
... multiply each by a factor that will make them whole numbers. Look for these fractions: ◦ 0.5 x 2 ◦ 0.33 x 3 ◦ 0.25 x 4 ...
... multiply each by a factor that will make them whole numbers. Look for these fractions: ◦ 0.5 x 2 ◦ 0.33 x 3 ◦ 0.25 x 4 ...
WA AP Chem gas law IMF MC Set C
... B. The intermolecular forces, such as hydrogen bonds, between the H2O (l) molecules require energy to be overcome, and the molecules can only separate to become a gas once these intermolecular forces are overcome. C. Water vapor is hotter than or equal in temperature to water at constant pressure, a ...
... B. The intermolecular forces, such as hydrogen bonds, between the H2O (l) molecules require energy to be overcome, and the molecules can only separate to become a gas once these intermolecular forces are overcome. C. Water vapor is hotter than or equal in temperature to water at constant pressure, a ...
Lab announcements – 2 lab quiz week before spring break
... photocopy before turning in or study before turning in -same as 101/other 102 sections be extra careful with dilutions this week (Expt. 21) Most chemical reactions do not go to completion. chemical equilibrium – two opposing reactions occur simultaneously at the same rate ‘equilibrium’ doesn’t neces ...
... photocopy before turning in or study before turning in -same as 101/other 102 sections be extra careful with dilutions this week (Expt. 21) Most chemical reactions do not go to completion. chemical equilibrium – two opposing reactions occur simultaneously at the same rate ‘equilibrium’ doesn’t neces ...
Chemical Equilibrium
... Note: You could also calculate Kp by using the ideal gas law - more on that later. ...
... Note: You could also calculate Kp by using the ideal gas law - more on that later. ...
Equilib - C.R.C.T.
... between H2O(g) [ideal gas phase] and H2O(L1) [i.e., the ideal boiling point] Equilib 9.2 ...
... between H2O(g) [ideal gas phase] and H2O(L1) [i.e., the ideal boiling point] Equilib 9.2 ...
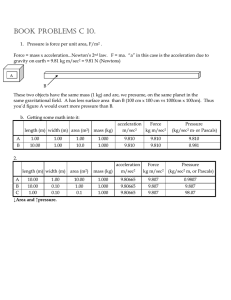
book problems c 10.
... atomic mass, and the unit cell length, determined from x-ray methods. To be useful for this purpose, the crystal must be free of defects. Very accurate values of these quantities for silicon have been measured at the National Institute for Standards and Technology (NIST). To use this approach, it is ...
... atomic mass, and the unit cell length, determined from x-ray methods. To be useful for this purpose, the crystal must be free of defects. Very accurate values of these quantities for silicon have been measured at the National Institute for Standards and Technology (NIST). To use this approach, it is ...
Chemistry 180-213B Introductory Physical
... points above zero and assign their temperature difference and the other is to use one fixed point and assign its numerical value. Until recently the calibration of the Kelvin temperature scale was performed using two fixed points: the ice point T 0 o K and the boiling point T 0 + 100o K of pure wate ...
... points above zero and assign their temperature difference and the other is to use one fixed point and assign its numerical value. Until recently the calibration of the Kelvin temperature scale was performed using two fixed points: the ice point T 0 o K and the boiling point T 0 + 100o K of pure wate ...
2.00atm x 1 .00L 0.0821 L.atm.mol K 298.15 = 8.17x10 mol. U = 8
... Answers: At a depth h, the pressure p = hρg, in which ρ is the liquid density and g = 9.8 m s–2. The density of sea water is about 1027 kg/m3. Hence the pressure: p = 1.5x103m (1027 kg/m3) (9.8 m s–2) = 1.5x107 Pa. = 149. 5 atm ( 1.0 atm = 101.3 kPa). In the oceans, the pressure increases by about 1 ...
... Answers: At a depth h, the pressure p = hρg, in which ρ is the liquid density and g = 9.8 m s–2. The density of sea water is about 1027 kg/m3. Hence the pressure: p = 1.5x103m (1027 kg/m3) (9.8 m s–2) = 1.5x107 Pa. = 149. 5 atm ( 1.0 atm = 101.3 kPa). In the oceans, the pressure increases by about 1 ...
Solidification in heat packs: I. Nucleation rate
... For predicted numbers well below unity, such as for the stable subcooled states in this work, there will almost always be zero incipient nuclei present, very occasionally one, and effectively never more than one. Neglecting any probability of finding more than one incipient nucleus means the value o ...
... For predicted numbers well below unity, such as for the stable subcooled states in this work, there will almost always be zero incipient nuclei present, very occasionally one, and effectively never more than one. Neglecting any probability of finding more than one incipient nucleus means the value o ...
EquiSage
... between H2O(g) [ideal gas phase] and H2O(L1) [i.e., the ideal boiling point] Equilib 9.2 ...
... between H2O(g) [ideal gas phase] and H2O(L1) [i.e., the ideal boiling point] Equilib 9.2 ...
Solutions
... concentration (in the case of ideal mixtures, only molar fractions, and sometimes mass fractions, were common). Generically speaking, concentrations in a mixture express the quantitative composition (although, under the SI standard, composition must refer to the unit volume of the system, i.e. mol/m ...
... concentration (in the case of ideal mixtures, only molar fractions, and sometimes mass fractions, were common). Generically speaking, concentrations in a mixture express the quantitative composition (although, under the SI standard, composition must refer to the unit volume of the system, i.e. mol/m ...
Phase Diagram Tutorial
... Between the liquidus and solidus, we have a two-phase region: liquid, L+Sn-rich solid, (Sn). As noted earlier, in this field the compositions of the phases are fixed at a given temperature and variations in overall composition cause changes in the proportions of solid and liquid. The phase composition ...
... Between the liquidus and solidus, we have a two-phase region: liquid, L+Sn-rich solid, (Sn). As noted earlier, in this field the compositions of the phases are fixed at a given temperature and variations in overall composition cause changes in the proportions of solid and liquid. The phase composition ...
Properties_problems 5
... where Ev is defined as the energy change upon isothermal vaporization of the saturated liquid to the ideal gas state at infinite dilution and Vi is the molar volume of the liquid. The solubility parameter of a polymer has to be determined indirectly or calculated by group-contribution methods. Calc ...
... where Ev is defined as the energy change upon isothermal vaporization of the saturated liquid to the ideal gas state at infinite dilution and Vi is the molar volume of the liquid. The solubility parameter of a polymer has to be determined indirectly or calculated by group-contribution methods. Calc ...
Physcal Chemistry ERT 108 semester II 2010/2011
... • No unbalanced forces act on or within the system; hence the system undergoes no acceleration, and there is no turbulence within the system. 2. Material equilibrium • No net chemical reactions are occurring in the system, nor is there any net transfer of matter from one part of the system to anothe ...
... • No unbalanced forces act on or within the system; hence the system undergoes no acceleration, and there is no turbulence within the system. 2. Material equilibrium • No net chemical reactions are occurring in the system, nor is there any net transfer of matter from one part of the system to anothe ...
Mole Fraction Molality Molarity
... The main effect of making a solution is that the entropy of the solution is higher than the separate solvent and solute T = 0°C and P = 1 atm G ...
... The main effect of making a solution is that the entropy of the solution is higher than the separate solvent and solute T = 0°C and P = 1 atm G ...
The Mole
... Double check the equation. I have seen lots of students go right ahead and solve using the unbalanced equation supplied in the problem (or test question for that matter). DON'T use the same molar mass in steps two and four. Don't multiply the molar mass of a substance by the coefficient in the probl ...
... Double check the equation. I have seen lots of students go right ahead and solve using the unbalanced equation supplied in the problem (or test question for that matter). DON'T use the same molar mass in steps two and four. Don't multiply the molar mass of a substance by the coefficient in the probl ...
Moles 2016
... 1) Obtain the mass of each element in the compound in grams. If percentages are given, just assume a 100 g sample and the masses will be equal to the percentages. ...
... 1) Obtain the mass of each element in the compound in grams. If percentages are given, just assume a 100 g sample and the masses will be equal to the percentages. ...
1970 - Warren County Schools
... p-dichlorobenzene (molecular weight 147.0) in Al(OH)3 is amphoteric. The product is a hydroxo25.86 grams of naphthalene (molecular weight aluminate ion, Fe(OH)3 is not amphoteric. 128.2). Calculate the molality of the pdichlorobenzene solution. 1984 C Give a scientific explanation for the following ...
... p-dichlorobenzene (molecular weight 147.0) in Al(OH)3 is amphoteric. The product is a hydroxo25.86 grams of naphthalene (molecular weight aluminate ion, Fe(OH)3 is not amphoteric. 128.2). Calculate the molality of the pdichlorobenzene solution. 1984 C Give a scientific explanation for the following ...