The Mole - C405 Chemistry
... A substance has the following composition by mass: 60.80 % Na ; 28.60 % B ; 10.60 % H What is the empirical formula of the substance? Consider a sample size of 100 grams This will contain 60.80 grams of Na, 28.60 grams of B and 10.60 grams H Determine the number of moles of each Determine the simple ...
... A substance has the following composition by mass: 60.80 % Na ; 28.60 % B ; 10.60 % H What is the empirical formula of the substance? Consider a sample size of 100 grams This will contain 60.80 grams of Na, 28.60 grams of B and 10.60 grams H Determine the number of moles of each Determine the simple ...
Chapter 11 Absorption Air Conditioners
... The crystallization limit shown in Figures 8.2a and 8.2b represents the condition for which the lithium bromide solution is too highly concentrated to remain liquid. For the high concentrations and low temperatures that are below this limit, crystals of lithium bromide would form in the liquid solut ...
... The crystallization limit shown in Figures 8.2a and 8.2b represents the condition for which the lithium bromide solution is too highly concentrated to remain liquid. For the high concentrations and low temperatures that are below this limit, crystals of lithium bromide would form in the liquid solut ...
Chapter 10 The Mole
... Avogadro proposed that equal volumes of gases contain the same number of gas particles at a given temperature & pressure. Therefore one mole of gas #1 would have the same volume as one mole of gas #2. It is observed that one mole of any gas occupies 22.4 liters @ STP (molar volume). STP = Standard ...
... Avogadro proposed that equal volumes of gases contain the same number of gas particles at a given temperature & pressure. Therefore one mole of gas #1 would have the same volume as one mole of gas #2. It is observed that one mole of any gas occupies 22.4 liters @ STP (molar volume). STP = Standard ...
BalanceEquationsetc
... Example: H2O → 1 mole = 18.01 grams of H2O 2 moles of H: 2 x 1.008 = 2.016 grams 1 mole of O: 1 x 16 = 16 grams % of H → 2.018 x 100% = 11.19% ...
... Example: H2O → 1 mole = 18.01 grams of H2O 2 moles of H: 2 x 1.008 = 2.016 grams 1 mole of O: 1 x 16 = 16 grams % of H → 2.018 x 100% = 11.19% ...
Conversion Problems
... The element discussed in question number one is a halogen. As a pure element, it exists as a diatomic molecule (at room temperature and 1 atm pressure it is most stable as a liquid). (a) What is the average mass of each of these diatomic molecules (in amu)? ans.: if two average atoms combine they wi ...
... The element discussed in question number one is a halogen. As a pure element, it exists as a diatomic molecule (at room temperature and 1 atm pressure it is most stable as a liquid). (a) What is the average mass of each of these diatomic molecules (in amu)? ans.: if two average atoms combine they wi ...
Introduction to Kinetics and Equilibrium
... Introduction to Kinetics and Equilibrium Kinetics and equilibrium are two of the most important areas in chemistry Entire books and important areas in chemistry. Entire books and courses at the undergraduate and graduate level are devoted to them. Chemical kinetics – the study of the rates of ...
... Introduction to Kinetics and Equilibrium Kinetics and equilibrium are two of the most important areas in chemistry Entire books and important areas in chemistry. Entire books and courses at the undergraduate and graduate level are devoted to them. Chemical kinetics – the study of the rates of ...
The Physical Chemistry, Theory and Technique of
... fugacity are all relative concepts, e.g., a solution does not have an osmotic pressure unless there is a semipermeable membrane present and another solution on the other side. When talking about these terms, then, reference must be made to the particular system being studied. ...
... fugacity are all relative concepts, e.g., a solution does not have an osmotic pressure unless there is a semipermeable membrane present and another solution on the other side. When talking about these terms, then, reference must be made to the particular system being studied. ...
Physical Chemistry 2.pdf
... The module, Physical Chemistry 2, focuses on five (5) areas of physical chemistry important to many aspects of our lives: solutions, colloids, phase equilibrium, electrochemistry and nuclear chemistry. Solutions are often necessary to facilitate many chemical reactions in life processes or industry ...
... The module, Physical Chemistry 2, focuses on five (5) areas of physical chemistry important to many aspects of our lives: solutions, colloids, phase equilibrium, electrochemistry and nuclear chemistry. Solutions are often necessary to facilitate many chemical reactions in life processes or industry ...
The Mole & Stoicheometry
... • What is the molecular formula of a substance that has an empirical formula of AgCO2and a formula mass of 304? • The formula mass of the empirical unit, AgCO2, is 152. If we divide the formula mass 304 by 152, we get 2. Therefore, the molecular formula must be 2 times the empirical formula, or Ag2C ...
... • What is the molecular formula of a substance that has an empirical formula of AgCO2and a formula mass of 304? • The formula mass of the empirical unit, AgCO2, is 152. If we divide the formula mass 304 by 152, we get 2. Therefore, the molecular formula must be 2 times the empirical formula, or Ag2C ...
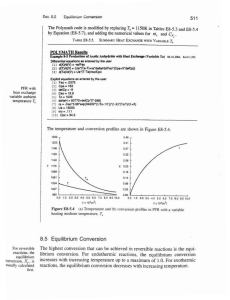
8-5 Equilibrium Conversion
... the inlet temperature were lowered to 500 K, the corresponding equilibrium conversion is increased to 0.38; however, the reaction rate is slower at this lower ternpentuse so that this conversion is not achieved until closer to the end of the reactor. V the entering temperature were lowered further t ...
... the inlet temperature were lowered to 500 K, the corresponding equilibrium conversion is increased to 0.38; however, the reaction rate is slower at this lower ternpentuse so that this conversion is not achieved until closer to the end of the reactor. V the entering temperature were lowered further t ...
Principles of Reactivity: Chemical Equilibria
... the equilibrium constant for the new equation (Knew) is the old equilibrium constant (Kold) raised to the power of the multiplication factor. The equilibrium constants for a reaction and its reverse are the reciprocals of each other. When two or more chemical equations are added to produce a net equ ...
... the equilibrium constant for the new equation (Knew) is the old equilibrium constant (Kold) raised to the power of the multiplication factor. The equilibrium constants for a reaction and its reverse are the reciprocals of each other. When two or more chemical equations are added to produce a net equ ...
Determination of Complete Melting and Surface Premelting
... determine the onset temperature of surface premelting (Tsm) for NPs in terms of the mean-field approximation.26 Complete melting points (Tm) of Ag NPs obtained experimentally have shown scattered data in the range of 383 to 953 K,27−32 while the only published molecular dynamics (MD) simulation was p ...
... determine the onset temperature of surface premelting (Tsm) for NPs in terms of the mean-field approximation.26 Complete melting points (Tm) of Ag NPs obtained experimentally have shown scattered data in the range of 383 to 953 K,27−32 while the only published molecular dynamics (MD) simulation was p ...
AT 25 °C - University of Bath
... Hence, at 450 °C, 108.24 kJ is evolved for each mole of the equation i.e 108.24 kJ is evolved per mole of nitrogen which reacts. Since, for a gas, volume ∝ number of moles, the 1:3 mixture is in the stoichiometric amount. At 0 °C and 1 atm pressure, 1 mole occupies 22.4 dm3. Thus, the total amount o ...
... Hence, at 450 °C, 108.24 kJ is evolved for each mole of the equation i.e 108.24 kJ is evolved per mole of nitrogen which reacts. Since, for a gas, volume ∝ number of moles, the 1:3 mixture is in the stoichiometric amount. At 0 °C and 1 atm pressure, 1 mole occupies 22.4 dm3. Thus, the total amount o ...
mole concept type 1 - teko classes bhopal
... If volume of a gas is given along with its temperature (T) and pressure (P) PV RT where R = 0.0821 lit-atm/mol-K (when P is in atmosphere and V is in litre.) ...
... If volume of a gas is given along with its temperature (T) and pressure (P) PV RT where R = 0.0821 lit-atm/mol-K (when P is in atmosphere and V is in litre.) ...
Temperature Dependence of Viscosity and Density of cis-1,4/trans
... Table 2. Values of Lg η ≡ log 10 ( f(T )/mPa s) calculated with Eq. (1) using the parameters in Table 1 at temperatures given in column 2. The values in brackets are extrapolated. The minimum and maximum measured values as well as their ratio (ηmin [mPa s], ηmax [mPa s], ηmax /ηmin ) are given for a ...
... Table 2. Values of Lg η ≡ log 10 ( f(T )/mPa s) calculated with Eq. (1) using the parameters in Table 1 at temperatures given in column 2. The values in brackets are extrapolated. The minimum and maximum measured values as well as their ratio (ηmin [mPa s], ηmax [mPa s], ηmax /ηmin ) are given for a ...
C - Thierry Karsenti
... The module, Physical Chemistry 2, focuses on five (5) areas of physical chemistry important to many aspects of our lives: solutions, colloids, phase equilibrium, electrochemistry and nuclear chemistry. Solutions are often necessary to facilitate many chemical reactions in life processes or industry ...
... The module, Physical Chemistry 2, focuses on five (5) areas of physical chemistry important to many aspects of our lives: solutions, colloids, phase equilibrium, electrochemistry and nuclear chemistry. Solutions are often necessary to facilitate many chemical reactions in life processes or industry ...
CHEM 122 - Nmt.edu
... amount of water to be close to 1 L; ~1 kg. What is the elevation in the boiling point of this solution? For water, Kb = 0.521 oC/m. a) ...
... amount of water to be close to 1 L; ~1 kg. What is the elevation in the boiling point of this solution? For water, Kb = 0.521 oC/m. a) ...
Chapter 11 Notes - Mr. Julien`s Homepage
... b. At higher temperatures, gas particles hit the walls of the container more often and with more force, producing higher pressures. 6. Kinetic Molecular Theory of Gases— a. Molecules from a perfume bottle move at about 1000 miles per hour when escaping from the bottle at room temperature. B. Pressur ...
... b. At higher temperatures, gas particles hit the walls of the container more often and with more force, producing higher pressures. 6. Kinetic Molecular Theory of Gases— a. Molecules from a perfume bottle move at about 1000 miles per hour when escaping from the bottle at room temperature. B. Pressur ...
Gases - chemmybear.com
... (b) SO2 is the least ideal gas. It has the largest size or volume. It has the strongest attractive forces (van der Waals forces or dipole-dipole interactions). (c) High temperature result in high kinetic energies. This energy overcomes the attractive forces. Low pressure increases the distance betwe ...
... (b) SO2 is the least ideal gas. It has the largest size or volume. It has the strongest attractive forces (van der Waals forces or dipole-dipole interactions). (c) High temperature result in high kinetic energies. This energy overcomes the attractive forces. Low pressure increases the distance betwe ...