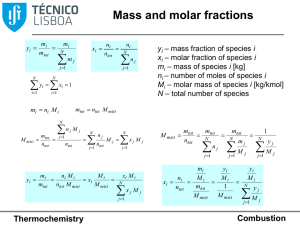
Document
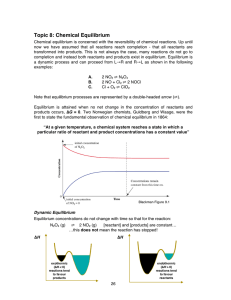
... • The proportion of CO2, CO and O2 at a particular temperature and pressure adjusts itself such that, after some time, the reaction rates of the direct and inverse reactions are equal, i.e., the number of moles of CO2 that dissociate, according to the direct reaction, is equal to the number of moles ...
... • The proportion of CO2, CO and O2 at a particular temperature and pressure adjusts itself such that, after some time, the reaction rates of the direct and inverse reactions are equal, i.e., the number of moles of CO2 that dissociate, according to the direct reaction, is equal to the number of moles ...
Chapter 6-States of Matter: Gases, Liquids, and Solids
... bonding, dipole-dipole attraction, Londodn dispersion forces. Such a system can only exist as a gas. Any real system will approach ideal gas behavior in the limit that the pressure is extremely low and the temperature is high enough to overcome attractive intermolecular forces. Measurement of Gases ...
... bonding, dipole-dipole attraction, Londodn dispersion forces. Such a system can only exist as a gas. Any real system will approach ideal gas behavior in the limit that the pressure is extremely low and the temperature is high enough to overcome attractive intermolecular forces. Measurement of Gases ...
Evaporation–glow discharge hybrid source for plasma immersion
... waveforms exhibit two spikes at the beginning and at the end of the pulse. These spikes are the sums of the true implanted ion current, secondary electron current, as well as system capacitance. To subtract the system capacitance, Fig. 3c,d shows the spike current waveforms attributable to the syste ...
... waveforms exhibit two spikes at the beginning and at the end of the pulse. These spikes are the sums of the true implanted ion current, secondary electron current, as well as system capacitance. To subtract the system capacitance, Fig. 3c,d shows the spike current waveforms attributable to the syste ...
Equilibrium - Clayton State University
... Higher moles of gaseous reactants than products (∆n is negative) - Increase in pressure shifts the equilibrium position to the right - Decrease in pressure shifts the equilibrium position to the left Higher moles of gaseous products than reactants (∆n is positive) - Increase in pressure shifts the e ...
... Higher moles of gaseous reactants than products (∆n is negative) - Increase in pressure shifts the equilibrium position to the right - Decrease in pressure shifts the equilibrium position to the left Higher moles of gaseous products than reactants (∆n is positive) - Increase in pressure shifts the e ...
b - PianetaChimica
... questions in this section on the multiple choice answer sheet, using a 2B pencil. Only one choice is allowed per question. If you make a mistake make sure that your incorrect answer is completely erased. Please note the following: • It is recommended that you first record your answer on this questio ...
... questions in this section on the multiple choice answer sheet, using a 2B pencil. Only one choice is allowed per question. If you make a mistake make sure that your incorrect answer is completely erased. Please note the following: • It is recommended that you first record your answer on this questio ...
Analysis of selected gaseous organic micro
... step) depends on the desorption time and the temperature of the injection port of the gas chromatograph. These parameters were determined in the previous studies [10]. The desorption temperature applied (250 °C) was within a 200 − 270 °C recommended range and below a 280 °C maximum working temperatu ...
... step) depends on the desorption time and the temperature of the injection port of the gas chromatograph. These parameters were determined in the previous studies [10]. The desorption temperature applied (250 °C) was within a 200 − 270 °C recommended range and below a 280 °C maximum working temperatu ...
Physical Chemistry Lab (Real Gas Behavior)
... error is due to a leak in the vessel. A leak would mean that less pressure and less CO2 would be in the vessel at the time of weighing, which would ultimately increase the Z value. This explains why the y-intercept is very positive as opposed to being closer to 1. The difference in the literature va ...
... error is due to a leak in the vessel. A leak would mean that less pressure and less CO2 would be in the vessel at the time of weighing, which would ultimately increase the Z value. This explains why the y-intercept is very positive as opposed to being closer to 1. The difference in the literature va ...
- sartep.com
... (A) Cu has two common oxidation states, but Ag has only one. (B) Cu has a higher melting point than Au has, but Ag has a lower melting point than Au has. (C) Cu atoms are smaller than Ag atoms, thus they interfere more with the displacement of atoms in the alloy. (D) Cu atoms are less polarizable th ...
... (A) Cu has two common oxidation states, but Ag has only one. (B) Cu has a higher melting point than Au has, but Ag has a lower melting point than Au has. (C) Cu atoms are smaller than Ag atoms, thus they interfere more with the displacement of atoms in the alloy. (D) Cu atoms are less polarizable th ...
for high volume liquid phosphate fertilizer applications for use on
... methods from those normally used are tried, test strips are recommended until such new DO NOT USE AVAIL HV for LIQUID PHOSPHATE methods have been proven to be effective. FERTILIZERS on granular phosphate fertilizers. AVAIL HV for LIQUID PHOSPHATE FERTILIZERS SOIL TYPES AND CROPS may be used with liq ...
... methods from those normally used are tried, test strips are recommended until such new DO NOT USE AVAIL HV for LIQUID PHOSPHATE methods have been proven to be effective. FERTILIZERS on granular phosphate fertilizers. AVAIL HV for LIQUID PHOSPHATE FERTILIZERS SOIL TYPES AND CROPS may be used with liq ...
Writing Equilibrium Cons... and Liquids - Chemwiki
... No state symbols have been given, but they will be all (g), or all (l), or all (aq) if the reaction was between substances in solution in water. If you allow this reaction to reach equilibrium and then measure the equilibrium concentrations of everything, you can combine these concentrations into an ...
... No state symbols have been given, but they will be all (g), or all (l), or all (aq) if the reaction was between substances in solution in water. If you allow this reaction to reach equilibrium and then measure the equilibrium concentrations of everything, you can combine these concentrations into an ...
Sample Exercise 19.1 Identifying Spontaneous Processes
... (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and oxygen gases spontaneously bubbling up out of water! Rather, the reverse process—the reaction o ...
... (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and oxygen gases spontaneously bubbling up out of water! Rather, the reverse process—the reaction o ...
Unit E Chemical Quantities
... • Gram atomic mass (gam) is the mass of 1 mole of atoms • Gram Molecular Mass (gmm) is the mass of 1 mole of molecules (covalent only!) • Gram Formula Mass (gfm) is the mass of 1 mole of ionic compounds These are all examples of the same thing: ...
... • Gram atomic mass (gam) is the mass of 1 mole of atoms • Gram Molecular Mass (gmm) is the mass of 1 mole of molecules (covalent only!) • Gram Formula Mass (gfm) is the mass of 1 mole of ionic compounds These are all examples of the same thing: ...
The Mole - Cloudfront.net
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
Growing Negative Pressure in Dissolved Solutes: Raman - HAL-Insu
... Raman spectrum was obtained with an accumulation time of 120s. Spectra were treated with the Renishaw Wire 3 software. The sensitivity and the stability of such recent Raman spectrometer make us able to extract unambiguously even very small features, which can be completely masked by noise and fluct ...
... Raman spectrum was obtained with an accumulation time of 120s. Spectra were treated with the Renishaw Wire 3 software. The sensitivity and the stability of such recent Raman spectrometer make us able to extract unambiguously even very small features, which can be completely masked by noise and fluct ...
Slide 1
... (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and oxygen gases spontaneously bubbling up out of water! Rather, the reverse process—the reaction o ...
... (thermal equilibrium), will be somewhere between the initial temperatures of the metal and the water. (b) Experience tells us that this process is not spontaneous—we certainly have never seen hydrogen and oxygen gases spontaneously bubbling up out of water! Rather, the reverse process—the reaction o ...
UNITS OF CONCENTRATION
... weight is half of the molecular weight. Similarly, for bases an equivalent amount of a base is defined as the amount of base that neutralizes one mole of H+. For CaCO3, one equivalent is ½ of one mole, since each mole of CaCO3 neutralizes two moles of H+. And again, the equivalent weight is ½ the mo ...
... weight is half of the molecular weight. Similarly, for bases an equivalent amount of a base is defined as the amount of base that neutralizes one mole of H+. For CaCO3, one equivalent is ½ of one mole, since each mole of CaCO3 neutralizes two moles of H+. And again, the equivalent weight is ½ the mo ...
Paper 41
... For the experimental data about soot mass growth rates, two temperature ranges can be distinguished: i) T < 2000 K, and ii) 2000 K < T < 2300 K. At T < 2000 K, the soot growth rate kf /[C] increases linearly with 1/T. These result holds for pure C6 H6, as well as for C6H6 /O2 mixtures. An activation ...
... For the experimental data about soot mass growth rates, two temperature ranges can be distinguished: i) T < 2000 K, and ii) 2000 K < T < 2300 K. At T < 2000 K, the soot growth rate kf /[C] increases linearly with 1/T. These result holds for pure C6 H6, as well as for C6H6 /O2 mixtures. An activation ...
Analytical Chemistry
... Since molarity involves a basis of solution volume, it is apparent that the molarity of a solution will change as volume changes which is associated with changes in temperature. Formal Concentration: Some substances do not exist in molecular form, whether in solid or solution form, they remain in io ...
... Since molarity involves a basis of solution volume, it is apparent that the molarity of a solution will change as volume changes which is associated with changes in temperature. Formal Concentration: Some substances do not exist in molecular form, whether in solid or solution form, they remain in io ...
Experiment 16: Spectrophotometric Determination of an Equilibrium Constant
... place a √ next to this wavelength. Choose Single 10 nm Band from the drop-down menu on the left. Click on OK. Fill the cuvet with your assigned standard solution, and place it in the SVP. Click once on the green arrow button. A circular button that is blue will appear next to the red rectangle. Watc ...
... place a √ next to this wavelength. Choose Single 10 nm Band from the drop-down menu on the left. Click on OK. Fill the cuvet with your assigned standard solution, and place it in the SVP. Click once on the green arrow button. A circular button that is blue will appear next to the red rectangle. Watc ...
Unit 13 Stoichiometry (Theoretical)
... f. What is the mole ratio between water and calcium hydroxide? 9. Methanol (CH4O) is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure, according to the following chemical equation: CO (g) + H2 (g) CH4O (g) a. Balan ...
... f. What is the mole ratio between water and calcium hydroxide? 9. Methanol (CH4O) is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure, according to the following chemical equation: CO (g) + H2 (g) CH4O (g) a. Balan ...
Chemistry Entrance Material for Grade 10 to 11 Answer Key
... Chapter 2: Revision of the Scientific Method In all multiple choice questions, more than answer could be correct Section №: 1 Experiments and Generalizations Know what an experiment is 01. An experiment is defined as: [-A-] controlled sequence of events [-B-] a rule framed on a collection of indivi ...
... Chapter 2: Revision of the Scientific Method In all multiple choice questions, more than answer could be correct Section №: 1 Experiments and Generalizations Know what an experiment is 01. An experiment is defined as: [-A-] controlled sequence of events [-B-] a rule framed on a collection of indivi ...