Atomic Structure LO Teacher
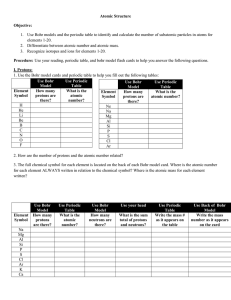
... Particles of the Atom The atom contains subatomic particles, which are very small particles that make up an atom. Three of these types of particles we have seen already: protons, neutrons, and electrons. The identity of an atom is determined by how many protons it has. atomic number = the number of ...
... Particles of the Atom The atom contains subatomic particles, which are very small particles that make up an atom. Three of these types of particles we have seen already: protons, neutrons, and electrons. The identity of an atom is determined by how many protons it has. atomic number = the number of ...
Atomic Structure LO Teacher
... Particles of the Atom The atom contains subatomic particles, which are very small particles that make up an atom. Three of these types of particles we have seen already: protons, neutrons, and electrons. The identity of an atom is determined by how many protons it has. atomic number = the number of ...
... Particles of the Atom The atom contains subatomic particles, which are very small particles that make up an atom. Three of these types of particles we have seen already: protons, neutrons, and electrons. The identity of an atom is determined by how many protons it has. atomic number = the number of ...
Atomic masses
... Alpha particles: carry two fundamental units of positive charge and have the same mass as helium atoms. They have +2 charges. Beta particles: are negatively charged particles produced by changes occuring within the nuclei of radioactive atoms and have the same properties as electrons. Gama rays are ...
... Alpha particles: carry two fundamental units of positive charge and have the same mass as helium atoms. They have +2 charges. Beta particles: are negatively charged particles produced by changes occuring within the nuclei of radioactive atoms and have the same properties as electrons. Gama rays are ...
Atomic Structure_Bohr Flashcards
... 1. Use the Bohr model cards and periodic table to help you fill out the following tables: Use Bohr Use Periodic Use Bohr Use Periodic Model Table Model Table Element How many What is the Element How many What is the Symbol protons are atomic Symbol protons are atomic number? there? number? there? H ...
... 1. Use the Bohr model cards and periodic table to help you fill out the following tables: Use Bohr Use Periodic Use Bohr Use Periodic Model Table Model Table Element How many What is the Element How many What is the Symbol protons are atomic Symbol protons are atomic number? there? number? there? H ...
chap7_nucleus
... Radioactivity must be associated with atomic nuclei because these are the only parts of atoms not affected by such treatments. The radioactivity of an element is due to the radioactivity of one or more of its isotopes. A nucleus is said to decay when it emits an alpha or beta particle or a gamma ray ...
... Radioactivity must be associated with atomic nuclei because these are the only parts of atoms not affected by such treatments. The radioactivity of an element is due to the radioactivity of one or more of its isotopes. A nucleus is said to decay when it emits an alpha or beta particle or a gamma ray ...
CHAPTER 11 Introduction to Atoms
... Scientists must determine the atomic number, or the number of protons, in the newly formed nucleus. The nucleus is that of a new element only if the number of protons is different from all known elements. ...
... Scientists must determine the atomic number, or the number of protons, in the newly formed nucleus. The nucleus is that of a new element only if the number of protons is different from all known elements. ...
Using the Periodic Table
... Using the Periodic Table • In a neutral atom (one with no electric charge) the atomic number is also the number of electrons – You remember electrons • The negatively charged particles orbiting the nucleus ...
... Using the Periodic Table • In a neutral atom (one with no electric charge) the atomic number is also the number of electrons – You remember electrons • The negatively charged particles orbiting the nucleus ...
mack atoms - McClymonds Chemistry
... • Electrons are negatively charged. • Electrons are much smaller and lighter than atoms. • Electrons are uniformly present in many different kinds of substances. • He proposed that atoms must contain positive charge that balanced the negative charge of electrons. ...
... • Electrons are negatively charged. • Electrons are much smaller and lighter than atoms. • Electrons are uniformly present in many different kinds of substances. • He proposed that atoms must contain positive charge that balanced the negative charge of electrons. ...
Chapter 4
... ■ Teacher- summarized results of his experiments and those of others. ■ Elements substances that can’t be broken down ■ In Dalton’s Atomic Theory ■ Combined idea of elements with that of atoms. ...
... ■ Teacher- summarized results of his experiments and those of others. ■ Elements substances that can’t be broken down ■ In Dalton’s Atomic Theory ■ Combined idea of elements with that of atoms. ...
U1 Atoms, Elements and Ions
... • As you move left to right across the table atom size decreases • Where are the largest atoms located? ...
... • As you move left to right across the table atom size decreases • Where are the largest atoms located? ...
ch03 - earthjay science
... Source materials for potassium-40 and its daughter isotope Argon 40 would include muscovite, biotite, hornblende, glauconite, potassium, feldspar, and whole volcanic rock. ...
... Source materials for potassium-40 and its daughter isotope Argon 40 would include muscovite, biotite, hornblende, glauconite, potassium, feldspar, and whole volcanic rock. ...
Atomic Structure PPQs 2
... State ONE similarity and ONE difference between these two isotopes in terms of the numbers of their fundamental particles. Similarity .......................................................................................................... Difference ................................................ ...
... State ONE similarity and ONE difference between these two isotopes in terms of the numbers of their fundamental particles. Similarity .......................................................................................................... Difference ................................................ ...
6-2 Notes: The Atom
... The charges or protons and electrons are opposite but _________, so the charges cancel out. If the numbers of electrons and protons become unequal, the atom becomes a charged particle called an ______. An atom that loses one or more electrons becomes a _______________ charged ion. An atom that gains ...
... The charges or protons and electrons are opposite but _________, so the charges cancel out. If the numbers of electrons and protons become unequal, the atom becomes a charged particle called an ______. An atom that loses one or more electrons becomes a _______________ charged ion. An atom that gains ...
PPT of Notes
... In the diagram above, ion stream A is most deflected - it will contain ions with the smallest mass/charge ratio. Ion stream C is the least deflected - it contains ions with the greatest mass/charge ratio. It makes it simpler to talk about this if we assume that the charge on all the ions is 1+. Most ...
... In the diagram above, ion stream A is most deflected - it will contain ions with the smallest mass/charge ratio. Ion stream C is the least deflected - it contains ions with the greatest mass/charge ratio. It makes it simpler to talk about this if we assume that the charge on all the ions is 1+. Most ...
Review # 3
... What is the total number of nucleons (protons and neutrons) in an atom of selenium ( at. # = 34; mass # = 79) a. 34 b. 45 c. 79 d. 113 ...
... What is the total number of nucleons (protons and neutrons) in an atom of selenium ( at. # = 34; mass # = 79) a. 34 b. 45 c. 79 d. 113 ...
Atomic Number
... Atomic Number The atomic number • is a whole number specific for each element. • is the same for all atoms of an element. • is equal to the number of protons in an atom. • appears above the symbol of an element in the periodic table. Atomic number ...
... Atomic Number The atomic number • is a whole number specific for each element. • is the same for all atoms of an element. • is equal to the number of protons in an atom. • appears above the symbol of an element in the periodic table. Atomic number ...
Atomic Theory
... Information about elements - the elements mercury, sulfur, and antimony were discovered - properties of some elements Develop lab apparatus / procedures / experimental techniques - alchemists learned how to prepare acids. - developed several alloys - new glassware ...
... Information about elements - the elements mercury, sulfur, and antimony were discovered - properties of some elements Develop lab apparatus / procedures / experimental techniques - alchemists learned how to prepare acids. - developed several alloys - new glassware ...
atoms of different elements differ in size, mass
... Atoms of different elements combine in simple whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, separated, or rearranged ...
... Atoms of different elements combine in simple whole-number ratios to form chemical compounds In chemical reactions, atoms are combined, separated, or rearranged ...
Isotope

Isotopes are variants of a particular chemical element which differ in neutron number, although all isotopes of a given element have the same number of protons in each atom. The term isotope is formed from the Greek roots isos (ἴσος ""equal"") and topos (τόπος ""place""), meaning ""the same place""; thus, the meaning behind the name it is that different isotopes of a single element occupy the same position on the periodic table. The number of protons within the atom's nucleus is called atomic number and is equal to the number of electrons in the neutral (non-ionized) atom. Each atomic number identifies a specific element, but not the isotope; an atom of a given element may have a wide range in its number of neutrons. The number of nucleons (both protons and neutrons) in the nucleus is the atom's mass number, and each isotope of a given element has a different mass number.For example, carbon-12, carbon-13 and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13 and 14 respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons, so that the neutron numbers of these isotopes are 6, 7 and 8 respectively.