The Structure of the Atom
... Radioactive decay produced particles about the mass of a _________but without any charge. Proved the particles produced were neutral because a charged object did not __________ ...
... Radioactive decay produced particles about the mass of a _________but without any charge. Proved the particles produced were neutral because a charged object did not __________ ...
ch10_sec1_rc
... predict the time required for half of the nuclei in a given radioactive sample to decay. • half-life: the time required for half of a sample of a radioactive isotope to break down by radioactive decay to form a daughter isotope ...
... predict the time required for half of the nuclei in a given radioactive sample to decay. • half-life: the time required for half of a sample of a radioactive isotope to break down by radioactive decay to form a daughter isotope ...
ppt
... The 235U nucleus captures a thermal (slow-moving) neutron This capture results in the formation of 236U*, and the excess energy of this nucleus causes it to undergo violent oscillations The 236U* nucleus becomes highly elongated, and the force of repulsion between the protons tends to increase ...
... The 235U nucleus captures a thermal (slow-moving) neutron This capture results in the formation of 236U*, and the excess energy of this nucleus causes it to undergo violent oscillations The 236U* nucleus becomes highly elongated, and the force of repulsion between the protons tends to increase ...
1 slide per page() - Wayne State University Physics and Astronomy
... The 235U nucleus captures a thermal (slow-moving) neutron This capture results in the formation of 236U*, and the excess energy of this nucleus causes it to undergo violent oscillations The 236U* nucleus becomes highly elongated, and the force of repulsion between the protons tends to increase ...
... The 235U nucleus captures a thermal (slow-moving) neutron This capture results in the formation of 236U*, and the excess energy of this nucleus causes it to undergo violent oscillations The 236U* nucleus becomes highly elongated, and the force of repulsion between the protons tends to increase ...
Chapter 3 - cloudfront.net
... Mass (not weight) of atoms is very, very small, i.e. around ____________ Better to use a relative scale to compare mass of atoms to each other, but you need a standard. Choose ______, which has 6 protons & 6 neutrons, 126C, also ...
... Mass (not weight) of atoms is very, very small, i.e. around ____________ Better to use a relative scale to compare mass of atoms to each other, but you need a standard. Choose ______, which has 6 protons & 6 neutrons, 126C, also ...
Radioactive decay of nucleus
... Isotopes – elements with same number of protons (Z) but differing in number of neutrons (N). Ex: H-1 , deuterium H-2, and tritium H-3 Have same chemical properties but different physical properties. one isotope of an element may be highly radioactive, while another is stable. Atomic Mass Units- ins ...
... Isotopes – elements with same number of protons (Z) but differing in number of neutrons (N). Ex: H-1 , deuterium H-2, and tritium H-3 Have same chemical properties but different physical properties. one isotope of an element may be highly radioactive, while another is stable. Atomic Mass Units- ins ...
Atomic Structure Subatomic Particles Atoms are made up of even
... Arrangement of subatomic particles ...
... Arrangement of subatomic particles ...
Chapter 4 and 5 study guide 2016-2017
... How is the atomic mass of an element determined? a. Count the number of protons and neutrons in an atom of the element. b. Use the atomic mass of the most abundant isotope. c. Take a weighted average of the masses of the isotopes present in nature. d. Average the atomic masses of all its isotopes. ...
... How is the atomic mass of an element determined? a. Count the number of protons and neutrons in an atom of the element. b. Use the atomic mass of the most abundant isotope. c. Take a weighted average of the masses of the isotopes present in nature. d. Average the atomic masses of all its isotopes. ...
Science Notes September 09/06/2016
... Dalton expanded into elements classification chart called periodic table with different masses & properties …Everything may be split into atoms (smallest particles) & compounds – combinations of elements JJ Thomson experimented with rays/beams of light to determine - electrons hold negative charge - ...
... Dalton expanded into elements classification chart called periodic table with different masses & properties …Everything may be split into atoms (smallest particles) & compounds – combinations of elements JJ Thomson experimented with rays/beams of light to determine - electrons hold negative charge - ...
CHAPTER 1 Practice Exercises 1.1 x = 12.3 g Cd 1.3 2.24845 ×12 u
... There is no space in the periodic table for another element of mass 73 u. Germanium has an atomic mass of 72.6 u and an atomic number of 32. Next to it on the periodic table is arsenic which has an atomic number of 33. In order for there to be a new element with an atomic mass of 73, it would be exp ...
... There is no space in the periodic table for another element of mass 73 u. Germanium has an atomic mass of 72.6 u and an atomic number of 32. Next to it on the periodic table is arsenic which has an atomic number of 33. In order for there to be a new element with an atomic mass of 73, it would be exp ...
Chapter 10 - Department Of Computer Science
... The atomic number (Z) is the number of protons in the nucleus of each atom of that element The atomic number also represents the number of electrons in a neutral atom ...
... The atomic number (Z) is the number of protons in the nucleus of each atom of that element The atomic number also represents the number of electrons in a neutral atom ...
Document
... statements is true? (a) The alpha particle has more kinetic energy than the daughter nucleus. (b) The daughter nucleus has more kinetic energy than the alpha particle. (c) The daughter nucleus and the alpha particle have the same kinetic energy. (a). Conservation of momentum requires the momenta of ...
... statements is true? (a) The alpha particle has more kinetic energy than the daughter nucleus. (b) The daughter nucleus has more kinetic energy than the alpha particle. (c) The daughter nucleus and the alpha particle have the same kinetic energy. (a). Conservation of momentum requires the momenta of ...
only that they did. democritus, an early greek philosopher, even had
... ELEMENTS, WE USE A SHORTHAND METHOD FOR DESIGNATING THE ELEMENTS. FOR EXAMPLE, THE SYMBOL FOR HYDROGEN IS ...
... ELEMENTS, WE USE A SHORTHAND METHOD FOR DESIGNATING THE ELEMENTS. FOR EXAMPLE, THE SYMBOL FOR HYDROGEN IS ...
s8.1toatomicmass
... 1. Discovered alpha (), beta (), and gamma () particles. 2. Correctly interpreted the nature of radioactivity. 3. Discovered the true nature of the alpha particles. 4. Discovered the atomic nucleus. - Most alpha particles shot through a thin gold foil, but a few alpha particles did not penetrate ...
... 1. Discovered alpha (), beta (), and gamma () particles. 2. Correctly interpreted the nature of radioactivity. 3. Discovered the true nature of the alpha particles. 4. Discovered the atomic nucleus. - Most alpha particles shot through a thin gold foil, but a few alpha particles did not penetrate ...
1_2133_201227212755_Unit_3(H)_TestA_2.7.12
... ____ 23. The atomic mass of an atom of carbon is 12, and the atomic mass of an atom of oxygen is 16. To produce CO, 16g of oxygen can be combined with 12g of carbon. According to the Law of Multiple Proportions, the ratio of oxygen to carbon when 32g of oxygen combine with 12g of carbon is a. 1:1 b. ...
... ____ 23. The atomic mass of an atom of carbon is 12, and the atomic mass of an atom of oxygen is 16. To produce CO, 16g of oxygen can be combined with 12g of carbon. According to the Law of Multiple Proportions, the ratio of oxygen to carbon when 32g of oxygen combine with 12g of carbon is a. 1:1 b. ...
Parts of the Atom & History of Discovery PPT
... Elements are made of tiny particles called atoms that are indivisible.. All atoms of a given element are identical. The atoms of a given element are different from those of any other element. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has ...
... Elements are made of tiny particles called atoms that are indivisible.. All atoms of a given element are identical. The atoms of a given element are different from those of any other element. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has ...
Name: Chapter 4 and 5 Study Guide Who was the Greek
... 16. What is going on inside the atoms when a neon light glows? 17. In a periodic table, a set of properties repeats from… a. Element to element b. Group to group c. Column to column d. Row to row 18. The usefulness of Mendeleev’s periodic table was confirmed by… a. The discovery of subatomic particl ...
... 16. What is going on inside the atoms when a neon light glows? 17. In a periodic table, a set of properties repeats from… a. Element to element b. Group to group c. Column to column d. Row to row 18. The usefulness of Mendeleev’s periodic table was confirmed by… a. The discovery of subatomic particl ...
Atomic Structure Notes
... identical. Atoms of any one element are different from those of any other element. ...
... identical. Atoms of any one element are different from those of any other element. ...
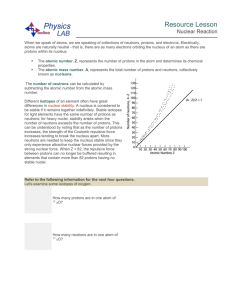
Resource Lesson Nuclear Reaction When we speak of atoms, we
... atoms are naturally neutral - that is, there are as many electrons orbiting the nucleus of an atom as there are protons within its nucleus. ...
... atoms are naturally neutral - that is, there are as many electrons orbiting the nucleus of an atom as there are protons within its nucleus. ...
THE ATOM: THE BASIC BUILDING BLOCK OF MATTER 12
... Note: Periodic Tables often display the information differently – watch out! Follow the key or legend. ...
... Note: Periodic Tables often display the information differently – watch out! Follow the key or legend. ...
Document
... 6. nucleus – The extremely dense central core of an atom. Almost the entire mass of the atom is concentrated in the nucleus, which occupies only a tiny fraction of the atom's volume. The nucleus of an atom consists of neutrons and protons. 7. element - Natural or synthetic (man-made) matter that is ...
... 6. nucleus – The extremely dense central core of an atom. Almost the entire mass of the atom is concentrated in the nucleus, which occupies only a tiny fraction of the atom's volume. The nucleus of an atom consists of neutrons and protons. 7. element - Natural or synthetic (man-made) matter that is ...
Ch.7 Summary Notes
... Radiocarbon dating is the process of determining the age of an object by measuring the amount of carbon-14 remaining in that object. Carbon’s isotopes include carbon-12 and carbon14. When an organism is alive, the ratio of carbon- 14 atoms to carbon-12 atoms in the organism remains nearly constant. ...
... Radiocarbon dating is the process of determining the age of an object by measuring the amount of carbon-14 remaining in that object. Carbon’s isotopes include carbon-12 and carbon14. When an organism is alive, the ratio of carbon- 14 atoms to carbon-12 atoms in the organism remains nearly constant. ...
View - Rutgers Physics
... formula might say, and those with an odd number a bit less well bound. This has a considerable effect on which nuclei are stable; If you look closely at the stable nuclei in Fig. 44.3, you see many more blue dots for even values of N and Z then for the odd values. Secondly, as you can imagine, the e ...
... formula might say, and those with an odd number a bit less well bound. This has a considerable effect on which nuclei are stable; If you look closely at the stable nuclei in Fig. 44.3, you see many more blue dots for even values of N and Z then for the odd values. Secondly, as you can imagine, the e ...