35. Number of reactions - Royal Society of Chemistry
... (Cu(OH)2.CuCO3) is formed initially. On adding excess A this dissolves to form a deep blue solution of Cu(NH3)42+(aq). ...
... (Cu(OH)2.CuCO3) is formed initially. On adding excess A this dissolves to form a deep blue solution of Cu(NH3)42+(aq). ...
Click here for the Reaction NOTES Handout
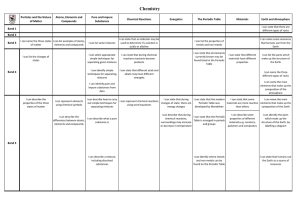
... a) Temperature 1. Decreasing heat will drive the reaction in the direction that will produce more heat (forward) 2. Increasing heat will drive the reaction in the direction that will use up the excess heat (reverse) b) Pressure 1. Decreasing pressure drives the reaction in the direction that will p ...
... a) Temperature 1. Decreasing heat will drive the reaction in the direction that will produce more heat (forward) 2. Increasing heat will drive the reaction in the direction that will use up the excess heat (reverse) b) Pressure 1. Decreasing pressure drives the reaction in the direction that will p ...
Preview Sample 1
... C) are generally 100 or more times sweeter than sucrose. D) are always some form of carbohydrate. E) are naturally similar to sugars. 102) Alaska Natives have a lower incidence of heart disease even though their diets are high in fat and cholesterol. This may be due to the large amount of ________ i ...
... C) are generally 100 or more times sweeter than sucrose. D) are always some form of carbohydrate. E) are naturally similar to sugars. 102) Alaska Natives have a lower incidence of heart disease even though their diets are high in fat and cholesterol. This may be due to the large amount of ________ i ...
CHEMISTRY The Central Science 9th Edition
... by symbols, using the initial letter of the name in capital form, starting by the old known elements, so Carbon is represented by the letter C, but Calcium is represented by the symbol Ca and Cobalt by the symbol Co, ……, Nitrogen is represented by the symbol N and Nickel by the symbol Ni, etc…. In g ...
... by symbols, using the initial letter of the name in capital form, starting by the old known elements, so Carbon is represented by the letter C, but Calcium is represented by the symbol Ca and Cobalt by the symbol Co, ……, Nitrogen is represented by the symbol N and Nickel by the symbol Ni, etc…. In g ...
ii. year course contents
... To teach the basic analytical chemistry concepts to the chemistry students and gain some skills with methods for the qualitative and quantitative analysis of samples. Chemistry students who will be work in research institution and industry, learn basic analytical chemistry concepts, errors in chemic ...
... To teach the basic analytical chemistry concepts to the chemistry students and gain some skills with methods for the qualitative and quantitative analysis of samples. Chemistry students who will be work in research institution and industry, learn basic analytical chemistry concepts, errors in chemic ...
Order date : 24-07-2010
... of different charge types due to Ingold Y-value and Dimroth parameter. Stereochemistry of SN1 and SN2 reactions. Reactions of epoxides and quaternary ammonium compounds. Neighbouring group participation- participation of carboxylate ion, halogen, hydroxyl group, acetoxy group, phenyl group and π bon ...
... of different charge types due to Ingold Y-value and Dimroth parameter. Stereochemistry of SN1 and SN2 reactions. Reactions of epoxides and quaternary ammonium compounds. Neighbouring group participation- participation of carboxylate ion, halogen, hydroxyl group, acetoxy group, phenyl group and π bon ...
Chem 150 Unit 2 - Hydrocarbons & Functional Groups
... The number of the first carbon in the double or triple bond is included in the name to locate the double or triple bond. ...
... The number of the first carbon in the double or triple bond is included in the name to locate the double or triple bond. ...
The format of this test is MULTIPLE CHOICE
... 2. All matter is made up of tiny particles called __atoms___. 3. When a solid becomes a liquid, _melting_____ occurs. 4. An _element_____ is made up of only one type of atom. 5. __freezing___ changes a liquid into a solid. 6. A mixture is made up of 2 or more substances that are physically combined ...
... 2. All matter is made up of tiny particles called __atoms___. 3. When a solid becomes a liquid, _melting_____ occurs. 4. An _element_____ is made up of only one type of atom. 5. __freezing___ changes a liquid into a solid. 6. A mixture is made up of 2 or more substances that are physically combined ...
The format of this test is MULTIPLE CHOICE
... 4. How does the size of an ion compare to the size of the neutral atom from which it was created? Ions are bigger 5. How does an atom’s position on the periodic table provide information on that atom’s size (atomic radius)? The farther left in the period, the larger the atom, the further down a grou ...
... 4. How does the size of an ion compare to the size of the neutral atom from which it was created? Ions are bigger 5. How does an atom’s position on the periodic table provide information on that atom’s size (atomic radius)? The farther left in the period, the larger the atom, the further down a grou ...
CH 115 Exam 2 - UAB General Chemistry Supplemental Instruction
... Assume the chemical equations on this exam are NOT balanced unless stated otherwise. 1. Balance the equation and give the stoichiometric coefficient for HCl ...
... Assume the chemical equations on this exam are NOT balanced unless stated otherwise. 1. Balance the equation and give the stoichiometric coefficient for HCl ...
Atoms, Molecules, and Ions
... understanding of math and algebra, including an understanding of decimals, exponents, logarithms, quadratics, and algebraic equations, is essential to success in this course (calculus is not required). You should not be taking remedial algebra concurrently with this course. Topics included are atomi ...
... understanding of math and algebra, including an understanding of decimals, exponents, logarithms, quadratics, and algebraic equations, is essential to success in this course (calculus is not required). You should not be taking remedial algebra concurrently with this course. Topics included are atomi ...
Molecular Evolution
... events that lead to the early building blocks of life. After one week, nearly 15% of carbon was in organic compounds. DNA and RNA were not formed but sugars, lipids and some building blocks for nucleic acids were formed. 2% of carbon formed amino acids! ...
... events that lead to the early building blocks of life. After one week, nearly 15% of carbon was in organic compounds. DNA and RNA were not formed but sugars, lipids and some building blocks for nucleic acids were formed. 2% of carbon formed amino acids! ...
Problem Set 3_Chem165_Sp2014
... For (c), which isomer of the alcohol product do you think you get from the hydration reaction, or do you think you get both isomers? 7. Predict the products of the following reactions. (No reaction could be the correct answer.) (a) 1-octene + MeOH (with an acid catalyst) (b) styrene + excess H2 (ove ...
... For (c), which isomer of the alcohol product do you think you get from the hydration reaction, or do you think you get both isomers? 7. Predict the products of the following reactions. (No reaction could be the correct answer.) (a) 1-octene + MeOH (with an acid catalyst) (b) styrene + excess H2 (ove ...
Ch 4 Review
... d. subatomic particle without charge e. positively charged subatomic particle ____ 30. chemical properties ____ 31. neutron ____ 32. proton ____ 33. physical properties ____ 34. electron Match each item with the correct statement below. a. the smallest unit of an element that maintains all of the el ...
... d. subatomic particle without charge e. positively charged subatomic particle ____ 30. chemical properties ____ 31. neutron ____ 32. proton ____ 33. physical properties ____ 34. electron Match each item with the correct statement below. a. the smallest unit of an element that maintains all of the el ...
Chapter 2
... Also called exchange reactions because electrons are exchanged or shared differently Electron donors lose electrons and are oxidized Electron acceptors receive electrons and become ...
... Also called exchange reactions because electrons are exchanged or shared differently Electron donors lose electrons and are oxidized Electron acceptors receive electrons and become ...
Syllabus of the International Chemistry Olympiad
... 1.3 Trends in physical properties (main groups) ...
... 1.3 Trends in physical properties (main groups) ...
Derivatization reagents
... ● Purified, dried and packaged under nitrogen in convenient 50mL Hypo-Vial Sample Storage Vials ● Supplied with elastomer septa, allowing immediate access to the sample without exposure to moisture and oxygen ● Use polar solvents (acetonitrile, dimethylformamide, dimethylsulfoxide, pyridine, tetrahy ...
... ● Purified, dried and packaged under nitrogen in convenient 50mL Hypo-Vial Sample Storage Vials ● Supplied with elastomer septa, allowing immediate access to the sample without exposure to moisture and oxygen ● Use polar solvents (acetonitrile, dimethylformamide, dimethylsulfoxide, pyridine, tetrahy ...
Re-typed from The Ultimate Chemical Equations Handbook by
... 1. A binary molecule is formed when two nonmetals or metalloids combine. Electrons are shared so the bonding involved is known as _________________________ bonding. 2. Sometimes these compounds have generic or common names (water) and they also have systemic names (dihydrogen monoxide). The common n ...
... 1. A binary molecule is formed when two nonmetals or metalloids combine. Electrons are shared so the bonding involved is known as _________________________ bonding. 2. Sometimes these compounds have generic or common names (water) and they also have systemic names (dihydrogen monoxide). The common n ...
Basic Background Review: Acid-Base , Redox, and Stable Isotopes
... •pH of a neutral solution of water = 7 •Acid‐base balances within Earth system generally involve elements of relatively high abundance ...
... •pH of a neutral solution of water = 7 •Acid‐base balances within Earth system generally involve elements of relatively high abundance ...
Unit 8 Powerpoint
... 4. Balance the elements one at a time by using coefficients. Begin by balancing elements that appear only once on each side of the equation. Unwritten coefficients are assumed to be 1 Once you are certain you have the correct chemical ...
... 4. Balance the elements one at a time by using coefficients. Begin by balancing elements that appear only once on each side of the equation. Unwritten coefficients are assumed to be 1 Once you are certain you have the correct chemical ...
Course 2.2. Organic matter
... It is highly recommendable to use the “Atoms system” (e.g. to make mass balances) Be very aware, in water quality data interpretation as well as in your own data reporting, of the way the results are expressed ! It’s a big source of errors! ...
... It is highly recommendable to use the “Atoms system” (e.g. to make mass balances) Be very aware, in water quality data interpretation as well as in your own data reporting, of the way the results are expressed ! It’s a big source of errors! ...
08 PowerPoint
... Chemical equations represent, with symbols and formulas, the reactants and products in a chemical reaction. ...
... Chemical equations represent, with symbols and formulas, the reactants and products in a chemical reaction. ...
Variation in Properties of Group II Compounds
... Each group of elements embodied in the periodic table has their own unique properties. As for group II elements, they are classified as one of the s-block elements, also named as alkaline earth metals. In this essay, the variation in properties of group II elements and their compounds are illustrate ...
... Each group of elements embodied in the periodic table has their own unique properties. As for group II elements, they are classified as one of the s-block elements, also named as alkaline earth metals. In this essay, the variation in properties of group II elements and their compounds are illustrate ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.