Ch. 7 & 8 Notes (Chemical Reactions) teacher
... compounds A reaction between _____ that are dissolved in water that produces _____ two ________________ compounds , one of which is ...
... compounds A reaction between _____ that are dissolved in water that produces _____ two ________________ compounds , one of which is ...
Chemical Equations
... Chemical formula A representation of a substance in which the elements are represented by their symbols and subscripts represent the number of atoms of each element ...
... Chemical formula A representation of a substance in which the elements are represented by their symbols and subscripts represent the number of atoms of each element ...
+ H 2 SO 4(aq) - Rothschild Science
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
... element you have NH3 (one nitrogen, three hydrogen)- DON’T mess with these!! Coefficients – small whole number that appears ...
Organic and Bio-Molecular Chemistry
... indicated by the name, the wide variety of compounds that constitute the living organisms; it includes however also structurally related compounds that have been mass-produced in laboratories and by industry in the 20th century. ...
... indicated by the name, the wide variety of compounds that constitute the living organisms; it includes however also structurally related compounds that have been mass-produced in laboratories and by industry in the 20th century. ...
Chemistry
... Properties of Matter • Property- a characteristic or feature that makes it possible to distinguish between kinds of matter • Physical Property- a characteristic that can be observed without altering the matter • Mass, color, shape, length ...
... Properties of Matter • Property- a characteristic or feature that makes it possible to distinguish between kinds of matter • Physical Property- a characteristic that can be observed without altering the matter • Mass, color, shape, length ...
File
... monatomic- meaning they naturally occur as stable, single atoms. Right: Many gases, such as Hydrogen, are diatomic, which means they naturally occur as a molecule with two atoms. Not shown: Some gasses, such as Ozone- a form of oxygen, occur in a three atom arrangement called triatomic. ...
... monatomic- meaning they naturally occur as stable, single atoms. Right: Many gases, such as Hydrogen, are diatomic, which means they naturally occur as a molecule with two atoms. Not shown: Some gasses, such as Ozone- a form of oxygen, occur in a three atom arrangement called triatomic. ...
UNIT 7 – CHEMICAL REACTIONS
... 5. Sometimes a metal will not always replace another metal in a compound dissolved in water. 6. This is because metals differ in their reactivities. A metal’s reactivity is its ability to react with another substance. 7. Chemists use an _________________________ to be able to know which metals will ...
... 5. Sometimes a metal will not always replace another metal in a compound dissolved in water. 6. This is because metals differ in their reactivities. A metal’s reactivity is its ability to react with another substance. 7. Chemists use an _________________________ to be able to know which metals will ...
Sample Exam 1 Key
... 10. Aspirin has the formula C9H8O4. A compound is isolated from sea urchins that also has the formula C9H8O4. What can you conclude? a) Sea urchins produce aspirin. b) Sea urchins might produce aspirin, but this isn’t sufficient evidence that they do. c) Because aspirin is a fairly small molecule, i ...
... 10. Aspirin has the formula C9H8O4. A compound is isolated from sea urchins that also has the formula C9H8O4. What can you conclude? a) Sea urchins produce aspirin. b) Sea urchins might produce aspirin, but this isn’t sufficient evidence that they do. c) Because aspirin is a fairly small molecule, i ...
Scientific Method - Virtual Medical Academy
... 2/ An atom of zinc has a mass number of 65. (A): Number of protons in the zinc atom >> ...
... 2/ An atom of zinc has a mass number of 65. (A): Number of protons in the zinc atom >> ...
Scientific Method - Virtual Medical Academy
... 2/ An atom of zinc has a mass number of 65. (A): Number of protons in the zinc atom >> ...
... 2/ An atom of zinc has a mass number of 65. (A): Number of protons in the zinc atom >> ...
1. Natures Chemistry Unit Questions
... In the reaction, the carbon atom next to the carbonyl functional group of one molecule forms a bond with the carbonyl carbon atom of the second molecule. (a) Draw a structural formula for the product formed when propanone is used instead of ethanal in this type of reaction. (1) (b) Name an aldehyde ...
... In the reaction, the carbon atom next to the carbonyl functional group of one molecule forms a bond with the carbonyl carbon atom of the second molecule. (a) Draw a structural formula for the product formed when propanone is used instead of ethanal in this type of reaction. (1) (b) Name an aldehyde ...
Export To Word
... B. Electrons are key to defining chemical and some physical properties, reactivity, and molecular structures. Repeating (periodic) patterns of physical and chemical properties occur among elements that define groups of elements with similar properties. The periodic table displays the repeating patte ...
... B. Electrons are key to defining chemical and some physical properties, reactivity, and molecular structures. Repeating (periodic) patterns of physical and chemical properties occur among elements that define groups of elements with similar properties. The periodic table displays the repeating patte ...
Document
... 1. Color the carbon atoms black, the oxygen atoms red, and leave the hydrogen atoms white. 2. Use scissors to carefully cut out the atoms. o Build the reactants: 3. On a sheet of paper, place the atoms together to make the molecules of the reactants on the left side of the chemical equation for the ...
... 1. Color the carbon atoms black, the oxygen atoms red, and leave the hydrogen atoms white. 2. Use scissors to carefully cut out the atoms. o Build the reactants: 3. On a sheet of paper, place the atoms together to make the molecules of the reactants on the left side of the chemical equation for the ...
CHEMISTRY
... Compounds contain more than one element. They always have the same composition, regardless of source (law of constant composition; law of definite ...
... Compounds contain more than one element. They always have the same composition, regardless of source (law of constant composition; law of definite ...
Semester 2 Review WS
... d. What is represented in letter C? _________________________________________________ e. How many total ATOMS are there in letter D? _____________________________________ f. How many total MOLECULES are there in letter D? ________________________________ 2. Distinguish between the structure of molec ...
... d. What is represented in letter C? _________________________________________________ e. How many total ATOMS are there in letter D? _____________________________________ f. How many total MOLECULES are there in letter D? ________________________________ 2. Distinguish between the structure of molec ...
Chemistry - talcher autonomous college
... X-ray diffraction, Bragg’s law, a simple account of rotating crystal method and powder pattern method. Analysis of powder diffraction patterns of NaCl, CsCl and KCl. Defects in crystals. Glasses and liquid crystals. (16 Lectures) ...
... X-ray diffraction, Bragg’s law, a simple account of rotating crystal method and powder pattern method. Analysis of powder diffraction patterns of NaCl, CsCl and KCl. Defects in crystals. Glasses and liquid crystals. (16 Lectures) ...
Summary of 5.4
... Evidence for the structure of benzene comes from x-ray diffraction. A pattern like the one above can be processed to indicate areas of high electron density in a molecule. See below for p-methoxybenzoic acid, which contains a benzene ring. ...
... Evidence for the structure of benzene comes from x-ray diffraction. A pattern like the one above can be processed to indicate areas of high electron density in a molecule. See below for p-methoxybenzoic acid, which contains a benzene ring. ...
Topic 2
... Atomic Theory of Matter – An element is a substance whose atoms all have the same atomic number (Z). The #protons defines the identity of an atom and can be found on the periodic table (large number in top of element box). – The neutron is a nuclear particle having a mass almost identical to that o ...
... Atomic Theory of Matter – An element is a substance whose atoms all have the same atomic number (Z). The #protons defines the identity of an atom and can be found on the periodic table (large number in top of element box). – The neutron is a nuclear particle having a mass almost identical to that o ...
31 BIOMOLECULES Y MODULE - 7
... of large number of amino acid units. There is no clear line of demarcation between polypeptides and proteins. For example insulin, although it contains only 51 amino acids, is generally considered a small protein. The amino acid unit with the free amino group is known as the N-terminal residue and t ...
... of large number of amino acid units. There is no clear line of demarcation between polypeptides and proteins. For example insulin, although it contains only 51 amino acids, is generally considered a small protein. The amino acid unit with the free amino group is known as the N-terminal residue and t ...
Topic 20 Organic Chemistry
... 2. Compound A is a bromoalkane. When compound A reacts with warm dilute sodium hydroxide solution an organic compound B is formed which has a relative molar mass of 74.12 and contains 64.81 % carbon, 13.60 % hydrogen and 21.59 % oxygen by mass. Compound B can be oxidised by an acidified solution of ...
... 2. Compound A is a bromoalkane. When compound A reacts with warm dilute sodium hydroxide solution an organic compound B is formed which has a relative molar mass of 74.12 and contains 64.81 % carbon, 13.60 % hydrogen and 21.59 % oxygen by mass. Compound B can be oxidised by an acidified solution of ...
What Can I Do With a Major In Chemistry
... and use of matter. Chemistry is divided into five main areas: analytical chemistry, biochemistry, inorganic chemistry, organic chemistry and physical chemistry. Analytical chemistry is the study of the physical and chemical properties of compounds and mixtures through qualitative and quantitative an ...
... and use of matter. Chemistry is divided into five main areas: analytical chemistry, biochemistry, inorganic chemistry, organic chemistry and physical chemistry. Analytical chemistry is the study of the physical and chemical properties of compounds and mixtures through qualitative and quantitative an ...
Chemistry Subject Matter Requirements Part I: Content Domains for
... (Next Generation Science Standards for California Public Schools, Kindergarten through Grade Twelve, Grades Nine through Twelve, Physical Sciences: PS1.B) Understand organic chemistry and biochemistry. a. Demonstrate knowledge of the bonding characteristics of carbon. b. Recognize the chemical struc ...
... (Next Generation Science Standards for California Public Schools, Kindergarten through Grade Twelve, Grades Nine through Twelve, Physical Sciences: PS1.B) Understand organic chemistry and biochemistry. a. Demonstrate knowledge of the bonding characteristics of carbon. b. Recognize the chemical struc ...
CHEMISTRY A
... (d) Chemical companies are using catalysts to develop processes that are more sustainable. These processes reduce costs and are less harmful to the environment. Suggest two ways in which the use of catalysts helps chemical companies to make their processes more sustainable. ...
... (d) Chemical companies are using catalysts to develop processes that are more sustainable. These processes reduce costs and are less harmful to the environment. Suggest two ways in which the use of catalysts helps chemical companies to make their processes more sustainable. ...
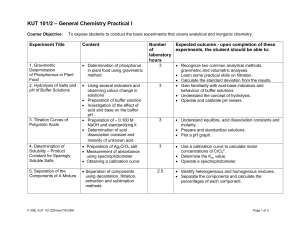
KUT 101/2 – General Chemistry Practical I
... • Determination of acid dissociation constant and molarity of unknown acid • Preparation of Ag2CrO4 salt • Measurement of absorbance using spectrophotometer • Obtaining a calibration curve ...
... • Determination of acid dissociation constant and molarity of unknown acid • Preparation of Ag2CrO4 salt • Measurement of absorbance using spectrophotometer • Obtaining a calibration curve ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.