Oxidation-Reduction Processes in Natural Waters
... the absence of organisms because of slow kinetics. (An exception is the oxidation of Fe+2 to Fe+3, which will occur spontaneously by chemical means alone.) Because of the combination of slow chemical kinetics and favorable thermodynamic conditions, opportunistic microbes catalyze all the important r ...
... the absence of organisms because of slow kinetics. (An exception is the oxidation of Fe+2 to Fe+3, which will occur spontaneously by chemical means alone.) Because of the combination of slow chemical kinetics and favorable thermodynamic conditions, opportunistic microbes catalyze all the important r ...
CHEM IB Lecture notes as of 8-29-06
... 1 : a science that deals with the composition, structure, and properties of substances and with the transformations that they undergo 2 a : the composition and chemical properties of a substance b : chemical
processes and phenomena (as of an organism)
3 a : ...
... 1 : a science that deals with the composition, structure, and properties of substances and with the transformations that they undergo 2 a : the composition and chemical properties of a substance
double-replacement reaction
... Predicting whether a reaction will occur In a single-replacement reaction, a more active metal displaces a less active metal according to the activity series. • In a double-replacement reaction, two aqueous solutions produce a precipitate of an insoluble compound. ...
... Predicting whether a reaction will occur In a single-replacement reaction, a more active metal displaces a less active metal according to the activity series. • In a double-replacement reaction, two aqueous solutions produce a precipitate of an insoluble compound. ...
State Standard - SchoolNotes.com
... Understand: Chemical elements are the fundamental building materials of matter. Elemental properties are determined by the structure of the nucleus and distribution of electrons. One element can change into another through one of several nuclear processes. ...
... Understand: Chemical elements are the fundamental building materials of matter. Elemental properties are determined by the structure of the nucleus and distribution of electrons. One element can change into another through one of several nuclear processes. ...
Chem 115 POGIL Worksheet
... elements are homonuclear, because they are composed of only one kind of atom. Molecules of compounds are heteronuclear, because they are composed of two or more different kinds of atoms. For a molecular substance (element or compound), the composition of the molecules is indicated by a molecular for ...
... elements are homonuclear, because they are composed of only one kind of atom. Molecules of compounds are heteronuclear, because they are composed of two or more different kinds of atoms. For a molecular substance (element or compound), the composition of the molecules is indicated by a molecular for ...
Chemical Reactions
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
... (BrINClHOF) For example, Oxygen is O2 as an element. In a compound, it can’t be a diatomic element because it’s not an element anymore, it’s a compound! ...
Unit 3 Practice Test
... 25. CH4, the first member of Group IVA hydrogen compounds, does not show the reversal in trend because CH4 is A. nonpolar and has only van der Waals forces C. nonpolar and has only London dispersion force ...
... 25. CH4, the first member of Group IVA hydrogen compounds, does not show the reversal in trend because CH4 is A. nonpolar and has only van der Waals forces C. nonpolar and has only London dispersion force ...
1st Olympiad of Metropolises Chemistry Theoretical Problems
... under heating of furan with ammonia (amines) above 400 C in the presence of alumina. In a laboratory, the sequence of furan hydrolysis followed by Paal-Knorr reaction with ammonia (amine) is used for this transformation. This sequence can be realized as a two-step procedure or as a domino reaction. ...
... under heating of furan with ammonia (amines) above 400 C in the presence of alumina. In a laboratory, the sequence of furan hydrolysis followed by Paal-Knorr reaction with ammonia (amine) is used for this transformation. This sequence can be realized as a two-step procedure or as a domino reaction. ...
Lecture 24 (Slides) October 18
... Ionic and Covalent Bonds • When Main Group elements react, electrons can be transferred (usually from a metal to a nonmetal) to form ionic bonds. In other cases, pairs of electrons can be shared (usually between nonmetal atoms) to form covalent bonds. In both cases valence electrons are somehow “re ...
... Ionic and Covalent Bonds • When Main Group elements react, electrons can be transferred (usually from a metal to a nonmetal) to form ionic bonds. In other cases, pairs of electrons can be shared (usually between nonmetal atoms) to form covalent bonds. In both cases valence electrons are somehow “re ...
A Study of Matter
... liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
... liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
Chem Bonding Notes
... (4)CH2O 23. Which formula represents a nonpolar molecule? (1)HC1 (3)NH3 (2)H 2 O (4)CF4 24. Which type of bond is formed when electrons are transferred from one atom to another? (1) covalent (3) hydrogen (2) ionic (4) metallic 25. Which Lewis electron-dot structure is drawn correctly for the atom it ...
... (4)CH2O 23. Which formula represents a nonpolar molecule? (1)HC1 (3)NH3 (2)H 2 O (4)CF4 24. Which type of bond is formed when electrons are transferred from one atom to another? (1) covalent (3) hydrogen (2) ionic (4) metallic 25. Which Lewis electron-dot structure is drawn correctly for the atom it ...
2.6 M - Thierry Karsenti
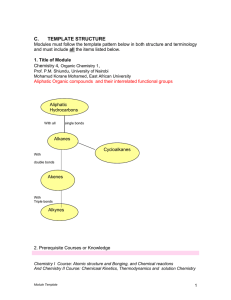
... .3. An alkane is a hydrocarbon that has only single bonds. Alkanes that do not contain rings have he formula CnH2n+2. An alkane in the shape of a ring is called a cycloalkane. Cycloalkanes have the formula CnH2n. ..4. An alkene is a compound that has at least one double bond. A straight-chain alkene ...
... .3. An alkane is a hydrocarbon that has only single bonds. Alkanes that do not contain rings have he formula CnH2n+2. An alkane in the shape of a ring is called a cycloalkane. Cycloalkanes have the formula CnH2n. ..4. An alkene is a compound that has at least one double bond. A straight-chain alkene ...
Importance of Molecular Simulation for Studying Structural Properties
... Naturally occurring cationic polysaccharide known for its biocompatibility and biodegradability is chitosan (QN). The QN is a copolymer of the amino polysaccharide which is obtained by alkaline deacetylation of chitin [11], the main structural material in the exoskeletons of crustaceans and other an ...
... Naturally occurring cationic polysaccharide known for its biocompatibility and biodegradability is chitosan (QN). The QN is a copolymer of the amino polysaccharide which is obtained by alkaline deacetylation of chitin [11], the main structural material in the exoskeletons of crustaceans and other an ...
Matter
... E) A cake bakes in the oven How do we know if a chemical change has taken place? Evidence of chemical change: ...
... E) A cake bakes in the oven How do we know if a chemical change has taken place? Evidence of chemical change: ...
ppt Sc10 Review Notes
... for the first part of the compound, then name the “xH2O” part as prefix + “hydrate” eg) NaF3H2O = sodium fluoride trihydrate CuSO45H2O = copper (II) sulphate pentahydrate ...
... for the first part of the compound, then name the “xH2O” part as prefix + “hydrate” eg) NaF3H2O = sodium fluoride trihydrate CuSO45H2O = copper (II) sulphate pentahydrate ...
Chapter 7 Chemical Reactions
... There are millions of compounds that will produce endless chemical reactions, therefore not all chemical reactions can be carried out in the laboratory A system is used to classify chemical reactions, which allows chemist to recognize patterns and predict the products of reactions One of these ...
... There are millions of compounds that will produce endless chemical reactions, therefore not all chemical reactions can be carried out in the laboratory A system is used to classify chemical reactions, which allows chemist to recognize patterns and predict the products of reactions One of these ...
*6th Grade Science-Chapter 5 Study Guide Lesson 5.1: Observing
... *Changes in matter can be described in terms of physical changes and chemical changes. *Chemical reactions involve changes in properties and changes in energy that you can often observe. Physical change-any change that alters the form or appearance of a substance but does not change it into another ...
... *Changes in matter can be described in terms of physical changes and chemical changes. *Chemical reactions involve changes in properties and changes in energy that you can often observe. Physical change-any change that alters the form or appearance of a substance but does not change it into another ...
CHAPTER 2
... • If only 2 similar formula type anions exist, the one containing the greater number of oxygen atoms have an “–ate” ending, and the smaller number of oxygen atoms have an “-ite” ending NO3NO2SO42SO32• If more than two exist, the one with the largest number of oxygen atoms have a prefix “per-” and an ...
... • If only 2 similar formula type anions exist, the one containing the greater number of oxygen atoms have an “–ate” ending, and the smaller number of oxygen atoms have an “-ite” ending NO3NO2SO42SO32• If more than two exist, the one with the largest number of oxygen atoms have a prefix “per-” and an ...
2 - My CCSD
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
File
... 21. The columns in the periodic table are called ______________________. They are grouped by ____________________________________________________________. 22. Most of the elements on the periodic table are __________________. 23. Matter is anything that takes up ____________ and has ____________. Al ...
... 21. The columns in the periodic table are called ______________________. They are grouped by ____________________________________________________________. 22. Most of the elements on the periodic table are __________________. 23. Matter is anything that takes up ____________ and has ____________. Al ...
Chemical Reactions PPT
... A boy and a girl are dancing, but then another boy “cuts in” and dances with the girl, leaving the first boy alone. Or a boy and a girl are dancing, but then another girl “cuts in” and dances with the boy, leaving the first girl alone. “Like” must replace “like” ...
... A boy and a girl are dancing, but then another boy “cuts in” and dances with the girl, leaving the first boy alone. Or a boy and a girl are dancing, but then another girl “cuts in” and dances with the boy, leaving the first girl alone. “Like” must replace “like” ...
Syracuse Syllabus
... understanding of math and algebra, including an understanding of decimals, exponents, logarithms, quadratics, and algebraic equations, is essential to success in this course (calculus is not required). You should not be taking remedial algebra concurrently with this course. Topics included are atomi ...
... understanding of math and algebra, including an understanding of decimals, exponents, logarithms, quadratics, and algebraic equations, is essential to success in this course (calculus is not required). You should not be taking remedial algebra concurrently with this course. Topics included are atomi ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... Herbicidal, Insecticidal, Antihistaminic, Anti-inflammatory and Anticancer. CNS stimulant and recent reports suggest that compounds containing piperidin-4-one moiety elicit excellent activity when aromatic substitutions are present at 2- and/or 6-positions. Mannich reaction is one of the multi-compo ...
... Herbicidal, Insecticidal, Antihistaminic, Anti-inflammatory and Anticancer. CNS stimulant and recent reports suggest that compounds containing piperidin-4-one moiety elicit excellent activity when aromatic substitutions are present at 2- and/or 6-positions. Mannich reaction is one of the multi-compo ...
What are the general types of reactions?
... – Mass is not created or destroyed in a chemical reaction – For practical purposes • Same types of atoms before and after a reaction • Same number of each type of atom before and after ...
... – Mass is not created or destroyed in a chemical reaction – For practical purposes • Same types of atoms before and after a reaction • Same number of each type of atom before and after ...
Study Island Copyright © 2012 Study Island
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
... 15. Which of the following is true about a compound and its elements? A. The properties of a compound are the same as the properties of its elements. B. The elements all share identical properties, but their properties are different than the compound's properties. C. The properties of a compound are ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.