200things2know
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
Chem 1411 Chapter 4
... A strong electrolyte is the one that has a high degree of dissociation and a weak electrolyte is the one that has a low degree of dissociation. Ex. NaCl, HCl, MgBr2 (Strong Electrolytes), Ca(OH)2, NH4OH(Weak Electrolytes) Acids and bases are also electrolytes. Non-electrolyte A substance that does n ...
... A strong electrolyte is the one that has a high degree of dissociation and a weak electrolyte is the one that has a low degree of dissociation. Ex. NaCl, HCl, MgBr2 (Strong Electrolytes), Ca(OH)2, NH4OH(Weak Electrolytes) Acids and bases are also electrolytes. Non-electrolyte A substance that does n ...
PPT format - Columbia University
... Avogadro’s Law: Equal volumes of different gases contain the same number of particles. The particles of a gas may be atoms or molecules. One liter of hydrogen = one liter of chlorine = one liter of hydrogen chloride in terms of particles (read molecules) ...
... Avogadro’s Law: Equal volumes of different gases contain the same number of particles. The particles of a gas may be atoms or molecules. One liter of hydrogen = one liter of chlorine = one liter of hydrogen chloride in terms of particles (read molecules) ...
Unit B Chemistry Unit study guide
... What is reactivity? Names, placement and characteristics of Families- Alkali metals, alkali earth metals, transition metals, halogens, noble gases as well as metals vs nonmetals Why are lanthanides and actinides on bottom? What are the only two liquids? Where are the gasses? Which element is in a gr ...
... What is reactivity? Names, placement and characteristics of Families- Alkali metals, alkali earth metals, transition metals, halogens, noble gases as well as metals vs nonmetals Why are lanthanides and actinides on bottom? What are the only two liquids? Where are the gasses? Which element is in a gr ...
Slide 1
... Most of these reactions have slow kinetics if not mediated by bacteria. Bacteria mediate most of these reactions and get the energy for their life processes. Because the energy of the sun is trapped in the C-C bonds, bacteria are indirectly using sunlight when they combust natural organic matter to ...
... Most of these reactions have slow kinetics if not mediated by bacteria. Bacteria mediate most of these reactions and get the energy for their life processes. Because the energy of the sun is trapped in the C-C bonds, bacteria are indirectly using sunlight when they combust natural organic matter to ...
Grades 9-12 Chemistry California Content Standards
... varied sizes, shapes, and chemical properties, providing the biochemical basis of life. As a basis for understanding this concept, students know: a. large molecules (polymers) such as proteins, nucleic acids, and starch are formed by repetitive combinations of simple sub-units. b. the bonding charac ...
... varied sizes, shapes, and chemical properties, providing the biochemical basis of life. As a basis for understanding this concept, students know: a. large molecules (polymers) such as proteins, nucleic acids, and starch are formed by repetitive combinations of simple sub-units. b. the bonding charac ...
Unit 3.2 worksheet 4 atomic model of matter
... Tips and tricks! Hope I help :)) Video Rating: / 5. Click Here - Movie Star Planet. Hi i am writing u to ask what is the state requirments while growing for person medical needs. what will make it completely legal where if visited by the law i wanna. Getting Started. USATestprep is very user-friendl ...
... Tips and tricks! Hope I help :)) Video Rating: / 5. Click Here - Movie Star Planet. Hi i am writing u to ask what is the state requirments while growing for person medical needs. what will make it completely legal where if visited by the law i wanna. Getting Started. USATestprep is very user-friendl ...
Chemistry - Gorman Learning Center
... varied sizes, shapes, and chemical properties, providing the biochemical basis of life. As a basis for understanding this concept, students know: a. large molecules (polymers) such as proteins, nucleic acids, and starch are formed by repetitive combinations of simple sub-units. b. the bonding charac ...
... varied sizes, shapes, and chemical properties, providing the biochemical basis of life. As a basis for understanding this concept, students know: a. large molecules (polymers) such as proteins, nucleic acids, and starch are formed by repetitive combinations of simple sub-units. b. the bonding charac ...
Unit 2.2 Test Review Key
... Are they, H2O2 and 2OH, the same substance? __No___ Explain your answer: No they are not the same substance even though they have the same amount and type of atoms. H2O2 is one molecule of Hydrogen Peroxide that has all of its atoms bonded together. Whereas, 2OH is two molecules of OH bonded togethe ...
... Are they, H2O2 and 2OH, the same substance? __No___ Explain your answer: No they are not the same substance even though they have the same amount and type of atoms. H2O2 is one molecule of Hydrogen Peroxide that has all of its atoms bonded together. Whereas, 2OH is two molecules of OH bonded togethe ...
Writing and Classifying Balanced Equations
... Instructions: Carefully study these five types of chemical reactions and answer the questions. **A, B, and C represent pure substances. AB, CD, AC, BC, AD, and CB are all compounds. **CxHyOz represents a fuel or any compound made up of carbon, hydrogen, and/or oxygen ...
... Instructions: Carefully study these five types of chemical reactions and answer the questions. **A, B, and C represent pure substances. AB, CD, AC, BC, AD, and CB are all compounds. **CxHyOz represents a fuel or any compound made up of carbon, hydrogen, and/or oxygen ...
2202 Chapter 1 - Eric G. Lambert School
... eg. calcium hydroxide ammonium sulfate PbSO4 Sn3(PO4)4 8:12 PM ...
... eg. calcium hydroxide ammonium sulfate PbSO4 Sn3(PO4)4 8:12 PM ...
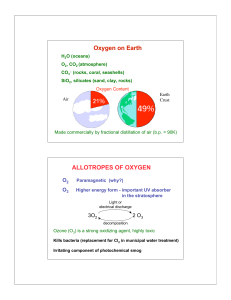
p-Block Elements, Part 1
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
- Department of Chemistry, York University
... CM Chondrites: •asteroidal origin (material from solar system) Amino acid composition in two CI (Ivuna and Orgeuil) and two CM (Murchison and Murray) meteorites: Amino acid Glycine -amino acids ...
... CM Chondrites: •asteroidal origin (material from solar system) Amino acid composition in two CI (Ivuna and Orgeuil) and two CM (Murchison and Murray) meteorites: Amino acid Glycine -amino acids ...
Chapter 1 - Manual Science Chemistry/Physics
... Molecule – smallest stable unit of a substance; can be composed of one or more kinds of atoms. Element – a pure substance that cannot be broken down into simpler, stable substances, and is only made up of one type of atom. Compound – a substance that can be broken down into simpler stable subs ...
... Molecule – smallest stable unit of a substance; can be composed of one or more kinds of atoms. Element – a pure substance that cannot be broken down into simpler, stable substances, and is only made up of one type of atom. Compound – a substance that can be broken down into simpler stable subs ...
urbano, mariajose
... A. Functional groups also contribute to the molecular diversity of life Small characteristic groups of atoms (functional groups) are frequently bonded to the carbon skeleton of organic molecules. These functional groups: • Have specific chemical and physical properties. • Are the regions of organic ...
... A. Functional groups also contribute to the molecular diversity of life Small characteristic groups of atoms (functional groups) are frequently bonded to the carbon skeleton of organic molecules. These functional groups: • Have specific chemical and physical properties. • Are the regions of organic ...
Chapter 3 – part I Sections 1-3
... ions) are these. You will learn later that strong acids and bases are strong electrolytes. • Weak electrolytes are weak conductors, ionic compounds that are insoluble are these. Insoluble = only a few dissolve into ions. You will learn later that weak acids and bases are weak electrolytes. • Non-ele ...
... ions) are these. You will learn later that strong acids and bases are strong electrolytes. • Weak electrolytes are weak conductors, ionic compounds that are insoluble are these. Insoluble = only a few dissolve into ions. You will learn later that weak acids and bases are weak electrolytes. • Non-ele ...
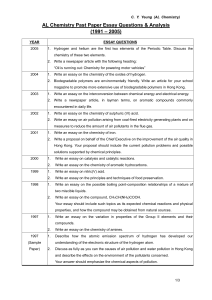
AL Chemistry Past paper essay questions
... Write an essay on amino acids, polypeptides and proteins. Your essay should include the properties of amino acids in aqueous solutions and a method of separation for a mixture of amino acids, as well as the constitution of polypeptides and proteins and their hydrolysis. ...
... Write an essay on amino acids, polypeptides and proteins. Your essay should include the properties of amino acids in aqueous solutions and a method of separation for a mixture of amino acids, as well as the constitution of polypeptides and proteins and their hydrolysis. ...
Biochemistry Assessment
... _______12. The four major macromolecules are _______________. A Proteins, carbohydrates, amino acids, steroids B Amino acids, lipids, carbohydrates, fats C Lipids, proteins, nucleic acids, carbohydrates D Steroids, nucleic acids, amino acids, lipids _______13. A common name for lipids is ___________ ...
... _______12. The four major macromolecules are _______________. A Proteins, carbohydrates, amino acids, steroids B Amino acids, lipids, carbohydrates, fats C Lipids, proteins, nucleic acids, carbohydrates D Steroids, nucleic acids, amino acids, lipids _______13. A common name for lipids is ___________ ...
Chapter 17: An Introduction to Organic Chemistry, Biochemistry, and
... that the bond angles around each carbon atom are either 90° or 180° and that the carbon atoms lie in a straight line. In contrast, the ball-and-stick and space-filling models in Figure 17.1 on the next page show that the angles are actually about 109° and that the carbons are in a zigzag arrangement ...
... that the bond angles around each carbon atom are either 90° or 180° and that the carbon atoms lie in a straight line. In contrast, the ball-and-stick and space-filling models in Figure 17.1 on the next page show that the angles are actually about 109° and that the carbons are in a zigzag arrangement ...
ch14
... Compounds of 3A elements have more covalent character than similar 2A compounds. Aluminum has the physical properties of a metal, but its halides exist as covalent dimers. ...
... Compounds of 3A elements have more covalent character than similar 2A compounds. Aluminum has the physical properties of a metal, but its halides exist as covalent dimers. ...
the ap chemistry summer assignment
... general chemistry class, but AP Chemistry is very different. Rather than memorizing how to do particular types of problems, you must really understand the chemistry and be able to apply it to different kinds of problems. AP Chemistry is a difficult course. To succeed you must keep up with the assign ...
... general chemistry class, but AP Chemistry is very different. Rather than memorizing how to do particular types of problems, you must really understand the chemistry and be able to apply it to different kinds of problems. AP Chemistry is a difficult course. To succeed you must keep up with the assign ...
powerpoint
... Do single replacement reactions always occur? NO! A metal will only replace a less reactive metal. The activity series is a way to predict whether or not certain reactions will occur. Any specific metal can replace any metal listed below it that is in a compound. It cannot replace any metal ...
... Do single replacement reactions always occur? NO! A metal will only replace a less reactive metal. The activity series is a way to predict whether or not certain reactions will occur. Any specific metal can replace any metal listed below it that is in a compound. It cannot replace any metal ...
7.2 Writing Chemical Equations
... In writing chemical equations, reactants are written to the left of the arrow and the products are written to the right. Separate reactants and products are separated by ...
... In writing chemical equations, reactants are written to the left of the arrow and the products are written to the right. Separate reactants and products are separated by ...
Chapter 11 Chemical Reactions
... If the combustion is incomplete, the products will be CO (or possibly just C) and H2O. ...
... If the combustion is incomplete, the products will be CO (or possibly just C) and H2O. ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.