Redox
... This method is typically used for organic compounds, which contain many carbon, hydrogen, and oxygen atoms The advantage of the effective charge method is that you can determine which atom has been oxidized or reduced To determine effective charges, we will need to use some more advanced topics, suc ...
... This method is typically used for organic compounds, which contain many carbon, hydrogen, and oxygen atoms The advantage of the effective charge method is that you can determine which atom has been oxidized or reduced To determine effective charges, we will need to use some more advanced topics, suc ...
Teacher Demo/Student Activity: Elephant`s Toothpaste
... occurring when magnesium burns or in the production of oxygen from hydrogen peroxide; the reaction of iron and copper sulphate; reactions occurring when fossil fuels burn) SNC2P A1.10 draw conclusions based on inquiry results and research findings, and justify their conclusions C2.3 conduct and ...
... occurring when magnesium burns or in the production of oxygen from hydrogen peroxide; the reaction of iron and copper sulphate; reactions occurring when fossil fuels burn) SNC2P A1.10 draw conclusions based on inquiry results and research findings, and justify their conclusions C2.3 conduct and ...
10th Carbon and Its Compounds Solved Paper-3
... 10. An organic compound A is widely used as a preservative in pickles and has a molecular formula C2H4O2. This compound reacts with ethanol to form a sweet smelling compound B. i) Identify the compound A. ii) Write the chemical equation for its reaction with Ethanol to form compound B. iii) How can ...
... 10. An organic compound A is widely used as a preservative in pickles and has a molecular formula C2H4O2. This compound reacts with ethanol to form a sweet smelling compound B. i) Identify the compound A. ii) Write the chemical equation for its reaction with Ethanol to form compound B. iii) How can ...
Undergraduate Chemistry Major Handbook - JHU Chemistry
... covered. These topics will be explored through the use of laboratory experiments and problem solving, and the use of these principles in current research areas will be discussed. 030.105-106: First-year chemistry laboratories. Students will develop laboratory skills and learn about topics that compl ...
... covered. These topics will be explored through the use of laboratory experiments and problem solving, and the use of these principles in current research areas will be discussed. 030.105-106: First-year chemistry laboratories. Students will develop laboratory skills and learn about topics that compl ...
Synthesis, Crystal-Structure Determination and Magnetic Properties
... fortunately, both substances were received as phase-pure products, as evidenced by X-ray powder diffraction (for example, see Figure 1) and elemental analysis based on atomic-absorption spectroscopy, the latter yielding metal contents of 99.92% (CoNCN) and 99.70% (NiNCN) compared to the ideal stoich ...
... fortunately, both substances were received as phase-pure products, as evidenced by X-ray powder diffraction (for example, see Figure 1) and elemental analysis based on atomic-absorption spectroscopy, the latter yielding metal contents of 99.92% (CoNCN) and 99.70% (NiNCN) compared to the ideal stoich ...
Lecture Notes
... which a substance changes from the solid state to the liquid state. For pure water themelting point is 32oF or 0oC. The Freezing point of a substance is the same as the melting point, since the process of freezing is the opposite of melting. The boiling point is defined as that temperature at which ...
... which a substance changes from the solid state to the liquid state. For pure water themelting point is 32oF or 0oC. The Freezing point of a substance is the same as the melting point, since the process of freezing is the opposite of melting. The boiling point is defined as that temperature at which ...
Atoms, Elements and Compounds Home
... a) Carbon, oxygen, iron and gold are all ______________ (metals/elements/compounds). b) Things which contain only one type of atom are called __________________ (elements/compounds). c) Compounds always contain __________________ (one/more than one) type of atom. d) The chemical name for common salt ...
... a) Carbon, oxygen, iron and gold are all ______________ (metals/elements/compounds). b) Things which contain only one type of atom are called __________________ (elements/compounds). c) Compounds always contain __________________ (one/more than one) type of atom. d) The chemical name for common salt ...
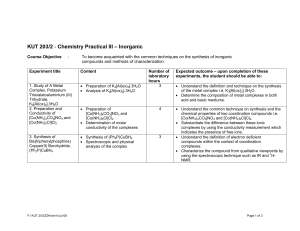
KUT 203/2 - Chemistry Practical III (Inorganic Chemistry)
... • Understand the correlation between the colors of transition metal or metal ion with its oxidation state i.e. VO2+ (yellow), VO2+ (blue) etc. • Determine the composition of a metal complex of which the metal exists in various oxidation states by using the titration technique. • Synthesize several c ...
... • Understand the correlation between the colors of transition metal or metal ion with its oxidation state i.e. VO2+ (yellow), VO2+ (blue) etc. • Determine the composition of a metal complex of which the metal exists in various oxidation states by using the titration technique. • Synthesize several c ...
Bioorganic chemistry-a scientific endeavour in continuous
... of the reagent has aided not only the correct sequencing of peptide segments of many proteins but also the production, through genetic engineering, of human insulin by means of a methionyl-containing precursor. Organic chemistry approaching 100% conversion at each. step provides the basis of modern ...
... of the reagent has aided not only the correct sequencing of peptide segments of many proteins but also the production, through genetic engineering, of human insulin by means of a methionyl-containing precursor. Organic chemistry approaching 100% conversion at each. step provides the basis of modern ...
Kinetics of the Selective Reaction of Diazonium Salts with Single
... Current methods of synthesis for carbon nanotubes (CNTs) usually produce heterogeneous mixtures of different nanotube diameters, thus a mixture of electronic properties. Consequently, many techniques have been developed in attempts to separate nanotubes according to their electronic type, w ith vary ...
... Current methods of synthesis for carbon nanotubes (CNTs) usually produce heterogeneous mixtures of different nanotube diameters, thus a mixture of electronic properties. Consequently, many techniques have been developed in attempts to separate nanotubes according to their electronic type, w ith vary ...
Elements and Compounds
... Matter has mass and takes up space. Mass measures how much matter is present and volume measures how much space the matter occupies. Matter occurs as elements, compounds or mixtures. An element is a pure substance that cannot be broken down into simpler different substances. A sample of an element m ...
... Matter has mass and takes up space. Mass measures how much matter is present and volume measures how much space the matter occupies. Matter occurs as elements, compounds or mixtures. An element is a pure substance that cannot be broken down into simpler different substances. A sample of an element m ...
Syllabus - The Society of Flavor Chemists
... No attempt has been made to divide the subject matter into Certified and Apprentice SFC membership categories. It is the expected that Certified members will have broader and deeper knowledge than Apprentice members, but it is not the intention here to elaborate who should know what. It is not likel ...
... No attempt has been made to divide the subject matter into Certified and Apprentice SFC membership categories. It is the expected that Certified members will have broader and deeper knowledge than Apprentice members, but it is not the intention here to elaborate who should know what. It is not likel ...
MIDTERM REVIEW UNIT 1: Mass/Measurement
... 11. In a reaction between lead (II) nitrate and copper (II) bromide, do the following: a) write the formulas for the reactants and the products and balance the equation b) If 0.67 moles of copper (II) ...
... 11. In a reaction between lead (II) nitrate and copper (II) bromide, do the following: a) write the formulas for the reactants and the products and balance the equation b) If 0.67 moles of copper (II) ...
Chapter 11 Chemical Reactions
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
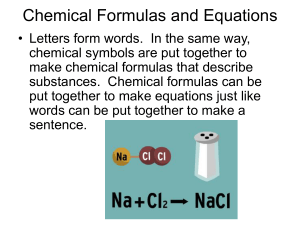
Chemical Formulas and Equations
... Chemical Formulas and Equations • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a sentence. ...
... Chemical Formulas and Equations • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a sentence. ...
Chemistry I Syllabus 2011-2012
... Essential Questions: 1. What specific properties of materials allow them to be classified as metals or nonmetals? 2. How is the relative mass of atoms determined? What does that indicate about the way in which they react? 3. What evidence is there for the existence of electrons and the nucleus? 4. H ...
... Essential Questions: 1. What specific properties of materials allow them to be classified as metals or nonmetals? 2. How is the relative mass of atoms determined? What does that indicate about the way in which they react? 3. What evidence is there for the existence of electrons and the nucleus? 4. H ...
Chapter 11 Chemical Reactions
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
Candle Mass Lab and the Law of Conservation of Matter Notes.
... • A. Subscripts • B. Mass numbers • C. Atomic numbers • D. coefficients ...
... • A. Subscripts • B. Mass numbers • C. Atomic numbers • D. coefficients ...
Chapter 11 Chemical Reactions
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
... 1) Assemble the correct formulas for all the reactants and products, using “+” and “→” 2) Count the number of atoms of each type appearing on both sides 3) Balance the elements one at a time by adding coefficients (the numbers in front) where you need more - save balancing the H and O until LAST! ...
Chapter 6
... Displacement Reaction): These are reactions where two reactants just exchange parts. ...
... Displacement Reaction): These are reactions where two reactants just exchange parts. ...
File - Flipped Out Science with Mrs. Thomas!
... coefficients mean? How can you tell which elements are present. Know the Law of Conservation of Mass and how it is applied to chemical reactions Know what makes a compound organic or not Know the difference between physical and chemical properties and changes Exothermic and endothermic react ...
... coefficients mean? How can you tell which elements are present. Know the Law of Conservation of Mass and how it is applied to chemical reactions Know what makes a compound organic or not Know the difference between physical and chemical properties and changes Exothermic and endothermic react ...
200 Things to Know to Pass the Chemistry Regents
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
200 Ways to Pass the Chemistry
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
200things2know
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
... 96. Metallic bonds can be thought of as a crystalline lattice of kernels surrounded by a “sea” of mobile valence electrons. Metallic bonding occurs between atoms of sulfur sodium fluoride sodium carbon 97. Atoms are most stable when they have 8 valence electrons (an octet) and tend to form ions to o ...
Organic chemistry

Organic chemistry is a chemistry subdiscipline involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms. Study of structure includes many physical and chemical methods to determine the chemical composition and the chemical constitution of organic compounds and materials. Study of properties includes both physical properties and chemical properties, and uses similar methods as well as methods to evaluate chemical reactivity, with the aim to understand the behavior of the organic matter in its pure form (when possible), but also in solutions, mixtures, and fabricated forms. The study of organic reactions includes probing their scope through use in preparation of target compounds (e.g., natural products, drugs, polymers, etc.) by chemical synthesis, as well as the focused study of the reactivities of individual organic molecules, both in the laboratory and via theoretical (in silico) study.The range of chemicals studied in organic chemistry include hydrocarbons (compounds containing only carbon and hydrogen), as well as myriad compositions based always on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (these, included in many organic chemicals in biology) and the radiostable elements of the halogens.In the modern era, the range extends further into the periodic table, with main group elements, including:Group 1 and 2 organometallic compounds, i.e., involving alkali (e.g., lithium, sodium, and potassium) or alkaline earth metals (e.g., magnesium)Metalloids (e.g., boron and silicon) or other metals (e.g., aluminium and tin)In addition, much modern research focuses on organic chemistry involving further organometallics, including the lanthanides, but especially the transition metals; (e.g., zinc, copper, palladium, nickel, cobalt, titanium and chromium)Finally, organic compounds form the basis of all earthly life and constitute a significant part of human endeavors in chemistry. The bonding patterns open to carbon, with its valence of four—formal single, double, and triple bonds, as well as various structures with delocalized electrons—make the array of organic compounds structurally diverse, and their range of applications enormous. They either form the basis of, or are important constituents of, many commercial products including pharmaceuticals; petrochemicals and products made from them (including lubricants, solvents, etc.); plastics; fuels and explosives; etc. As indicated, the study of organic chemistry overlaps with organometallic chemistry and biochemistry, but also with medicinal chemistry, polymer chemistry, as well as many aspects of materials science.