Part 2-ICHO-26-30
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
... The overall catalytic reaction is simple, whereas the reaction mechanism in the homogeneous phase is very complicated with a large number of reaction steps, and the course is difficult to control owing to a distinct chain character. With platinum as catalyst the significant reaction steps are: (i) A ...
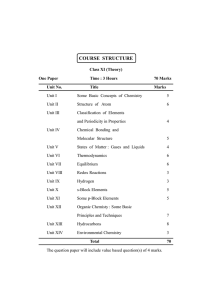
Grossmont College Chemistry 120 Laboratory Manual 6th Edition
... data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answer important questions that are intended to give you an understanding of the principles in which the experimental procedure is based as you perform the experiment. Scientists learn much by discu ...
... data, but do not clutter the calculation section with arithmetic details. Likewise, think through and answer important questions that are intended to give you an understanding of the principles in which the experimental procedure is based as you perform the experiment. Scientists learn much by discu ...
FREE Sample Here - We can offer most test bank and
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
... 3. The French chemist Antoine Lavoisier found that the weight of objects before burning and the weight of the products after burning were equal. He concluded that the total weight did not change during a process. Which of these best describes Lavoisier's conclusion? a. From observation, Lavoisier cr ...
ap 2005 chemistry_b scoring guidelines - AP Central
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
Higher Chemistry Resources Guide - Glow Blogs
... provided in the decolourisation of permanganate using rhubarb as described in the Practical Chemistry website from the Royal Society of Chemistry and the Nuffield Foundation. ...
... provided in the decolourisation of permanganate using rhubarb as described in the Practical Chemistry website from the Royal Society of Chemistry and the Nuffield Foundation. ...
Unit F325 - Equilibria, energetics and elements - High band
... Fuels cells based on systems such as III and IV are increasingly being used to generate energy. Discuss two advantages and two disadvantages of using fuels cells for energy rather than using fossil fuels. ...
... Fuels cells based on systems such as III and IV are increasingly being used to generate energy. Discuss two advantages and two disadvantages of using fuels cells for energy rather than using fossil fuels. ...
Worked solutions to the problems
... we know that you will employ best-practice to responsibly dispose or recycle the materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these ...
... we know that you will employ best-practice to responsibly dispose or recycle the materials that your students use and produce. Students should of course also make themselves aware of any hazards associated with the chemicals that they will be using in any exercise and we encourage you to bring these ...
Topic 3: Chemical Kinetics - Manitoba Education and Training
... Chemical kinetics crosses over into many other areas of science and engineering. Rates of metabolic reaction and the progress of reactions involved in growth and bone regeneration are studied by biologists. Automobile engineers want to decrease the rate of rusting of car bodies, while agricultural s ...
... Chemical kinetics crosses over into many other areas of science and engineering. Rates of metabolic reaction and the progress of reactions involved in growth and bone regeneration are studied by biologists. Automobile engineers want to decrease the rate of rusting of car bodies, while agricultural s ...
Higher Chemistry Resources Guide - Glow Blogs
... The following pages show the Mandatory Course key areas table from the SQA Higher Chemistry Course and Unit Support Notes. An additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are o ...
... The following pages show the Mandatory Course key areas table from the SQA Higher Chemistry Course and Unit Support Notes. An additional fourth column has been included which contains hyperlinks to useful resources. Please note: Practitioners are not required to use the resources listed – they are o ...
Guide to the Environmental Engineering Minor - 2015
... To enter the minor, students must be in good academic standing (2.0 grade-point average or better) and file a petition in the Office of Academic and Student Affairs, 6426 Boelter Hall. Required Lower Division Course (4 units): Mathematics 3C or 32A. Required Upper Division Courses (24 units minimum) ...
... To enter the minor, students must be in good academic standing (2.0 grade-point average or better) and file a petition in the Office of Academic and Student Affairs, 6426 Boelter Hall. Required Lower Division Course (4 units): Mathematics 3C or 32A. Required Upper Division Courses (24 units minimum) ...
Part 3-ICHO-31-35
... Edited by Anton Sirota ICHO International Information Centre, Bratislava, Slovakia ...
... Edited by Anton Sirota ICHO International Information Centre, Bratislava, Slovakia ...
Department of Chemistry
... or at the interface between two of them. A student admitted to a graduate program in chemistry in the United States is usually offered a teaching assistantship or fellowship. Chemists have a wide choice of academic, governmental, or industrial positions. By far the greatest percentage accept industr ...
... or at the interface between two of them. A student admitted to a graduate program in chemistry in the United States is usually offered a teaching assistantship or fellowship. Chemists have a wide choice of academic, governmental, or industrial positions. By far the greatest percentage accept industr ...
Chemistry Content Review Notes
... The document is a compilation of information found in the Virginia Department of Education (VDOE) Curriculum Framework, Enhanced Scope and Sequence, and Released Test items. In addition to VDOE information, Glencoe Textbook Series and resources have been used. Finally, information from various websi ...
... The document is a compilation of information found in the Virginia Department of Education (VDOE) Curriculum Framework, Enhanced Scope and Sequence, and Released Test items. In addition to VDOE information, Glencoe Textbook Series and resources have been used. Finally, information from various websi ...
Materials Chemistry Prof. S. Sunder Manoharan Department of
... Now, when we think of the synthesis we always think is very easy because it does not involve much of wet chemical route. Therefore, we often think solid state chemistry synthesis is often a very simple phenomenon, but what we see is it looks deceptively simple, but when you actually go through the ...
... Now, when we think of the synthesis we always think is very easy because it does not involve much of wet chemical route. Therefore, we often think solid state chemistry synthesis is often a very simple phenomenon, but what we see is it looks deceptively simple, but when you actually go through the ...
Equilibrium Notes - Chemistry Teaching Resources
... H + (aq) + OH – (aq) → H 2 O(l) As research continued throughout the nineteenth and early twentieth centuries, the definitions had to be refined. For example, pure hydrogen chloride is a gas that contains no H + ions and ammonia has no OH – ions but can neutralise an acid. It was also discovered tha ...
... H + (aq) + OH – (aq) → H 2 O(l) As research continued throughout the nineteenth and early twentieth centuries, the definitions had to be refined. For example, pure hydrogen chloride is a gas that contains no H + ions and ammonia has no OH – ions but can neutralise an acid. It was also discovered tha ...
Part-1
... Osmotic pressure of a solution is directly proportional to the number of moles of solute dissolved per litre of solution at a given temperature. Solutions having equal molar concentration and equal osmotic pressure at a given temperature are called isotonic solutions, e.g., A 0.90% (mass/volume) sol ...
... Osmotic pressure of a solution is directly proportional to the number of moles of solute dissolved per litre of solution at a given temperature. Solutions having equal molar concentration and equal osmotic pressure at a given temperature are called isotonic solutions, e.g., A 0.90% (mass/volume) sol ...
Chapter 6 Thermochemistry - Suffolk County Community College
... attachment between atoms, the atoms’ positions relative to each other in the molecule, or the molecules, relative positions in the structure Tro: Chemistry: A Molecular Approach, 2/e ...
... attachment between atoms, the atoms’ positions relative to each other in the molecule, or the molecules, relative positions in the structure Tro: Chemistry: A Molecular Approach, 2/e ...
Examiners` Report November 2012 GCSE Chemistry
... In common with all the other science GCSE examination papers for the current specification, this was a one hour, 60-mark paper. Like the previous paper, the paper contained six questions based largely around one of the topics in the specification. The last two questions each contained an extended wr ...
... In common with all the other science GCSE examination papers for the current specification, this was a one hour, 60-mark paper. Like the previous paper, the paper contained six questions based largely around one of the topics in the specification. The last two questions each contained an extended wr ...
Chapter 6 - Suffolk County Community College
... type of material 1000 J of heat energy will raise the temperature of 100 g of sand 12 °C, but only raise the temperature of 100 g of water by 2.4 °C Tro: Chemistry: A Molecular Approach, 2/e ...
... type of material 1000 J of heat energy will raise the temperature of 100 g of sand 12 °C, but only raise the temperature of 100 g of water by 2.4 °C Tro: Chemistry: A Molecular Approach, 2/e ...
chm 205 - National Open University of Nigeria
... manufactured on a large scale by heating coke with silica at ~2775/K for 25-35 hours. You may be aware that a new form of element carbon called fullerenes in which the atoms are arranged in closed shells was discovered in 1985 by Dr Robert F. Curl, Dr. Harold W. Croto and Dr. Richard E. Smalley who ...
... manufactured on a large scale by heating coke with silica at ~2775/K for 25-35 hours. You may be aware that a new form of element carbon called fullerenes in which the atoms are arranged in closed shells was discovered in 1985 by Dr Robert F. Curl, Dr. Harold W. Croto and Dr. Richard E. Smalley who ...
SAMPLE QUESTION PAPER SIR.S.M.TAHIR CHEMISTRY Mob: 9557076999
... Explain, why does the atomic radii increases considerably from N to P but very little increase is observed from As to Bi. ...
... Explain, why does the atomic radii increases considerably from N to P but very little increase is observed from As to Bi. ...
Chemistry and Biochemistry
... Graduate students must maintain a GPA of 3.0/4.0. Only courses applicable to the graduate program, excluding those for making up deficiencies or satisfying proficiencies, are counted in the GPA. If the cumulative GPA drops below 3.0 for one semester, the student will be placed on academic probation. ...
... Graduate students must maintain a GPA of 3.0/4.0. Only courses applicable to the graduate program, excluding those for making up deficiencies or satisfying proficiencies, are counted in the GPA. If the cumulative GPA drops below 3.0 for one semester, the student will be placed on academic probation. ...