Challenge Problems
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
... was made by the German chemist Johann Wolfgang Döbereiner (1780–1849). In 1816, Döbereiner noticed that the then accepted atomic mass of strontium (50) was midway between the atomic masses of calcium (27.5) and barium (72.5). Note that the accepted atomic masses for these elements today are very dif ...
Unit 8: Reactions
... The mass on the reactants (left) side of the arrow and the mass on the products (right) side of the arrow MUST equal each other as the Law of Conservation of Mass states that mass may not be created or destroyed in any chemical reaction or physical change. Missing Mass examples: 1. Given that 35.0 g ...
... The mass on the reactants (left) side of the arrow and the mass on the products (right) side of the arrow MUST equal each other as the Law of Conservation of Mass states that mass may not be created or destroyed in any chemical reaction or physical change. Missing Mass examples: 1. Given that 35.0 g ...
Chapter 4
... related terms: oxidizing agent = the substance that is reduced (Cl2) reducing agent = the substance that is oxidized (Na) Oxidation and reduction always occur together so that there is no net loss or gain of electrons overall. ...
... related terms: oxidizing agent = the substance that is reduced (Cl2) reducing agent = the substance that is oxidized (Na) Oxidation and reduction always occur together so that there is no net loss or gain of electrons overall. ...
Atomic Structure
... The statements (i) “In filling a group of orbitals of equal energy it is energetically preferable to assign electrons to empty orbitals rather than pair them into a particular orbital. (ii) When two electrons are placed in two different orbitals, energy is lower if the espins are parallel” are valid ...
... The statements (i) “In filling a group of orbitals of equal energy it is energetically preferable to assign electrons to empty orbitals rather than pair them into a particular orbital. (ii) When two electrons are placed in two different orbitals, energy is lower if the espins are parallel” are valid ...
GCE Chemistry Specification (From 2015 - WALES ONLY
... higher education, including Chemistry at degree level and in related areas such as Medicine, Biochemistry and Chemical Engineering. It also develops a range of knowledge and skills essential for direct entry into employment in many chemistryrelated fields. In addition, the specification provides a c ...
... higher education, including Chemistry at degree level and in related areas such as Medicine, Biochemistry and Chemical Engineering. It also develops a range of knowledge and skills essential for direct entry into employment in many chemistryrelated fields. In addition, the specification provides a c ...
19_Worked_Examples
... (a) The reactants are six molecules of gas, and the products are three molecules of gas and four molecules of liquid. Thus, the number of molecules of gas has decreased significantly during the reaction. By using the general rules discussed in Section 19.3, we expect a decrease in the number of gas ...
... (a) The reactants are six molecules of gas, and the products are three molecules of gas and four molecules of liquid. Thus, the number of molecules of gas has decreased significantly during the reaction. By using the general rules discussed in Section 19.3, we expect a decrease in the number of gas ...
17 ADSORPTION AND CATALYSIS S MODULE - 5
... Adsorption Isotherm : One of the drawbacks of the Freundlich adsorption isotherm is that it fails at high pressure of the gas. Langmuir derived an adsorption isotherm on theoretical considerations based on kinetic theory of gases. This is named as the Langmuir adosrption isotherm. This isotherm is b ...
... Adsorption Isotherm : One of the drawbacks of the Freundlich adsorption isotherm is that it fails at high pressure of the gas. Langmuir derived an adsorption isotherm on theoretical considerations based on kinetic theory of gases. This is named as the Langmuir adosrption isotherm. This isotherm is b ...
CHM203 - National Open University of Nigeria
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
... The melting point of a substance can be defined as the temperature at which it undergoes the transition from the solid to the liquid state. Pure crystalline solids have sharp melting points. Thus, melting point is used as an important physical property both for the identification of organic compound ...
Association in Solution III - Engineering Conferences International
... Hydrophobic interactions, driven by the inability of water molecules to hydrogen bond in the vicinity of hydrophobic molecules and surfaces resulting in strong, long range attractive interactions between hydrophobic moieties, are ubiquitous in the self-assembly of complex structures and many physica ...
... Hydrophobic interactions, driven by the inability of water molecules to hydrogen bond in the vicinity of hydrophobic molecules and surfaces resulting in strong, long range attractive interactions between hydrophobic moieties, are ubiquitous in the self-assembly of complex structures and many physica ...
The integration of flow reactors into synthetic organic chemistry
... conduct chemical synthesis has remained relatively unchanged for over two centuries.12 – 14 Remarkably, apparatus such as standard glass round bottom flasks, condensers, measuring cylinders, test tubes and Bunsen burners are all still commonly in use today despite them being invented over 160 years ...
... conduct chemical synthesis has remained relatively unchanged for over two centuries.12 – 14 Remarkably, apparatus such as standard glass round bottom flasks, condensers, measuring cylinders, test tubes and Bunsen burners are all still commonly in use today despite them being invented over 160 years ...
chemistry - The Aga Khan University
... Normal Oxides, Peroxides, Super Oxides and their Stability 13.2.3.2 Reactions of Oxides with Water and Dilute Acids 13.2.4 Reactions with Chlorine 13.2.5 Effect of Heat on Nitrates, Carbonates and Hydrogen-Carbonates explaining the Trend in Terms of the Polarizing Ability of the ...
... Normal Oxides, Peroxides, Super Oxides and their Stability 13.2.3.2 Reactions of Oxides with Water and Dilute Acids 13.2.4 Reactions with Chlorine 13.2.5 Effect of Heat on Nitrates, Carbonates and Hydrogen-Carbonates explaining the Trend in Terms of the Polarizing Ability of the ...
Unit_1_Introduction_to_Chemistry_Student[1]
... 1. Safety goggles must be worn at all times during a lab. This rule must be followed not matter what you are doing during the lab period. 2. Contact lenses are not allowed. Even when worn under safety goggles, since fumes from the chemicals can get under them and cause serious injuries or blindness. ...
... 1. Safety goggles must be worn at all times during a lab. This rule must be followed not matter what you are doing during the lab period. 2. Contact lenses are not allowed. Even when worn under safety goggles, since fumes from the chemicals can get under them and cause serious injuries or blindness. ...
Part 1-ICHO-21-25
... This publication contains the competition problems (Volume 2) from the 21st – 40th International Chemistry Olympiads (ICHO) organized in the years 1989 – 2008 and is a continuation of the publication that appeared last year as Volume 1 and contained competition problems from the first twenty ICHOs. ...
... This publication contains the competition problems (Volume 2) from the 21st – 40th International Chemistry Olympiads (ICHO) organized in the years 1989 – 2008 and is a continuation of the publication that appeared last year as Volume 1 and contained competition problems from the first twenty ICHOs. ...
Sample Exercise 19.1 Identifying Spontaneous Processes
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
An Empirical Survey of the Ramifications of a Green
... Green, smart and sustainable are ‘buzz words’ that in recent years have increasingly been used in association with development and the environment arenas. Great emphasis is placed upon economic development which enhances general human development in its many dimensions. The truth is that Green Econo ...
... Green, smart and sustainable are ‘buzz words’ that in recent years have increasingly been used in association with development and the environment arenas. Great emphasis is placed upon economic development which enhances general human development in its many dimensions. The truth is that Green Econo ...
Slide 1
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
19 BROWN Chemical Thermodynamics PPTSExercise
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
... Analyze: We are asked to judge whether each process will proceed spontaneously in the direction indicated, in the reverse direction, or in neither direction. Plan: We need to think about whether each process is consistent with our experience about the natural direction of events or whether we expect ...
Chemistry Standards Clarification
... expected to know and be able to do by the end of high school and outline the parameters for receiving high school credit as recently mandated by the Merit Curriculum legislation in the state of Michigan. The Science Content Expectations Documents and the Michigan Merit Curriculum Document have raise ...
... expected to know and be able to do by the end of high school and outline the parameters for receiving high school credit as recently mandated by the Merit Curriculum legislation in the state of Michigan. The Science Content Expectations Documents and the Michigan Merit Curriculum Document have raise ...
Physical Chemistry Problems. ©Mike Lyons 2009
... A compendium of past examination questions set on Physical Chemistry on the JF Chemistry paper and problem sheets associated with CH1101 Physical Chemistry (Lyons). You will not fully understand Physical Chemistry if you cannot solve numerical problems on the material delivered in lectures. Formulat ...
... A compendium of past examination questions set on Physical Chemistry on the JF Chemistry paper and problem sheets associated with CH1101 Physical Chemistry (Lyons). You will not fully understand Physical Chemistry if you cannot solve numerical problems on the material delivered in lectures. Formulat ...
Specification and sample assessment material - Edexcel
... investigative skills based on correct and safe laboratory techniques ...
... investigative skills based on correct and safe laboratory techniques ...
O - FH Münster
... decomposes at high temp. to NaF and HF decomposes at high temp. to NH3 and HF somewhat hygroscopic ...
... decomposes at high temp. to NaF and HF decomposes at high temp. to NH3 and HF somewhat hygroscopic ...
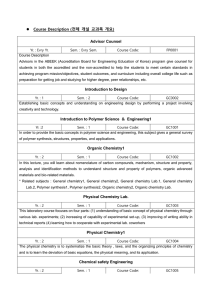
Course Description Word File
... Design for Polymer Analysis Yr. : 3 Sem. : 2 Course Code: GC1022 To increase the design ability of polymer materials by studying the fundamentals of polymer materials and characterization, by selecting polymer materials, which can be found in our living environment and industries, and performing tea ...
... Design for Polymer Analysis Yr. : 3 Sem. : 2 Course Code: GC1022 To increase the design ability of polymer materials by studying the fundamentals of polymer materials and characterization, by selecting polymer materials, which can be found in our living environment and industries, and performing tea ...
Unit 12: Electrochemistry
... Redox Reactions Objective: What steps do we take to balance Redox Reactions? Balancing Redox Reactions: Previous examples have shown how the spectator ions in a redox reaction may be ignored during redox reactions. We can therefore eliminate any spectator ions from the balancing of half-reactions. ...
... Redox Reactions Objective: What steps do we take to balance Redox Reactions? Balancing Redox Reactions: Previous examples have shown how the spectator ions in a redox reaction may be ignored during redox reactions. We can therefore eliminate any spectator ions from the balancing of half-reactions. ...
Stoichiometry - VernonScienceLSA
... Stoichiometry calculations allow us to find out how much of chemical #1 is involved in a chemical reaction based on the amount of chemical #2 involved. A typical problem might be “How many grams of chemical #1 must be reacted to produce 25.0 g of chemical #2?” or “What volume of chemical #1 at STP w ...
... Stoichiometry calculations allow us to find out how much of chemical #1 is involved in a chemical reaction based on the amount of chemical #2 involved. A typical problem might be “How many grams of chemical #1 must be reacted to produce 25.0 g of chemical #2?” or “What volume of chemical #1 at STP w ...
PART 3-ICHO 11-15
... substances: barium chloride, sodium sulphate, potassium chloride, magnesium nitrate, sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the abo ...
... substances: barium chloride, sodium sulphate, potassium chloride, magnesium nitrate, sodium orthophosphate, barium hydroxide, lead nitrate, potassium hydroxide, aluminium sulphate, sodium carbonate. Using only these solutions as reagents, determine in which of the numbered test tubes each of the abo ...









![Unit_1_Introduction_to_Chemistry_Student[1]](http://s1.studyres.com/store/data/022016930_1-98bbde1835e65f7e36a50862854faaef-300x300.png)