NOBLE-GAS CHEMISTRY
... 40 years after the initial preparation.2 The history of the breaking of the inertness of the noble gases is certainly complex and fascinating, full of many misleading reports, surprises, and ingenuity.3 The chemical bonds between noble gas and other atoms are usually quite fragile due to various red ...
... 40 years after the initial preparation.2 The history of the breaking of the inertness of the noble gases is certainly complex and fascinating, full of many misleading reports, surprises, and ingenuity.3 The chemical bonds between noble gas and other atoms are usually quite fragile due to various red ...
Chemical Reactivity as Described by Quantum Chemical Methods
... aspects can be included when further linking reactivity to the property vertex. In the field of organic chemistry, the ab initio calculation of functional group properties and their use in studies on acidity and basicity is discussed together with the use of DFT descriptors to study the kinetics of ...
... aspects can be included when further linking reactivity to the property vertex. In the field of organic chemistry, the ab initio calculation of functional group properties and their use in studies on acidity and basicity is discussed together with the use of DFT descriptors to study the kinetics of ...
Chemical Monitoring and Management by Ahmad Shah Idil
... Hence, collaboration between chemists is essential for solving chemical issues, or when dealing with large amounts of data being collected, as the chemists provide input and expertise from their own particular field, for a common goal. ...
... Hence, collaboration between chemists is essential for solving chemical issues, or when dealing with large amounts of data being collected, as the chemists provide input and expertise from their own particular field, for a common goal. ...
CHemiStrY - Cabrillo College
... science and much of a student’s time will be spent in the laboratory. A chemistry major usually transfers to a four-year institution to complete a bachelor’s degree. Many also go on to earn Masters or Ph.D.s, since advanced degrees generally lead to more rewarding careers. Cabrillo’s chemistry progr ...
... science and much of a student’s time will be spent in the laboratory. A chemistry major usually transfers to a four-year institution to complete a bachelor’s degree. Many also go on to earn Masters or Ph.D.s, since advanced degrees generally lead to more rewarding careers. Cabrillo’s chemistry progr ...
Enhancing the secondary-tertiary transition in
... a new world that is very different to their high-school environment. At many Australian tertiary institutions, incoming students experience very large lecture theatres with classes of hundreds of students, and the delivery and assessment of content is markedly different to high school. From the inst ...
... a new world that is very different to their high-school environment. At many Australian tertiary institutions, incoming students experience very large lecture theatres with classes of hundreds of students, and the delivery and assessment of content is markedly different to high school. From the inst ...
Instruments for Radiation Detection and Measurement
... exposure to light, change in temperature, and radiolysis The longer a compound is exposed to these conditions, the more it will tend to break down How to lessen the degradation of the material? Substances such as sodium ascorbate, ascorbic acid, and sodium sulfite are often added to maintain the sta ...
... exposure to light, change in temperature, and radiolysis The longer a compound is exposed to these conditions, the more it will tend to break down How to lessen the degradation of the material? Substances such as sodium ascorbate, ascorbic acid, and sodium sulfite are often added to maintain the sta ...
NUCL 1 Early life of Albert Ghiorso: Preparation for future role as
... using prosthetic groups such as N-succinimidyl-4-[18F]fluorobenzoate ([18F]SFB) and N[6-(4-[18F]fluorobenzylidene)aminooxyhexyl]maleimide ([18F]FBAM). However, these labeling methods often require multiple steps and results in low radiochemical yields. Consequently, the search has been ongoing to de ...
... using prosthetic groups such as N-succinimidyl-4-[18F]fluorobenzoate ([18F]SFB) and N[6-(4-[18F]fluorobenzylidene)aminooxyhexyl]maleimide ([18F]FBAM). However, these labeling methods often require multiple steps and results in low radiochemical yields. Consequently, the search has been ongoing to de ...
lecture ch1-3 chem161pikul
... Precision – How well set of repeated measurements of same quantity agree with each other – More significant figures equals more precise measurement Jesperson, Brady, Hyslop. Chemistry: The Molecular Nature of Matter, 6E ...
... Precision – How well set of repeated measurements of same quantity agree with each other – More significant figures equals more precise measurement Jesperson, Brady, Hyslop. Chemistry: The Molecular Nature of Matter, 6E ...
Science - AP Chemistry - Grades 11-12
... exams. The College Board AP Chemistry exam will be administered. Laboratory work is an essential part of the AP Chemistry course. At least six class periods per week are scheduled for the class. Within that time, a minimum of one double period per week should be spent conducting laboratory work. The ...
... exams. The College Board AP Chemistry exam will be administered. Laboratory work is an essential part of the AP Chemistry course. At least six class periods per week are scheduled for the class. Within that time, a minimum of one double period per week should be spent conducting laboratory work. The ...
Physical chemistry and transition elements 5.1 Rates, equilibrium
... reaction is −292.7 J mol−1 K−1. Thus no matter what the value of T, ∆G can never be negative [2 marks for worked example]. The ∆H value for N2 + O2 → 2NO is −180.4 kJ mol−1 for 2 moles of NO formed. Thus the reactants are thermodynamically unstable with respect to the products [1]. However, because ...
... reaction is −292.7 J mol−1 K−1. Thus no matter what the value of T, ∆G can never be negative [2 marks for worked example]. The ∆H value for N2 + O2 → 2NO is −180.4 kJ mol−1 for 2 moles of NO formed. Thus the reactants are thermodynamically unstable with respect to the products [1]. However, because ...
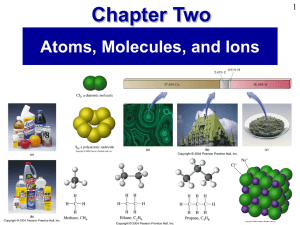
Prentice Hall Ch 02 Atoms Molecules Ions
... • How many protons are in chlorine-35? • How many protons are in chlorine-37? • How many neutrons are in chlorine-37? ...
... • How many protons are in chlorine-35? • How many protons are in chlorine-37? • How many neutrons are in chlorine-37? ...
Sample Exercise 20.1 Identifying Oxidizing and Reducing Agents
... is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solution (such as platinum foil) is suspended in each solution, and the two ...
... is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solution (such as platinum foil) is suspended in each solution, and the two ...
Worked out problems
... is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solution (such as platinum foil) is suspended in each solution, and the two ...
... is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solution (such as platinum foil) is suspended in each solution, and the two ...
MOLES! - ChemGod.com
... large numbers of things. After all, atoms and molecules are very small; if you have a collection of them that you can see, it has a lot of particles in it! ...
... large numbers of things. After all, atoms and molecules are very small; if you have a collection of them that you can see, it has a lot of particles in it! ...
Tutorial 4 (PowerPoint)
... • Tip can be operated in “dynamic mode” • The tip and cantilever (beam with the tip on it) have a mechanical natural resonance • The resonance will change as external forces from the sample are exerted on it • The tip’s vibration amplitude must be much less than the distance between it and the sampl ...
... • Tip can be operated in “dynamic mode” • The tip and cantilever (beam with the tip on it) have a mechanical natural resonance • The resonance will change as external forces from the sample are exerted on it • The tip’s vibration amplitude must be much less than the distance between it and the sampl ...
Brochure BITSAT-2011
... the use of keyboard or mouse. Each computer is connected to a server, which prepares the question set and delivers it to the candidate on the computer. This is unlike the traditional paper-pencil based test, which is generally offered on a single day to all candidates. BITSAT-2011 will be offered ov ...
... the use of keyboard or mouse. Each computer is connected to a server, which prepares the question set and delivers it to the candidate on the computer. This is unlike the traditional paper-pencil based test, which is generally offered on a single day to all candidates. BITSAT-2011 will be offered ov ...
graduate school of pharmaceutical sciences faculty of
... chemistry covering the basic concepts of organic reactions, stereochemistry, synthesis, and spectroscopic methods for undergraduate students. The Department provides graduate students with seminars which review and discuss recent important organic and organometallic reactions. Laboratory practice in ...
... chemistry covering the basic concepts of organic reactions, stereochemistry, synthesis, and spectroscopic methods for undergraduate students. The Department provides graduate students with seminars which review and discuss recent important organic and organometallic reactions. Laboratory practice in ...
chemistry
... Record the number of your choice for each Part A and Part B–1 multiple-choice question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You ...
... Record the number of your choice for each Part A and Part B–1 multiple-choice question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You ...
Fundamentals of Chemistry
... The diameter of the atom is determined by the range of the electrons in their travels around the nucleus and is approximately 10-8 cm. The diameter of the nucleus is roughly 10,000 times smaller, approximately 10-13 to 10-12 cm. Because the nucleus is composed of neutrons and protons that are about ...
... The diameter of the atom is determined by the range of the electrons in their travels around the nucleus and is approximately 10-8 cm. The diameter of the nucleus is roughly 10,000 times smaller, approximately 10-13 to 10-12 cm. Because the nucleus is composed of neutrons and protons that are about ...
Chapter 12
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
Chapter 12
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
... It is important to understand that when we say that the atomic mass of carbon is 12.01 amu, we are referring to the average value. If carbon atoms could be examined individually, we would find either an atom of atomic mass 12.00000 amu or one of 13.00335 amu, but never one of 12.01 amu. Example 3.1 ...
Congratulations! You have signed up for AP Chemistry for this year
... 400 B.C.—Greeks—proposed all matter was make up of 4 “elements” : fire, earth, water and air Democritus—first to use the term atomos to describe the ultimate, smallest particles of matter Next 2,000 years—alchemy—a pseudoscience where people thought they could turn metals into gold. Some good chemis ...
... 400 B.C.—Greeks—proposed all matter was make up of 4 “elements” : fire, earth, water and air Democritus—first to use the term atomos to describe the ultimate, smallest particles of matter Next 2,000 years—alchemy—a pseudoscience where people thought they could turn metals into gold. Some good chemis ...
Chemistry 30 - SharpSchool
... this led to the empirical generalization known as the ______________ ___________________________________________ which says that there is a ________________________________ between the concentrations of the products and the concentrations of the reactants at equilibrium ...
... this led to the empirical generalization known as the ______________ ___________________________________________ which says that there is a ________________________________ between the concentrations of the products and the concentrations of the reactants at equilibrium ...
Chemistry 30 - SharpSchool
... this led to the empirical generalization known as the ______________ ___________________________________________ which says that there is a ________________________________ between the concentrations of the products and the concentrations of the reactants at equilibrium ...
... this led to the empirical generalization known as the ______________ ___________________________________________ which says that there is a ________________________________ between the concentrations of the products and the concentrations of the reactants at equilibrium ...
Chapter - WTPS.org
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
Analytical chemistry

Analytical chemistry is the study of the separation, identification, and quantification of the chemical components of natural and artificial materials. Qualitative analysis gives an indication of the identity of the chemical species in the sample, and quantitative analysis determines the amount of certain components in the substance. The separation of components is often performed prior to analysis.Analytical methods can be separated into classical and instrumental. Classical methods (also known as wet chemistry methods) use separations such as precipitation, extraction, and distillation and qualitative analysis by color, odor, or melting point. Classical quantitative analysis is achieved by measurement of weight or volume. Instrumental methods use an apparatus to measure physical quantities of the analyte such as light absorption, fluorescence, or conductivity. The separation of materials is accomplished using chromatography, electrophoresis or field flow fractionation methods.Analytical chemistry is also focused on improvements in experimental design, chemometrics, and the creation of new measurement tools to provide better chemical information. Analytical chemistry has applications in forensics, bioanalysis, clinical analysis, environmental analysis, and materials analysis.