Chapter22_LEC
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
... • the most abundant elements of the Earth’s crust are O and Si • silicates are covalent atomic solids of Si and O and minor amounts of other elements found in rocks, soils, and clays silicates have variable structures – leading to the variety of properties found in rocks, clays, and soils Tro, Ch ...
Document
... In the process of cheese making a bacterial culture is added to milk, which causes the milk to separate into the curds (solid cheese) and whey (remaining liquid). During this process the bacteria convert the lactose present in milk, into lactic acid, CH3CHOHCOOH. Lactic acid is a weak, monoprotic, o ...
... In the process of cheese making a bacterial culture is added to milk, which causes the milk to separate into the curds (solid cheese) and whey (remaining liquid). During this process the bacteria convert the lactose present in milk, into lactic acid, CH3CHOHCOOH. Lactic acid is a weak, monoprotic, o ...
Hadronic Chemistry and Binding Energies
... equal, they cause repulsion. However, since the coupling is in singlet, the magnetic properties are opposite, thus implying an attraction. The calculations have shown that magnetostatic attractions are equal to the electrostastic repulsions at a mutual distance of the order of 1fm, while it becomes ...
... equal, they cause repulsion. However, since the coupling is in singlet, the magnetic properties are opposite, thus implying an attraction. The calculations have shown that magnetostatic attractions are equal to the electrostastic repulsions at a mutual distance of the order of 1fm, while it becomes ...
Chemical-Principles-7th-Edition-Zumdahl-Test-Bank
... KEY: general chemistry | early atomic theory | atomic theory of matter | isotope 8. The element rhenium (Re) exists as two stable isotopes and 18 unstable isotopes. Rhenium-185 has in its nucleus A) 75 protons, 110 neutrons. B) 75 protons, 75 neutrons. C) 75 protons, 130 neutrons. D) 130 protons, 75 ...
... KEY: general chemistry | early atomic theory | atomic theory of matter | isotope 8. The element rhenium (Re) exists as two stable isotopes and 18 unstable isotopes. Rhenium-185 has in its nucleus A) 75 protons, 110 neutrons. B) 75 protons, 75 neutrons. C) 75 protons, 130 neutrons. D) 130 protons, 75 ...
PHYSICAL SETTING CHEMISTRY
... 67 A spectral line in the infrared region of the spectrum of hydrogen has a frequency of 2.3 × 1014 hertz. Using your graph, estimate the energy associated with this spectral line. [1] 68 Explain, in terms of subatomic particles and energy states, why light is emitted by the hydrogen gas. [1] 69 Ide ...
... 67 A spectral line in the infrared region of the spectrum of hydrogen has a frequency of 2.3 × 1014 hertz. Using your graph, estimate the energy associated with this spectral line. [1] 68 Explain, in terms of subatomic particles and energy states, why light is emitted by the hydrogen gas. [1] 69 Ide ...
Many Chemistries Could Be Used to Build Living Systems
... changed. This leads to the requirement of a general method for generating a wide range of arbitrary chemicals, and not generating many others that are not wanted. This points to the absolute requirement for a battery of specific catalysts. (I assume throughout that life is chemical: Nonchemical life ...
... changed. This leads to the requirement of a general method for generating a wide range of arbitrary chemicals, and not generating many others that are not wanted. This points to the absolute requirement for a battery of specific catalysts. (I assume throughout that life is chemical: Nonchemical life ...
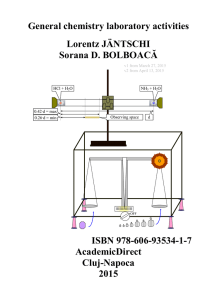
General chemistry laboratory activities, Lorentz
... Laboratory flasks are vessels (containers) which in laboratory and other scientific settings, they are usually referred to simply as flasks. Flasks come in a number of shapes and a wide range of sizes which are specified by the volume they can hold, typically in metric units such as millilitres (mL) ...
... Laboratory flasks are vessels (containers) which in laboratory and other scientific settings, they are usually referred to simply as flasks. Flasks come in a number of shapes and a wide range of sizes which are specified by the volume they can hold, typically in metric units such as millilitres (mL) ...
AP Chemistry: Course Introduction Sheet
... The particle that defines the identity of an atom is the _____________ Every hydrogen atom has ___ proton. Every magnesium atom has ___ protons. Any atom that has 23 protons is _________________. Any atom that has 92 protons is _________________. The mass of an atom is mostly from the ___________ an ...
... The particle that defines the identity of an atom is the _____________ Every hydrogen atom has ___ proton. Every magnesium atom has ___ protons. Any atom that has 23 protons is _________________. Any atom that has 92 protons is _________________. The mass of an atom is mostly from the ___________ an ...
Chem 12 Prov Exam PLO Review
... • reactions are the result of collisions between reactant particles • not all collisions are successful • sufficient kinetic energy (KE) and favourable geometry are required • to increase the rate of a reaction one must increase the frequency of successful collisions • energy changes are involved in ...
... • reactions are the result of collisions between reactant particles • not all collisions are successful • sufficient kinetic energy (KE) and favourable geometry are required • to increase the rate of a reaction one must increase the frequency of successful collisions • energy changes are involved in ...
Determination of Cystein and Methionine by Oscillating Chemical
... them is a fascinating subject in itself. In addition, this understanding facilitates the further design of new chemical oscillators. A successful mechanistic study also contributes to the knowledge of the general chemistry and kinetics of the chemical species that are involved. In this study conduct ...
... them is a fascinating subject in itself. In addition, this understanding facilitates the further design of new chemical oscillators. A successful mechanistic study also contributes to the knowledge of the general chemistry and kinetics of the chemical species that are involved. In this study conduct ...
day_3_main_lecture - the Essentially Science Wiki!
... 6.7 x 1025 molecules Or 111 mol meaning 111 moles of water molecules. This is a much more manageable number. ...
... 6.7 x 1025 molecules Or 111 mol meaning 111 moles of water molecules. This is a much more manageable number. ...
File
... Which of the following best describes the variation of the electronegativity of the elements with respect to their position on the periodic table? a. Increases across a period; increases down a group. b. Increases across a period; decreases down a group. c. Decreases across a period; increases down ...
... Which of the following best describes the variation of the electronegativity of the elements with respect to their position on the periodic table? a. Increases across a period; increases down a group. b. Increases across a period; decreases down a group. c. Decreases across a period; increases down ...
formula
... They go through several examples of the types of problems I have assigned. If you cannot find my webpage, email me and I will send you the link. •AP Chemistry Boot Camp: AP Chemistry Boot Camp will run July 14th-17th. You are highly encouraged to sign up. We will mostly be covering Units 3 and 4 dur ...
... They go through several examples of the types of problems I have assigned. If you cannot find my webpage, email me and I will send you the link. •AP Chemistry Boot Camp: AP Chemistry Boot Camp will run July 14th-17th. You are highly encouraged to sign up. We will mostly be covering Units 3 and 4 dur ...
Lecture 1 and 2 Volumetric analysis Zuhair Khammas
... Titrimetric Methods: these include a large powerful group of quantitative procedures, based on measuring the amount of a reagent of known concentration that is consumed by an analyte. Volumetric titrimetry: these involve measuring the volume of standard solution of knowing concentration that is need ...
... Titrimetric Methods: these include a large powerful group of quantitative procedures, based on measuring the amount of a reagent of known concentration that is consumed by an analyte. Volumetric titrimetry: these involve measuring the volume of standard solution of knowing concentration that is need ...
CHEMISTRY 2202
... ALL MATERIALS MUST BE PASSED IN WITH THIS EXAM. Use your time wisely. Good luck! ...
... ALL MATERIALS MUST BE PASSED IN WITH THIS EXAM. Use your time wisely. Good luck! ...
Chapter 2: Atoms, Molecules and Ions
... Chapter 2: Atoms, Molecules and Ions The topics in this chapter should be review from a previous course. It is expected that you are able to review and master this material quickly and somewhat independently. ...
... Chapter 2: Atoms, Molecules and Ions The topics in this chapter should be review from a previous course. It is expected that you are able to review and master this material quickly and somewhat independently. ...
PREPARATORY PROBLEMS
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
PREPARATORY PROBLEMS (Theoretical)
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
PREPARATORY PROBLEMS (Theoretical)
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
... The natural tendency of any chemical reaction to proceed in a certain direction at constant temperature and pressure is determined by the sign of the Gibbs energy of the reaction, DG. This is the universal principle. If DG < 0, the reaction can proceed predominantly in the forward direction (a produ ...
Introduction to Qualitative Analysis
... The techniques used in qualitative analysis depend upon the material being analyzed. For inorganic compounds, qualitative analysis often involves the identification of ions present in a sample. This is the type of analysis you will be involved with for your unknown; you will be given an aqueous solu ...
... The techniques used in qualitative analysis depend upon the material being analyzed. For inorganic compounds, qualitative analysis often involves the identification of ions present in a sample. This is the type of analysis you will be involved with for your unknown; you will be given an aqueous solu ...
Problem 14. MAGNESIUM DETERMINATION
... Many chemical phenomena can be explained by physical theories. The main theory for chemistry is quantum mechanics, which gives the solid foundation for the observed chemical periodicity. One of the cornerstones of quantum mechanics is the superposition principle that says: “If a quantum system can b ...
... Many chemical phenomena can be explained by physical theories. The main theory for chemistry is quantum mechanics, which gives the solid foundation for the observed chemical periodicity. One of the cornerstones of quantum mechanics is the superposition principle that says: “If a quantum system can b ...
Module 2 Alcohols, halogenoalkanes and analysis
... Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceuticals, refrigerants, solvents and plas ...
... Throughout the centuries, chemists have synthesised new substances and investigated their properties in the search for more useful materials. In the recent past, organic chemists have developed a broad range of original and exciting materials, such as pharmaceuticals, refrigerants, solvents and plas ...
chemistry-c7-what-you-should
... nitrogen to form ammonia is a reversible reaction b. I understand how the yield of ammonia is increased by recycling unreacted hydrogen and nitrogen c. I can explain the effect of changing temperature and pressure on the yield of ammonia at equilibrium d. I understand that the gases do not stay in t ...
... nitrogen to form ammonia is a reversible reaction b. I understand how the yield of ammonia is increased by recycling unreacted hydrogen and nitrogen c. I can explain the effect of changing temperature and pressure on the yield of ammonia at equilibrium d. I understand that the gases do not stay in t ...
Analytical chemistry

Analytical chemistry is the study of the separation, identification, and quantification of the chemical components of natural and artificial materials. Qualitative analysis gives an indication of the identity of the chemical species in the sample, and quantitative analysis determines the amount of certain components in the substance. The separation of components is often performed prior to analysis.Analytical methods can be separated into classical and instrumental. Classical methods (also known as wet chemistry methods) use separations such as precipitation, extraction, and distillation and qualitative analysis by color, odor, or melting point. Classical quantitative analysis is achieved by measurement of weight or volume. Instrumental methods use an apparatus to measure physical quantities of the analyte such as light absorption, fluorescence, or conductivity. The separation of materials is accomplished using chromatography, electrophoresis or field flow fractionation methods.Analytical chemistry is also focused on improvements in experimental design, chemometrics, and the creation of new measurement tools to provide better chemical information. Analytical chemistry has applications in forensics, bioanalysis, clinical analysis, environmental analysis, and materials analysis.