Chemical Reaction Equations
... 2) Dissociate all high-solubility ionic compounds, and ionize all strong acids to show the complete ionic equation 3) Cancel identical entities that appear on both the reactant and ...
... 2) Dissociate all high-solubility ionic compounds, and ionize all strong acids to show the complete ionic equation 3) Cancel identical entities that appear on both the reactant and ...
Chemistry Atoms, Molecules, and Ions 2.1 Multiple
... B) carbon C) hydrogen D) mercury Answer: C Topic: Section 2.9 Nuclear Stability 75) As the atomic number of the elements increases, the ratio of neutrons to protons in stable nuclei A) decreases. B) stays the same. C) increases. D) is unrelated to stability. Answer: C Topic: Section 2.9 Nuclear Stab ...
... B) carbon C) hydrogen D) mercury Answer: C Topic: Section 2.9 Nuclear Stability 75) As the atomic number of the elements increases, the ratio of neutrons to protons in stable nuclei A) decreases. B) stays the same. C) increases. D) is unrelated to stability. Answer: C Topic: Section 2.9 Nuclear Stab ...
Stoichiometery
... A chemical equation is a recipe for making a molecule. This can be written in “shopping list” format: H2 + O2 → H2O But this doesn’t help with specific amounts ...
... A chemical equation is a recipe for making a molecule. This can be written in “shopping list” format: H2 + O2 → H2O But this doesn’t help with specific amounts ...
Answers
... the empirical formula, which shows only the simplest whole number ratio of one atom to another. It conveys the least information about a molecule. ...
... the empirical formula, which shows only the simplest whole number ratio of one atom to another. It conveys the least information about a molecule. ...
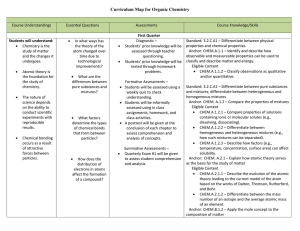
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... Chemistry is the study of matter and the changes it undergoes. ...
... Chemistry is the study of matter and the changes it undergoes. ...
document
... Calculate the number of moles of oxygen required to react exactly with 4.3 moles of propane, C3H8, in the above reaction 4.3 moles of C3H8 requires how many moles of O2 There is a 1:5 ratio So 4.3(1) : 4.3(5) ...
... Calculate the number of moles of oxygen required to react exactly with 4.3 moles of propane, C3H8, in the above reaction 4.3 moles of C3H8 requires how many moles of O2 There is a 1:5 ratio So 4.3(1) : 4.3(5) ...
CHEM 1411 – STUDY-GUIDE-for-TEST-2
... 40. If equal masses of O2(g) and HBr(g) are in separate containers of equal volume and temperature, which one of these statements is true? A) The pressure in the O2 container is greater than that in the HBr container. B) There are more HBr molecules than O2 molecules. C) The average velocity of the ...
... 40. If equal masses of O2(g) and HBr(g) are in separate containers of equal volume and temperature, which one of these statements is true? A) The pressure in the O2 container is greater than that in the HBr container. B) There are more HBr molecules than O2 molecules. C) The average velocity of the ...
9/11/01
... But what if we had only gotten 100. g of SO 2 with those amounts of CS2 and O2? - reaction did not go to completion - several competing reactions involving CS2 and/or O2 were going on at the same time. Theoretical yield – the amount of product we should get when a reactant is completely consumed acc ...
... But what if we had only gotten 100. g of SO 2 with those amounts of CS2 and O2? - reaction did not go to completion - several competing reactions involving CS2 and/or O2 were going on at the same time. Theoretical yield – the amount of product we should get when a reactant is completely consumed acc ...
Chemistry I Honors Semester I FINAL EXAM REVIEW Atomic
... ____ 7. Under ordinary conditions of temperature and pressure, the particles in a gas are a. closely packed. c. held in fixed positions. b. very far from one another. d. unevenly distributed. ____ 8. The solid state of matter can be described as a. having definite shape and definite volume. b. havin ...
... ____ 7. Under ordinary conditions of temperature and pressure, the particles in a gas are a. closely packed. c. held in fixed positions. b. very far from one another. d. unevenly distributed. ____ 8. The solid state of matter can be described as a. having definite shape and definite volume. b. havin ...
Chapter 3 HWsolutions (from Handout)
... nK 60.1 g K 1 mol K 1.537 mol K 39.10 g K Dividing by the smallest number of moles (1.532 mol) gives the empirical formula, KCN. ...
... nK 60.1 g K 1 mol K 1.537 mol K 39.10 g K Dividing by the smallest number of moles (1.532 mol) gives the empirical formula, KCN. ...
molar mass
... Law of Conservation of Mass (p. 118) In a chemical reaction, the total mass of reactants always equals the total mass of products. eg. 2 Na3N → 6 Na + N2 When 500.00 g of Na3N decomposes 323.20 g of N2 is produced. How much Na is produced in this decomposition? 2:26 AM ...
... Law of Conservation of Mass (p. 118) In a chemical reaction, the total mass of reactants always equals the total mass of products. eg. 2 Na3N → 6 Na + N2 When 500.00 g of Na3N decomposes 323.20 g of N2 is produced. How much Na is produced in this decomposition? 2:26 AM ...
5. Homework 5-Answers
... E) release CO2. C) expand. Ans: A 8. Copper metal has a specific heat of 0.385 J/g·°C. Calculate the amount of heat required to raise the temperature of 22.8 g of Cu from 20.0°C to 875°C. A) 1.97 10–5 J B) 1.0 10–2 J C) 329 J D) 7.51 kJ E) 10.5 kJ Ans: D 9. Calculate the amount of heat necessary ...
... E) release CO2. C) expand. Ans: A 8. Copper metal has a specific heat of 0.385 J/g·°C. Calculate the amount of heat required to raise the temperature of 22.8 g of Cu from 20.0°C to 875°C. A) 1.97 10–5 J B) 1.0 10–2 J C) 329 J D) 7.51 kJ E) 10.5 kJ Ans: D 9. Calculate the amount of heat necessary ...
Rubidium
... This element is considered to be the 16th most abundant element in the earth's crust. It occurs naturally in the minerals leucite, pollucite, and zinnwaldite, which contains traces of up to 1% of its oxide. Lepidolite contains 1.5% rubidium and this is the commercial source of the element. Some pota ...
... This element is considered to be the 16th most abundant element in the earth's crust. It occurs naturally in the minerals leucite, pollucite, and zinnwaldite, which contains traces of up to 1% of its oxide. Lepidolite contains 1.5% rubidium and this is the commercial source of the element. Some pota ...
Chapter 1 Introduction to Forensic Chemistry
... Many professions use the scientific method in order to solve a problem by gathering information, forming a hypothesis, and then testing it. ...
... Many professions use the scientific method in order to solve a problem by gathering information, forming a hypothesis, and then testing it. ...
Stoichiometry
... 6. Compound X2Y is 60% X by mass. Calculate the percent Y by mass of the compound XY3? If you have 100 g of X2Y there would be 60g of X and 40g of Y For XY3 since it has only one X atom you can think of that as ½(60g) or 30 g of X and since there’s three Y atoms you can think of that as 3(40g) or 12 ...
... 6. Compound X2Y is 60% X by mass. Calculate the percent Y by mass of the compound XY3? If you have 100 g of X2Y there would be 60g of X and 40g of Y For XY3 since it has only one X atom you can think of that as ½(60g) or 30 g of X and since there’s three Y atoms you can think of that as 3(40g) or 12 ...
Chapter 14
... Based on low temperature experiments, it appears that the entropy of every pure substance approaches the same value as T 0. K. Third law of thermodynamics: The absolute entropy (S) of a perfect crystal of any pure substance at absolute zero is 0.0 J/mol.K. Because there are standard ways of find ...
... Based on low temperature experiments, it appears that the entropy of every pure substance approaches the same value as T 0. K. Third law of thermodynamics: The absolute entropy (S) of a perfect crystal of any pure substance at absolute zero is 0.0 J/mol.K. Because there are standard ways of find ...
Final Exam
... long, 2.50 cm wide, and 1.00 cm thick. What is a possible identity of the element? a. nickel, 8.90 g/cm3 b. aluminum, 2.70 g/cm3 c. titanium, 4.54 g/cm3 d. zirconium, 6.51 g/cm3 e. chromium, 7.20 g/cm3 ...
... long, 2.50 cm wide, and 1.00 cm thick. What is a possible identity of the element? a. nickel, 8.90 g/cm3 b. aluminum, 2.70 g/cm3 c. titanium, 4.54 g/cm3 d. zirconium, 6.51 g/cm3 e. chromium, 7.20 g/cm3 ...
File
... less than what was expected. They identify and list the most likely errors in the activity. Which is a systematic error? A. B. C. D. ...
... less than what was expected. They identify and list the most likely errors in the activity. Which is a systematic error? A. B. C. D. ...
29 Sept 08 - Seattle Central
... Reactants and products must occur in numbers that give the same number of each type of atom on both sides of the arrow. ...
... Reactants and products must occur in numbers that give the same number of each type of atom on both sides of the arrow. ...
Exam Review
... 18. As the atomic number in a period increases, the degree of nonmetallic character __. a) increases c) increases then decreases b) decreases d) remains the same Chemistry 11 Exam Review – Answers - Page 2 ...
... 18. As the atomic number in a period increases, the degree of nonmetallic character __. a) increases c) increases then decreases b) decreases d) remains the same Chemistry 11 Exam Review – Answers - Page 2 ...
Introduction to Chemistry
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
Chapter 3:Mass Relationships in Chemical Reactions
... H2 (g) + Cl2 (g) HCl (g) • Notice the subscript for H and Cl is 2, therefore we have 2 atoms of each substance. In the products, we have HCl, 1 atom of each. We can balance the equation by putting a 2 in front HCl. H2 (g) + Cl2(g) 2 HCl (g) ...
... H2 (g) + Cl2 (g) HCl (g) • Notice the subscript for H and Cl is 2, therefore we have 2 atoms of each substance. In the products, we have HCl, 1 atom of each. We can balance the equation by putting a 2 in front HCl. H2 (g) + Cl2(g) 2 HCl (g) ...
g - mrnicholsscience
... yield is the lesser of the two. • The limiting reactant gives this answer • The excess (XS) reactant is left over. (How much?) ...
... yield is the lesser of the two. • The limiting reactant gives this answer • The excess (XS) reactant is left over. (How much?) ...
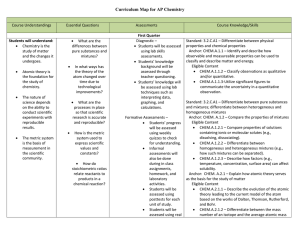
AP Chemistry Curriculum Map - Belle Vernon Area School District
... Dalton (atomic theory), Thomson (the electron), Rutherford (the nucleus), and Bohr (planetary model of the atom) and understand how each discovery leads to modern theory. Anchor: CHEM.A.1.1 Identify and describe how observable and measurable properties can be used to classify and describe matter and ...
... Dalton (atomic theory), Thomson (the electron), Rutherford (the nucleus), and Bohr (planetary model of the atom) and understand how each discovery leads to modern theory. Anchor: CHEM.A.1.1 Identify and describe how observable and measurable properties can be used to classify and describe matter and ...