Support Material
... Law of Multiple Proportions (John Dalton) : When two elements combine to form two or more compounds, then the different masses of one element, which combine with a ®xed mass of the other, bear a simple ratio to one another. Gay Lussac’s Law : When gases combine or are produced in a chemical reac ...
... Law of Multiple Proportions (John Dalton) : When two elements combine to form two or more compounds, then the different masses of one element, which combine with a ®xed mass of the other, bear a simple ratio to one another. Gay Lussac’s Law : When gases combine or are produced in a chemical reac ...
The Coordination Chemistry of Solvated Metal Ions in DMPU
... iron(II), iron(III), zinc(II), cadmium(II), and lanthanoid(III) ions. These studies have shown that the solvation process in DMPU is sometimes very different to those in corresponding aqueous systems. This is due to the the space-demanding properties the DMPU molecule has when coordinating to metal ...
... iron(II), iron(III), zinc(II), cadmium(II), and lanthanoid(III) ions. These studies have shown that the solvation process in DMPU is sometimes very different to those in corresponding aqueous systems. This is due to the the space-demanding properties the DMPU molecule has when coordinating to metal ...
Atoms and Elements
... The first people to propose that matter was composed of small, indestructible particles were Leucippus (fifth century B.C., exact dates unknown) and his student Democritus (460–370 B.C.). These Greek philosophers theorized that matter was ultimately composed of small, indivisible particles called at ...
... The first people to propose that matter was composed of small, indestructible particles were Leucippus (fifth century B.C., exact dates unknown) and his student Democritus (460–370 B.C.). These Greek philosophers theorized that matter was ultimately composed of small, indivisible particles called at ...
2 Atoms and Molecules
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
... In Chapter 1, we defined elements as homogeneous pure substances made up of identical atoms. At least 115 different elements are known to exist. This leads to the conclusion that a minimum of 115 different kinds of atoms exist. Eighty-eight of the elements are naturally occurring and therefore are f ...
9.2 Oxidation Numbers
... When fluorine atoms are combined with atoms of other elements, their oxidation number is ‒1. When oxygen atoms are combined with atoms of other elements, their oxidation number is ‒2, except in peroxides, such as hydrogen peroxide, H2O2, where their oxidation number is ‒1. (There are other exception ...
... When fluorine atoms are combined with atoms of other elements, their oxidation number is ‒1. When oxygen atoms are combined with atoms of other elements, their oxidation number is ‒2, except in peroxides, such as hydrogen peroxide, H2O2, where their oxidation number is ‒1. (There are other exception ...
11 myp covalent bonding
... energetically unstable and therefore they react with other elements to be stable. That atoms of elements, apart from the noble gases, try to attain a stable state by – either losing electrons, or – gaining electrons, or – sharing electrons. • so that they have a completely filled valence (outer) she ...
... energetically unstable and therefore they react with other elements to be stable. That atoms of elements, apart from the noble gases, try to attain a stable state by – either losing electrons, or – gaining electrons, or – sharing electrons. • so that they have a completely filled valence (outer) she ...
Calculations with Chemical Formulas and Equations
... The trick: • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
... The trick: • By definition, these are the mass of 1 mol of a substance (i.e., g/mol) – The molar mass of an element is the mass number for the element that we find on the periodic table – The formula weight (in amu’s) will be the same number as the molar mass (in g/mol) Stoichiometry ...
Stoichiometry notes 1
... 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the mole ratio from the balanced equation. 5. Convert moles of target substance to the ...
... 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the mole ratio from the balanced equation. 5. Convert moles of target substance to the ...
Final Exam - KFUPM Faculty List
... Se is in representative group VI, thus it has 6 s,p valence electrons ns2np4 It is in period 4, thus n = 4 and (n-1)d is filled: ns2(n-1)d10np4 The next smaller noble gas to Se is Ar, thus Se has an [Ar] core: [Ar]4s23d104p4 Sec# 7-11 Grade# 65 Q18. Given the following 1st, 2nd, and 3rd ionization e ...
... Se is in representative group VI, thus it has 6 s,p valence electrons ns2np4 It is in period 4, thus n = 4 and (n-1)d is filled: ns2(n-1)d10np4 The next smaller noble gas to Se is Ar, thus Se has an [Ar] core: [Ar]4s23d104p4 Sec# 7-11 Grade# 65 Q18. Given the following 1st, 2nd, and 3rd ionization e ...
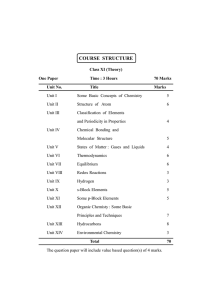
COURSE STRUCTURE
... de Broglie concept as well as uncertainty principle have no significance in everyday life because they have significance only for microscopic particles but we come across macroscopic bodies in everyday life. Quantum numbers The four quantum numbers provide the following informations : (1) Principal ...
... de Broglie concept as well as uncertainty principle have no significance in everyday life because they have significance only for microscopic particles but we come across macroscopic bodies in everyday life. Quantum numbers The four quantum numbers provide the following informations : (1) Principal ...
Disproportionation of Gold(II)
... showed this conclusion could be extended to neutral, threecoordinate AuI complexes of the type Au(phosphine)2X (X ) univalent anion, e.g., chloride, bromide or iodide). The results of these theoretical predictions have been utilized by the experimental group of Omary to design series of novel lumine ...
... showed this conclusion could be extended to neutral, threecoordinate AuI complexes of the type Au(phosphine)2X (X ) univalent anion, e.g., chloride, bromide or iodide). The results of these theoretical predictions have been utilized by the experimental group of Omary to design series of novel lumine ...
Chapter 2 Matter and Components F11 110pt
... 1. Some compounds have been known and used for so long that their trivial (or common names) have become accepted by the IUPAC as official: ...
... 1. Some compounds have been known and used for so long that their trivial (or common names) have become accepted by the IUPAC as official: ...
Chapter 2 Matter and Components F11 110
... is found in a fixed amount in nature, and rarely are these amounts equal among the given isotopes of an element we must have a way to take this into account when talking about a naturally occurring element; enter Average Mass: ...
... is found in a fixed amount in nature, and rarely are these amounts equal among the given isotopes of an element we must have a way to take this into account when talking about a naturally occurring element; enter Average Mass: ...
FREE Sample Here
... B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einstein. D) Dalton, and widely accepted within a few decades. Answer: A Topic: Section 2.1 Conservation of Mass and the Law of Definite Proportions 2) The observation that 15.0 g of ...
... B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einstein. D) Dalton, and widely accepted within a few decades. Answer: A Topic: Section 2.1 Conservation of Mass and the Law of Definite Proportions 2) The observation that 15.0 g of ...
apch04 test review_ans
... 2 AgNO3 (aq) + CaCl2 (aq) 2 AgCl (s) + Ca(NO3)2 (aq) Need to calculate moles of CaCl2 to determine the molarity (moles CaCl2 per L solution). Use stoichiometry mass to mole calculation. 0.9256 g AgCl ...
... 2 AgNO3 (aq) + CaCl2 (aq) 2 AgCl (s) + Ca(NO3)2 (aq) Need to calculate moles of CaCl2 to determine the molarity (moles CaCl2 per L solution). Use stoichiometry mass to mole calculation. 0.9256 g AgCl ...
General Chemistry: Atoms First (McMurry/Fay/Pribush)
... B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einstein. D) Dalton, and widely accepted within a few decades. Answer: A Topic: Section 2.1 Conservation of Mass and the Law of Definite Proportions 2) The observation that 15.0 g of ...
... B) Dalton, but not widely accepted until the work of Mendeleev. C) Dalton, but not widely accepted until the work of Einstein. D) Dalton, and widely accepted within a few decades. Answer: A Topic: Section 2.1 Conservation of Mass and the Law of Definite Proportions 2) The observation that 15.0 g of ...
Atomic Models 100
... Answer = Helps compare elements similarities and differences Helps explain how elements will combine with other elements to make compounds. ...
... Answer = Helps compare elements similarities and differences Helps explain how elements will combine with other elements to make compounds. ...
Chapter 1
... b. one- or two-letters derived from the element’s name with both letters always capitalized. c. the first two letters of the element’s name with both letters capitalized. d. one- or two-letters derived from the element’s name with the first letter capitalized and the second in lower case. Return ...
... b. one- or two-letters derived from the element’s name with both letters always capitalized. c. the first two letters of the element’s name with both letters capitalized. d. one- or two-letters derived from the element’s name with the first letter capitalized and the second in lower case. Return ...
Unit 1 Section 4 - Atomic Structure PPT
... • In 1900 a physcist named Max Planck proposed that matter does not emit electromagnetic energy continuously. • Max Planck suggested that the object emits energy in small specific amounts called quanta. • Quantum - the minimum quantity of energy that can be lost or gained by an atom. ...
... • In 1900 a physcist named Max Planck proposed that matter does not emit electromagnetic energy continuously. • Max Planck suggested that the object emits energy in small specific amounts called quanta. • Quantum - the minimum quantity of energy that can be lost or gained by an atom. ...
Review Study Guide for the Final
... What is it called when you have more electrons than protons? ...
... What is it called when you have more electrons than protons? ...
class notes 4
... solid when the reactants switch partners. Must have a solid precipitate form or it won’t go. b. Acid-Base Reaction: An acid and a base are mixed and we get water and a salt, when the acid and base switch partners. Acid-base reactions always go. c. Gas-Evolution Reaction: Two aqueous solutions are mi ...
... solid when the reactants switch partners. Must have a solid precipitate form or it won’t go. b. Acid-Base Reaction: An acid and a base are mixed and we get water and a salt, when the acid and base switch partners. Acid-base reactions always go. c. Gas-Evolution Reaction: Two aqueous solutions are mi ...
No Slide Title
... 1. What criteria did you use for grouping the magazines? 2. Once you arrange the magazines into groups, could you sort the material further to make it even more organized? ...
... 1. What criteria did you use for grouping the magazines? 2. Once you arrange the magazines into groups, could you sort the material further to make it even more organized? ...
Chem I Review Part 2
... 27. Which of these choices is the electron configuration of an excited state of an oxygen atom? A. 1s22s22p4 B. 1s22s22p5 C. 1s22s22p33s1 D. 1s22s22p6 E. 1s22s22p3 28. Which of these ground-state atoms is diamagnetic? A. Ca B. As C. Cu D. Fe E. none of these 29. The nineteenth century chemists arran ...
... 27. Which of these choices is the electron configuration of an excited state of an oxygen atom? A. 1s22s22p4 B. 1s22s22p5 C. 1s22s22p33s1 D. 1s22s22p6 E. 1s22s22p3 28. Which of these ground-state atoms is diamagnetic? A. Ca B. As C. Cu D. Fe E. none of these 29. The nineteenth century chemists arran ...
Stoichiometry1
... Not whole numbers No mass unit Also applies to relative molecular mass (Mr) ...
... Not whole numbers No mass unit Also applies to relative molecular mass (Mr) ...
Student Study Guide 1999
... The authors still consider this document to be a working document. They would, therefore, ask that, in exchange for using this Guide, teachers provide feedback. Please use the attached evaluation form to make corrections of suggestions. Special thanks to Jill Butler of the Lester B. Pearson School B ...
... The authors still consider this document to be a working document. They would, therefore, ask that, in exchange for using this Guide, teachers provide feedback. Please use the attached evaluation form to make corrections of suggestions. Special thanks to Jill Butler of the Lester B. Pearson School B ...