File
... (Bosch Proces, Lane’s Process, Electrolysis of water, None of these) 22. When steam is passed over iron at 500°C, magnetic oxide and hydrogen gas is produced. This is called __________. (Bosch Process, Lane’s Process, Electrolysis, All of these) 23. The presence of an acid or base renders water ___ ...
... (Bosch Proces, Lane’s Process, Electrolysis of water, None of these) 22. When steam is passed over iron at 500°C, magnetic oxide and hydrogen gas is produced. This is called __________. (Bosch Process, Lane’s Process, Electrolysis, All of these) 23. The presence of an acid or base renders water ___ ...
STOICHIOMETRY via ChemLog - Small
... Determining the mass percent composition of a compound refers to the proportion of one element expressed as a percentage of the total mass of the compound. Knowing the mass percent composition of a compound can help determine environmental effects from that compound. For example, carbon dioxide (CO2 ...
... Determining the mass percent composition of a compound refers to the proportion of one element expressed as a percentage of the total mass of the compound. Knowing the mass percent composition of a compound can help determine environmental effects from that compound. For example, carbon dioxide (CO2 ...
Chapter 1 – Reaction Kinetics Answer Key
... during a chemical reaction. The amount (number of moles) certainly does change. However, we must realize that as more moles of solid or liquid are formed, the volume of the solid or liquid al ...
... during a chemical reaction. The amount (number of moles) certainly does change. However, we must realize that as more moles of solid or liquid are formed, the volume of the solid or liquid al ...
2 - OnCourse
... Mass of NH3 is determined by calculating mass from moles using molar mass as before. ...
... Mass of NH3 is determined by calculating mass from moles using molar mass as before. ...
08272012BC Science Chem 12 Chapter 1 Answer Key
... significant than the pressure as we are very close to sea level. This means molar volume is likely a bit smaller than 24.5 L/mol (or 26.5 L/mol for trial two). Dividing by a smaller number would lead to a LARGER MASS of zinc expected. 1.2 Review Questions (page 25) 1. Factors affecting heterogeneous ...
... significant than the pressure as we are very close to sea level. This means molar volume is likely a bit smaller than 24.5 L/mol (or 26.5 L/mol for trial two). Dividing by a smaller number would lead to a LARGER MASS of zinc expected. 1.2 Review Questions (page 25) 1. Factors affecting heterogeneous ...
Chem Course Desc2. New
... Students in Chemistry will progress toward attainment of the district ESLR's while completing the course work required for this class. Self-directed Learner- Students in chemistry will recognize when they have not mastered a concept and get the appropriate help to master it. This skill is essential ...
... Students in Chemistry will progress toward attainment of the district ESLR's while completing the course work required for this class. Self-directed Learner- Students in chemistry will recognize when they have not mastered a concept and get the appropriate help to master it. This skill is essential ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... • Interpretation: ratio of number of moles of reactant required to give the ratio of number of moles of product. • These ratios are called stoichiometric ratios. NB: Stoichiometric ratios are ideal proportions • Real ratios of reactants and products in the laboratory need to be measured (in grams an ...
... • Interpretation: ratio of number of moles of reactant required to give the ratio of number of moles of product. • These ratios are called stoichiometric ratios. NB: Stoichiometric ratios are ideal proportions • Real ratios of reactants and products in the laboratory need to be measured (in grams an ...
Chemistry 11 Final Examination Review
... 31. A certain atom contains 34 protons, 34 electrons, and 45 neutrons. This atom has a mass number of __. a) 34 b) 45 c) 68 d) 79 32. An example of a compound is a) oxygen b) mercury c) salt d) diamond 33. Carbon is classed as an element rather than as a compound because it a) cannot be chemically d ...
... 31. A certain atom contains 34 protons, 34 electrons, and 45 neutrons. This atom has a mass number of __. a) 34 b) 45 c) 68 d) 79 32. An example of a compound is a) oxygen b) mercury c) salt d) diamond 33. Carbon is classed as an element rather than as a compound because it a) cannot be chemically d ...
The Mole Concept A. Atomic Masses and Avogadro`s Hypothesis 1
... Dalton assumed that elements formed simple compounds in a 1:1 ratio and he assigned an ARBITRARY MASS to each element. Since hydrogen was the lightest element, it was assigned a mass of “1” and all other elements were compared to that of hydrogen. Carbon was found to be 6 times heavier than hydrogen ...
... Dalton assumed that elements formed simple compounds in a 1:1 ratio and he assigned an ARBITRARY MASS to each element. Since hydrogen was the lightest element, it was assigned a mass of “1” and all other elements were compared to that of hydrogen. Carbon was found to be 6 times heavier than hydrogen ...
48th CHEMISTRY OLYMPIAD CHEMISTRY
... formulas to write the equation of the reaction of hydrolysis with sodium hydroxide. b) Write and substantiate the structural formulas of the compounds A and B. c) Using structural formulas write i) the scheme of the reaction A → B and ii) equation for the reaction A + B → C. d) Determine the oxidati ...
... formulas to write the equation of the reaction of hydrolysis with sodium hydroxide. b) Write and substantiate the structural formulas of the compounds A and B. c) Using structural formulas write i) the scheme of the reaction A → B and ii) equation for the reaction A + B → C. d) Determine the oxidati ...
Devillez (ld2653) – Test 1 Review – Devillez – (99998)
... In Rutherford’s gold foil experiment in 1910, α (alpha) particles were fired at gold foil, and the resulting deflection of the particles were observed. Most of the α particles went through the sample undeflected, suggesting that much of the atom was empty space. But of the few α particles that were ...
... In Rutherford’s gold foil experiment in 1910, α (alpha) particles were fired at gold foil, and the resulting deflection of the particles were observed. Most of the α particles went through the sample undeflected, suggesting that much of the atom was empty space. But of the few α particles that were ...
Calculations and the Chemical Equation
... The chemical equation enables the determination of the quantity of reactants needed to produce a certain quantity of products, and expresses these quantities in terms of moles. The number of moles of each product and reactant is indicated by placing a whole-number coefficient before the formula of e ...
... The chemical equation enables the determination of the quantity of reactants needed to produce a certain quantity of products, and expresses these quantities in terms of moles. The number of moles of each product and reactant is indicated by placing a whole-number coefficient before the formula of e ...
Week 3 July 22, 2016 Worksheet Review III 1 mol = 6.022 × 1023 1
... 1.807 × 1024 atoms are present in the sample True. We know that for every 1 molecule of H2O, we have 2 atoms of H and 1 atom of O. So we every 1 molecule of H2O has 3 atoms. 3× (6.022 × 1023 molecules H 2O) = 1.807 × 1024 atoms ...
... 1.807 × 1024 atoms are present in the sample True. We know that for every 1 molecule of H2O, we have 2 atoms of H and 1 atom of O. So we every 1 molecule of H2O has 3 atoms. 3× (6.022 × 1023 molecules H 2O) = 1.807 × 1024 atoms ...
Preview Sample 2
... Antoine Lavoisier's experiments showed that the mass of the products of a chemical reaction equals the mass of the reacting substances. When wood is burned, the ashes weigh less than the original wood, but this is not a violation of the law of conservation of matter. Dalton's atomic theory says that ...
... Antoine Lavoisier's experiments showed that the mass of the products of a chemical reaction equals the mass of the reacting substances. When wood is burned, the ashes weigh less than the original wood, but this is not a violation of the law of conservation of matter. Dalton's atomic theory says that ...
- Catalyst
... the atomic masses of each element multiplied by the subscripts. Solution: The formula is Na3PO4. The molar mass is 163.94 g/mol. Calculating the moles in a 38.6 g sample: moles of Na3PO4 = 38.6 g sample 1 mol 163.94 g sample ...
... the atomic masses of each element multiplied by the subscripts. Solution: The formula is Na3PO4. The molar mass is 163.94 g/mol. Calculating the moles in a 38.6 g sample: moles of Na3PO4 = 38.6 g sample 1 mol 163.94 g sample ...
Introductory Chemistry, 2nd Edition Nivaldo Tro
... number of molecules as well as the number of moles of each substance ...
... number of molecules as well as the number of moles of each substance ...
for the exam on 14 feb
... to reach the second equivalence point, you’d need 0.00200 mol – double the first.) You’re only halfway to the second equivalence point, here. But when you’re halfway there, you’re at the half-equivalence point for the second pKa value, meaning [HA] = [A-], and you can say that pH = pKa2. Therefore, ...
... to reach the second equivalence point, you’d need 0.00200 mol – double the first.) You’re only halfway to the second equivalence point, here. But when you’re halfway there, you’re at the half-equivalence point for the second pKa value, meaning [HA] = [A-], and you can say that pH = pKa2. Therefore, ...
Changing Matter
... chemicals in the world. • Most of the world’s phosphoric acid is produced by the wet process which involves the reaction of phosphate rock, Ca5(PO4)3F, with sulfuric acid (H2SO4). Ca5(PO4)3F(s) + 5H2SO4 3H3PO4 + HF + 5CaSO4 ...
... chemicals in the world. • Most of the world’s phosphoric acid is produced by the wet process which involves the reaction of phosphate rock, Ca5(PO4)3F, with sulfuric acid (H2SO4). Ca5(PO4)3F(s) + 5H2SO4 3H3PO4 + HF + 5CaSO4 ...
Objectives - hartman
... Chlorobenzene, C6H5Cl, is used in the production of many important chemicals, such as aspirin, dyes, and disinfectants. One industrial method of preparing chlorobenzene is to react benzene, C6H6, with chlorine, as represented by the following equation. C6H6 (l) + Cl2(g) → C6H5Cl(l) + HCl(g) ...
... Chlorobenzene, C6H5Cl, is used in the production of many important chemicals, such as aspirin, dyes, and disinfectants. One industrial method of preparing chlorobenzene is to react benzene, C6H6, with chlorine, as represented by the following equation. C6H6 (l) + Cl2(g) → C6H5Cl(l) + HCl(g) ...
Name:
... changes only slightly and therefore does not offset the increase in size due to the increase in energy levels. Atomic radius decreases as you go left to right across a period in the periodic table. The valence electrons are found in orbitals of the same energy level. At the same time, the effective ...
... changes only slightly and therefore does not offset the increase in size due to the increase in energy levels. Atomic radius decreases as you go left to right across a period in the periodic table. The valence electrons are found in orbitals of the same energy level. At the same time, the effective ...
Chapter 03 - KFUPM Faculty List
... So when 10 g SO3 are produced, then all O2 is used up, the reaction stops and some S is left over: O2 is the limiting reactant, S the excess reactant. The other possible method (to calculate how much oxygen is needed to react with all the sulfur) gives the same result: ...
... So when 10 g SO3 are produced, then all O2 is used up, the reaction stops and some S is left over: O2 is the limiting reactant, S the excess reactant. The other possible method (to calculate how much oxygen is needed to react with all the sulfur) gives the same result: ...
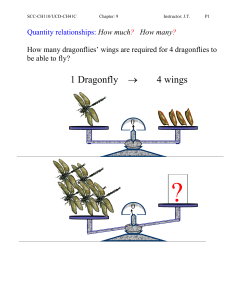
Quantity relationships: How much
... The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yield, and it is ca ...
... The calculated amount of product if is based on the assumption that all of the reactant is converted into product is called the theoretical yield. In laboratory or in industrial production, the actual amount of product isolated from a reaction is usually less than the theoretical yield, and it is ca ...
Chemistry 110 Oxidation Reduction Reactions Oxidation Number
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
... We can see that the oxidation number of C increases from -4 to +4 in this reaction, so C is oxidized. We can also see that the oxidation number of O decreases from zero (0) to -2, so O is reduced. Notice that the oxidation number of hydrogen does not change. It is always the case that if any element ...
1 General Chemistry II Jasperse Entropy, Spontaneity, and Free
... a. only that ΔSsys < 0 But if it's unfavorable, it must not be bad enough to b. only that ΔSsys > 0 win over the good enthalpy. c. both ΔSsys < 0 and the magnitude of ΔSsys < the magnitude of ΔSsurr d. both ΔSsys < 0 and the magnitude of ΔSsys > the magnitude of ΔSsurr e. either ΔSsys > 0, or else Δ ...
... a. only that ΔSsys < 0 But if it's unfavorable, it must not be bad enough to b. only that ΔSsys > 0 win over the good enthalpy. c. both ΔSsys < 0 and the magnitude of ΔSsys < the magnitude of ΔSsurr d. both ΔSsys < 0 and the magnitude of ΔSsys > the magnitude of ΔSsurr e. either ΔSsys > 0, or else Δ ...
Chemistry 211
... •How much product is formed from some given amount of reactant? (Theoretical Yield) •How much of one reactant is required to bring a reaction to completion given some amount of another reactant? (Limiting Reagent) ...
... •How much product is formed from some given amount of reactant? (Theoretical Yield) •How much of one reactant is required to bring a reaction to completion given some amount of another reactant? (Limiting Reagent) ...