AP Chemistry: Course Introduction Sheet
... State the three particles that make up an atom, their symbol, their charge, their mass, and their location. State the number of protons, neutrons, and electrons in any atom or ion. Explain that isotopes are two atoms with the same atomic number (number of protons) but different mass numbers (n ...
... State the three particles that make up an atom, their symbol, their charge, their mass, and their location. State the number of protons, neutrons, and electrons in any atom or ion. Explain that isotopes are two atoms with the same atomic number (number of protons) but different mass numbers (n ...
Document
... of electrons are available for bonding. There is only a ____________________ in successive ionization energies of the d electrons. Thus when elements of the first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons ...
... of electrons are available for bonding. There is only a ____________________ in successive ionization energies of the d electrons. Thus when elements of the first transition series react to form compounds, they can form ions of roughly the ___________________ by losing different numbers of electrons ...
Some basic concepts of chemistry
... The three basic units, i.e., units of mass, length and time are independent units and cannot be derived from any other units, hence they are called fundamental units. The three fundamental units cannot describe all the physical quantities such as temperature, intensity of luminosity, electric curren ...
... The three basic units, i.e., units of mass, length and time are independent units and cannot be derived from any other units, hence they are called fundamental units. The three fundamental units cannot describe all the physical quantities such as temperature, intensity of luminosity, electric curren ...
Chapter 4 What Are Atoms?
... • Locate semiconductors, halogens, and noble gases in the periodic table. • Relate an element’s chemical properties to the electron arrangement of its atoms. ...
... • Locate semiconductors, halogens, and noble gases in the periodic table. • Relate an element’s chemical properties to the electron arrangement of its atoms. ...
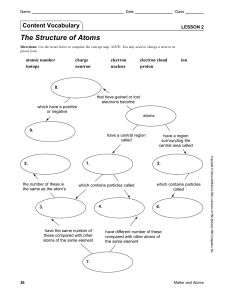
Lesson 2 | The Structure of Atoms
... 3. The region at the center of an atom that contains most of the mass of the atom is called the nucleus. a. A positively charged particle in the nucleus of an atom is called a(n) proton. b. An uncharged particle in the nucleus of an atom is called a(n) neutron. 4. A negatively charged particle that ...
... 3. The region at the center of an atom that contains most of the mass of the atom is called the nucleus. a. A positively charged particle in the nucleus of an atom is called a(n) proton. b. An uncharged particle in the nucleus of an atom is called a(n) neutron. 4. A negatively charged particle that ...
Chapter 2 – Atoms, Ions, and the Periodic Table
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
FREE Sample Here
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
Mendeleev`s Periodic Table
... peak in a given plot corresponds to the electron density in a given principal shell. Because helium has only one filled shell (n = 1), it shows only a single peak. In contrast, neon, with filled n = 1 and 2 principal shells, has two peaks. Argon, with filled n = 1, 2, and 3 principal shells, has thr ...
... peak in a given plot corresponds to the electron density in a given principal shell. Because helium has only one filled shell (n = 1), it shows only a single peak. In contrast, neon, with filled n = 1 and 2 principal shells, has two peaks. Argon, with filled n = 1, 2, and 3 principal shells, has thr ...
Chapter 2 – Atoms, Ions, and the Periodic Table
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
... chemical reactions, mass must be conserved. If elements could be changed into other elements during chemical reactions (as the alchemists were trying to do), then masses of atoms would change during reactions and mass would not be conserved. ...
NCERT SOLUTIONS STRUCTURE OF ATOM Question 1: What are
... is determined by the number of valence electrons present in the atom of that element. If the number of valence electrons of the atom of an element is less than or equal to four, then the valency of that element is equal to the number of valence electrons. For example, the atom of silicon has four va ...
... is determined by the number of valence electrons present in the atom of that element. If the number of valence electrons of the atom of an element is less than or equal to four, then the valency of that element is equal to the number of valence electrons. For example, the atom of silicon has four va ...
Possible pieces of introduction:
... to see again the earth, air, and water from which I was separated by a gulf that grew larger every day” (137). This statement alludes to three of the four fundamental elements earth, air, fire and water studied by the earliest of chemists before an understanding of the atom and the periodic table of ...
... to see again the earth, air, and water from which I was separated by a gulf that grew larger every day” (137). This statement alludes to three of the four fundamental elements earth, air, fire and water studied by the earliest of chemists before an understanding of the atom and the periodic table of ...
Chapter 2: Atoms, Molecules and Ions
... increase, decrease, or stay the same? As the mass of the particle is increased while the speed of the particles remains the same, would you expect the bending to increase, decrease, or stay the same? ...
... increase, decrease, or stay the same? As the mass of the particle is increased while the speed of the particles remains the same, would you expect the bending to increase, decrease, or stay the same? ...
1 Structure of Atom - Viva Online Learning
... Maharishi Kanada, a great Indian philosopher of 600 BC, proposed that matter consists of indestructible particles called paramanu (param means ultimate and anu means particle). A paramanu does not exist in free state, rather it combines with other paramanus to form a bigger particle called the anu. ...
... Maharishi Kanada, a great Indian philosopher of 600 BC, proposed that matter consists of indestructible particles called paramanu (param means ultimate and anu means particle). A paramanu does not exist in free state, rather it combines with other paramanus to form a bigger particle called the anu. ...
Hewitt/Lyons/Suchocki/Yeh, Conceptual Integrated Science
... – lightest and most abundant is hydrogen • to date, 115 are known – 90 occur in nature – others produced in laboratory are unstable Words atom and element can be used interchangeably Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
... – lightest and most abundant is hydrogen • to date, 115 are known – 90 occur in nature – others produced in laboratory are unstable Words atom and element can be used interchangeably Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
chapter 1–introduction to earth history
... this continuous change, or evolution, only rocks formed during the same age contain similar assemblages of fossils. Darwin also theorized about the mechanism for evolution which he called natural selection. Those individuals with the most favorable variations would have the best chance of surviving ...
... this continuous change, or evolution, only rocks formed during the same age contain similar assemblages of fossils. Darwin also theorized about the mechanism for evolution which he called natural selection. Those individuals with the most favorable variations would have the best chance of surviving ...
Chemistry - Resonance
... About 3 million organic compounds are known today. The main reasons for this huge number of organic compounds are (i) Catenation : The property of self linking of carbon atoms through covalent bonds to form long straight or branched chains and rings of different sizes is called catenation.Carbon sho ...
... About 3 million organic compounds are known today. The main reasons for this huge number of organic compounds are (i) Catenation : The property of self linking of carbon atoms through covalent bonds to form long straight or branched chains and rings of different sizes is called catenation.Carbon sho ...
Atoms and Molecules
... Each element is composed of atoms – which are incredibly small. All atoms of a given element are identical to one another in mass and other properties, and different from all other atoms. That atoms were indivisible, and were not created or destroyed in chemical reactions. When atoms of different el ...
... Each element is composed of atoms – which are incredibly small. All atoms of a given element are identical to one another in mass and other properties, and different from all other atoms. That atoms were indivisible, and were not created or destroyed in chemical reactions. When atoms of different el ...
Chapter 9 Atomic Absorption and Atomic Fluorescence Spectrometry
... Several common fuels and oxidants can be employed in flame spectroscopy depending on temperature needed. Temperatures of 1700oC to 2400oC are obtained with the various fuels when air serves as the oxidant. At these temperature, only easily decomposed samples are atomized. For more refractory samples ...
... Several common fuels and oxidants can be employed in flame spectroscopy depending on temperature needed. Temperatures of 1700oC to 2400oC are obtained with the various fuels when air serves as the oxidant. At these temperature, only easily decomposed samples are atomized. For more refractory samples ...
SOME BASIC CONCEPTS OF CHEMISTRY NOTES
... same element can have different masses also. 3. Dalton said that atoms of different elements have got different mass but during certain conditions they can have the same mass. ATOMIC AND MOLECULAR MASSES: The atomic mass of an atom is extremely small. Earlier, scientists used to determine the mass o ...
... same element can have different masses also. 3. Dalton said that atoms of different elements have got different mass but during certain conditions they can have the same mass. ATOMIC AND MOLECULAR MASSES: The atomic mass of an atom is extremely small. Earlier, scientists used to determine the mass o ...
09 - Northern Highlands
... element. Only a substance that could not be broken apart could truly be an element. As of this writing, scientists have identified 118 elements. Only 88 of these occur naturally. The others are made in laboratories. The modern As chemists worked on determining which substances were elements, they pe ...
... element. Only a substance that could not be broken apart could truly be an element. As of this writing, scientists have identified 118 elements. Only 88 of these occur naturally. The others are made in laboratories. The modern As chemists worked on determining which substances were elements, they pe ...
Dalton`s Atomic Theory
... • All substances are made of atoms. Atoms are the smallest particles of matter. They cannot be divided into smaller particles, created, or destroyed. • All atoms of the same element are alike and have the same mass. Atoms of different elements are different and have different masses. • Atoms join to ...
... • All substances are made of atoms. Atoms are the smallest particles of matter. They cannot be divided into smaller particles, created, or destroyed. • All atoms of the same element are alike and have the same mass. Atoms of different elements are different and have different masses. • Atoms join to ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
Reading 1.4 What Are The Parts Of An Atom and How Are They
... Electrons have a negative charge. As a result, they are attracted to positive objects and repelled from negative objects, including other electrons (illustrated below). To minimize repulsion, each electron is capable of staking out a “territory” and “defending” itself from other electrons. Protons a ...
... Electrons have a negative charge. As a result, they are attracted to positive objects and repelled from negative objects, including other electrons (illustrated below). To minimize repulsion, each electron is capable of staking out a “territory” and “defending” itself from other electrons. Protons a ...
Chemical element
A chemical element (or element) is a chemical substance consisting of atoms having the same number of protons in their atomic nuclei (i.e. the same atomic number, Z). There are 118 elements that have been identified, of which the first 94 occur naturally on Earth with the remaining 24 being synthetic elements. There are 80 elements that have at least one stable isotope and 38 that have exclusively radioactive isotopes, which decay over time into other elements. Iron is the most abundant element (by mass) making up the Earth, while oxygen is the most common element in the crust of the earth.Chemical elements constitute approximately 15% of the matter in the universe: the remainder is dark matter, the composition of it is unknown, but it is not composed of chemical elements.The two lightest elements, hydrogen and helium were mostly formed in the Big Bang and are the most common elements in the universe. The next three elements (lithium, beryllium and boron) were formed mostly by cosmic ray spallation, and are thus more rare than those that follow. Formation of elements with from six to twenty six protons occurred and continues to occur in main sequence stars via stellar nucleosynthesis. The high abundance of oxygen, silicon, and iron on Earth reflects their common production in such stars. Elements with greater than twenty six protons are formed by supernova nucleosynthesis in supernovae, which, when they explode, blast these elements far into space as planetary nebulae, where they may become incorporated into planets when they are formed.When different elements are chemically combined, with the atoms held together by chemical bonds, they form chemical compounds. Only a minority of elements are found uncombined as relatively pure minerals. Among the more common of such ""native elements"" are copper, silver, gold, carbon (as coal, graphite, or diamonds), and sulfur. All but a few of the most inert elements, such as noble gases and noble metals, are usually found on Earth in chemically combined form, as chemical compounds. While about 32 of the chemical elements occur on Earth in native uncombined forms, most of these occur as mixtures. For example, atmospheric air is primarily a mixture of nitrogen, oxygen, and argon, and native solid elements occur in alloys, such as that of iron and nickel.The history of the discovery and use of the elements began with primitive human societies that found native elements like carbon, sulfur, copper and gold. Later civilizations extracted elemental copper, tin, lead and iron from their ores by smelting, using charcoal. Alchemists and chemists subsequently identified many more, with almost all of the naturally-occurring elements becoming known by 1900. The properties of the chemical elements are summarized on the periodic table, which organizes the elements by increasing atomic number into rows (""periods"") in which the columns (""groups"") share recurring (""periodic"") physical and chemical properties. Save for unstable radioactive elements with short half-lives, all of the elements are available industrially, most of them in high degrees of purity.