W9e „Heat Capacity of Solids and Liquids“
... up as a voltage correct circuit. (see Fig. 1 below). In case of a voltage-correct circuit the voltage drop U is directly measured at the resistance heater using a digital multimeter in the ACV range. The current I is measured with a digital multimeter in the ACA range. The correct choice of the meas ...
... up as a voltage correct circuit. (see Fig. 1 below). In case of a voltage-correct circuit the voltage drop U is directly measured at the resistance heater using a digital multimeter in the ACV range. The current I is measured with a digital multimeter in the ACA range. The correct choice of the meas ...
Heat Transfer/ Specific Heat Problems Worksheet
... 2. How much heat is lost when a 64 g piece of copper cools from 375 oC, to 26 oC? (The specific heat of copper is 0.38452 J/g x oC). Place your answer in kJ. 3. The specific heat of iron is 0.4494 J/g x oC. How much heat is transferred when a 4.7 kg piece of iron is cooled from 180 oC to 13 oC? Reme ...
... 2. How much heat is lost when a 64 g piece of copper cools from 375 oC, to 26 oC? (The specific heat of copper is 0.38452 J/g x oC). Place your answer in kJ. 3. The specific heat of iron is 0.4494 J/g x oC. How much heat is transferred when a 4.7 kg piece of iron is cooled from 180 oC to 13 oC? Reme ...
ch5 notesheet naked outline0015
... Potential Energy Potential energy is Forms of potential energy ...
... Potential Energy Potential energy is Forms of potential energy ...
Chap19Class2
... a new metal alloy. A 0.150-kg sample of the alloy is heated to 540°C. It is then quickly placed in 0.400 kg of water at 10.0°C, which is contained in a 0.200-kg aluminum calorimeter cup. (We do not need to know the mass of the insulating jacket since we assume the air space between it and the cup in ...
... a new metal alloy. A 0.150-kg sample of the alloy is heated to 540°C. It is then quickly placed in 0.400 kg of water at 10.0°C, which is contained in a 0.200-kg aluminum calorimeter cup. (We do not need to know the mass of the insulating jacket since we assume the air space between it and the cup in ...
15.3 The First Law of Thermodynamics
... Since the volume increases, the work is positive. Estimate that there are 8.9 colored squares in the drawing. ...
... Since the volume increases, the work is positive. Estimate that there are 8.9 colored squares in the drawing. ...
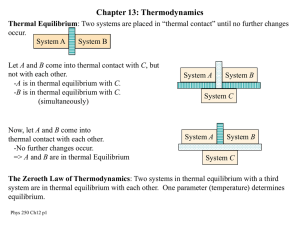
p250c13
... Example: A heat pump is used to maintain an inside temperature of 20 ºC when the outside temperature is 10ºC. What is the theoretical maximum cp for this heat pump? If the pump is to deliver heat at a rate of 15 kW, how much power must be supplied to run the pump? ...
... Example: A heat pump is used to maintain an inside temperature of 20 ºC when the outside temperature is 10ºC. What is the theoretical maximum cp for this heat pump? If the pump is to deliver heat at a rate of 15 kW, how much power must be supplied to run the pump? ...
Heat pipe
A heat pipe is a heat-transfer device that combines the principles of both thermal conductivity and phase transition to efficiently manage the transfer of heat between two solid interfaces.At the hot interface of a heat pipe a liquid in contact with a thermally conductive solid surface turns into a vapor by absorbing heat from that surface. The vapor then travels along the heat pipe to the cold interface and condenses back into a liquid - releasing the latent heat. The liquid then returns to the hot interface through either capillary action, centrifugal force, or gravity, and the cycle repeats. Due to the very high heat transfer coefficients for boiling and condensation, heat pipes are highly effective thermal conductors. The effective thermal conductivity varies with heat pipe length, and can approach 7002100000000000000♠100 kW/(m⋅K) for long heat pipes, in comparison with approximately 6999400000000000000♠0.4 kW/(m⋅K) for copper.