Implimenting a Simple Heat Exchanger Unit with
... the 80 °F air temperature, meaning that there is actually a negative temperature difference. This causes the system to operate at peak efficiency until a water temperature of 80 °F is reached. At this point a temperature difference starts to build up, reducing the cooling potential of the system. Th ...
... the 80 °F air temperature, meaning that there is actually a negative temperature difference. This causes the system to operate at peak efficiency until a water temperature of 80 °F is reached. At this point a temperature difference starts to build up, reducing the cooling potential of the system. Th ...
Radiant Barrier Training 7-2013 - Fi-Foil
... to reach 160 to 190 degrees Fahrenheit. Heat is conducted through roofing materials and reradiated to the mass insulation and ceiling below. Attic air temperatures climb to superheated levels…typically 140 degrees Fahrenheit in the summer. Radiant heat transfers into air conditioning ducts increasin ...
... to reach 160 to 190 degrees Fahrenheit. Heat is conducted through roofing materials and reradiated to the mass insulation and ceiling below. Attic air temperatures climb to superheated levels…typically 140 degrees Fahrenheit in the summer. Radiant heat transfers into air conditioning ducts increasin ...
Thermodynamic functions - Phase Transformations Group
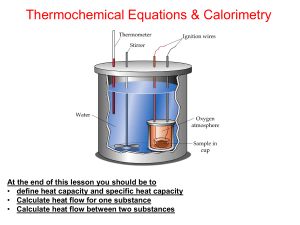
... functions of state. More About the Heat Capacity The heat capacity can be determined experimentally using calorimetry. The data can then be related directly to the functions of state H, G and S. The heat capacity varies with temperature and other factors and hence is important in determining the sta ...
... functions of state. More About the Heat Capacity The heat capacity can be determined experimentally using calorimetry. The data can then be related directly to the functions of state H, G and S. The heat capacity varies with temperature and other factors and hence is important in determining the sta ...
- Uponorpro.com
... the strategies used in forcedair systems are not necessarily applicable for radiant systems. The way in which energy is evaluated and managed is on a more finite level with radiant systems. The temperature in one room will not impact the temperature in the next room. This is why it is easier and les ...
... the strategies used in forcedair systems are not necessarily applicable for radiant systems. The way in which energy is evaluated and managed is on a more finite level with radiant systems. The temperature in one room will not impact the temperature in the next room. This is why it is easier and les ...
Set 3
... This is because in almost all cases, reverseable processes do something to change the macroscopic configuration of the system. This in general can be undone. Consider the compression of an ideal gas by a piston. Of the system is adiabatically isolated, then by “easing up” on the piston, I allow the ...
... This is because in almost all cases, reverseable processes do something to change the macroscopic configuration of the system. This in general can be undone. Consider the compression of an ideal gas by a piston. Of the system is adiabatically isolated, then by “easing up” on the piston, I allow the ...
Chapter 18
... Heat of vaporization LV: For liquid vaporizing to gas, or gas condenses to liquid. water, LV = 539 cal/g = 2256 kJ/kg Heat of fusion LF: For solid melting to liquid, or liquid freezing to solid. water, LF = 79.5 cal/g = 333 kJ/kg ...
... Heat of vaporization LV: For liquid vaporizing to gas, or gas condenses to liquid. water, LV = 539 cal/g = 2256 kJ/kg Heat of fusion LF: For solid melting to liquid, or liquid freezing to solid. water, LF = 79.5 cal/g = 333 kJ/kg ...
Unit 4: Themodynamics
... The overall heat of a reaction (ΔH ) is the sum of the ΔHs of each step in the process Can obtain “heat of formation” (ΔH) data for compounds from tables Formation of 1 mole of compounds from their elements ...
... The overall heat of a reaction (ΔH ) is the sum of the ΔHs of each step in the process Can obtain “heat of formation” (ΔH) data for compounds from tables Formation of 1 mole of compounds from their elements ...
Basic Thermodynamics Goals The ideal gas Entropy, Heat and Work
... Calculate the entropy changes in the system consisting of block plus baths in cooling from 200K to 100K in these two cases. Prove that in the limit of an infinite number of intermediate baths the total entropy change is zero. 6. Two identical bodies of constant heat capacity Cp at temperatures T1 an ...
... Calculate the entropy changes in the system consisting of block plus baths in cooling from 200K to 100K in these two cases. Prove that in the limit of an infinite number of intermediate baths the total entropy change is zero. 6. Two identical bodies of constant heat capacity Cp at temperatures T1 an ...
Heat pipe
A heat pipe is a heat-transfer device that combines the principles of both thermal conductivity and phase transition to efficiently manage the transfer of heat between two solid interfaces.At the hot interface of a heat pipe a liquid in contact with a thermally conductive solid surface turns into a vapor by absorbing heat from that surface. The vapor then travels along the heat pipe to the cold interface and condenses back into a liquid - releasing the latent heat. The liquid then returns to the hot interface through either capillary action, centrifugal force, or gravity, and the cycle repeats. Due to the very high heat transfer coefficients for boiling and condensation, heat pipes are highly effective thermal conductors. The effective thermal conductivity varies with heat pipe length, and can approach 7002100000000000000♠100 kW/(m⋅K) for long heat pipes, in comparison with approximately 6999400000000000000♠0.4 kW/(m⋅K) for copper.










![科目名 Course Title Thermal Engineering [熱工学E] 講義題目 Subtitle](http://s1.studyres.com/store/data/022970293_1-8d5861074e83e836baec8d9b5d560a01-300x300.png)