What do the numbers 238, 235 written against the name of the
... Mass of neutron = 1.00867 amu; mass of proton = 1.00728 amu; mass of electron = 0.00055 amu. The sum of the masses of all the protons, neutrons and electrons in O 16 gives 16.13200 amu. The mass of a 16O atom, however, is 15.99491 amu, relative to 12.00000 amu for 12C. This leaves a discrepancy of ...
... Mass of neutron = 1.00867 amu; mass of proton = 1.00728 amu; mass of electron = 0.00055 amu. The sum of the masses of all the protons, neutrons and electrons in O 16 gives 16.13200 amu. The mass of a 16O atom, however, is 15.99491 amu, relative to 12.00000 amu for 12C. This leaves a discrepancy of ...
1 slide per page() - Wayne State University Physics and Astronomy
... Nuclear Reactor A nuclear reactor is a system designed to maintain a self-sustained chain reaction The reproduction constant, K, is defined as the average number of neutrons from each fission ...
... Nuclear Reactor A nuclear reactor is a system designed to maintain a self-sustained chain reaction The reproduction constant, K, is defined as the average number of neutrons from each fission ...
Variant 1. 1) There are four different ways in which the various
... 1) There are four different ways in which the various particles that make up the Universe can interact with one another. Each of these is a particular variety of interaction or to use a more old-fashioned but more common term a force. These four forces are the nuclear, the electromagnetic, the weak ...
... 1) There are four different ways in which the various particles that make up the Universe can interact with one another. Each of these is a particular variety of interaction or to use a more old-fashioned but more common term a force. These four forces are the nuclear, the electromagnetic, the weak ...
The Density Matrix Renormalization Group Method for Realistic
... alternative program for doing nuclear structure calculations with the DMRG: •DMRG with sweeping in the m-scheme ...
... alternative program for doing nuclear structure calculations with the DMRG: •DMRG with sweeping in the m-scheme ...
The Big Bang, the LHC and the God Particle
... Electrons from nucleus? no p+ + e- ? But: energy, momentum missing New particle; tiny mass, zero charge ...
... Electrons from nucleus? no p+ + e- ? But: energy, momentum missing New particle; tiny mass, zero charge ...
The Weak and Strong Nuclear Interactions
... then resurfaced. Between 1900 and 1930 quantum mechanics, the theory that described microscopic phenomena, had become fairly well developed. The Dirac theory of the hydrogen atom electron (1928) led to the discovery of the positron, the antiparticle of the electron, in 1932. By the year 1932 it had ...
... then resurfaced. Between 1900 and 1930 quantum mechanics, the theory that described microscopic phenomena, had become fairly well developed. The Dirac theory of the hydrogen atom electron (1928) led to the discovery of the positron, the antiparticle of the electron, in 1932. By the year 1932 it had ...
In Search of Giants Worksheet
... a. While gravity and electromagnetic forces can account for all of the phenomenon that we encounter in our everyday world, but they cannot account for phenomenon where? ...
... a. While gravity and electromagnetic forces can account for all of the phenomenon that we encounter in our everyday world, but they cannot account for phenomenon where? ...
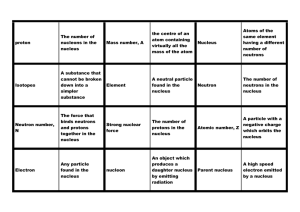
Atomic mass
... Most of the particles passed right through A few particles were deflected VERY FEW were greatly deflected “Like howitzer shells bouncing off of ...
... Most of the particles passed right through A few particles were deflected VERY FEW were greatly deflected “Like howitzer shells bouncing off of ...
STEM Fair Introduction Beanium Isotopes Lab
... Neutrons are made of one “up” quark and two “down” quarks ...
... Neutrons are made of one “up” quark and two “down” quarks ...
IB HL Physics More Problems on Quantum and Nuclear Physics_
... palladium nucleus. It then moves back along the path from which it came as shown in the diagram below. palladium nucleus -particle ...
... palladium nucleus. It then moves back along the path from which it came as shown in the diagram below. palladium nucleus -particle ...
4 slides per page() - Wayne State University Physics and
... Elementary particle interactions The scattering of two electrons via a coulomb force This virtual photon is said to mediate the electromagnetic force. The virtual photon can never be detected because it only lasts for a vanishing small time. ...
... Elementary particle interactions The scattering of two electrons via a coulomb force This virtual photon is said to mediate the electromagnetic force. The virtual photon can never be detected because it only lasts for a vanishing small time. ...
“Subatomic Physics” explores the phenomena which take place at
... phenomena we_8#/,&`O-"( need: ...
... phenomena we_8#/,&`O-"( need: ...
Heisenburg uncertainty principle
... forces both have infinite range but gravity is 1036 times weaker at a given distance The strong and weak forces are both short range forces (<10-14 m) The weak force is 108 times weaker than the strong force within a nucleus ...
... forces both have infinite range but gravity is 1036 times weaker at a given distance The strong and weak forces are both short range forces (<10-14 m) The weak force is 108 times weaker than the strong force within a nucleus ...
Lesson 15: Nuclear Quest - Highline Public Schools
... Alpha particle: A particle composed of two protons and two neutrons, equivalent to the nucleus of a helium atom. Beta particle: An electron emitted from the nucleus of an atom during beta decay. Gamma ray: A form of high-energy electromagnetic radiation emitted during nuclear reactions. ...
... Alpha particle: A particle composed of two protons and two neutrons, equivalent to the nucleus of a helium atom. Beta particle: An electron emitted from the nucleus of an atom during beta decay. Gamma ray: A form of high-energy electromagnetic radiation emitted during nuclear reactions. ...
Chapter 4 Atoms and Elements
... electrons have a negative (-) charge neutrons are neutral and have no charge neutral atoms have an equal number of protons and electrons • ions are atoms that have an unequal number of protons and electrons • an atom’s net charge = # of protons - # electrons ...
... electrons have a negative (-) charge neutrons are neutral and have no charge neutral atoms have an equal number of protons and electrons • ions are atoms that have an unequal number of protons and electrons • an atom’s net charge = # of protons - # electrons ...
University of Surrey Nuclear Physics Research Group 4 Professors
... * joint positions within AIR Division at NPL. Fundamental research based on synthesis and structure studies of exotic isotopes and evolution of nuclear structure as function of changing proton and neutron number & parameters such as angular momentum (spin) and binding energy ...
... * joint positions within AIR Division at NPL. Fundamental research based on synthesis and structure studies of exotic isotopes and evolution of nuclear structure as function of changing proton and neutron number & parameters such as angular momentum (spin) and binding energy ...
Nuclear force

The nuclear force (or nucleon–nucleon interaction or residual strong force) is the force between protons and neutrons, subatomic particles that are collectively called nucleons. The nuclear force is responsible for binding protons and neutrons into atomic nuclei. Neutrons and protons are affected by the nuclear force almost identically. Since protons have charge +1 e, they experience a Coulomb repulsion that tends to push them apart, but at short range the nuclear force is sufficiently attractive as to overcome the electromagnetic repulsive force. The mass of a nucleus is less than the sum total of the individual masses of the protons and neutrons which form it. The difference in mass between bound and unbound nucleons is known as the mass defect. Energy is released when nuclei break apart, and it is this energy that used in nuclear power and nuclear weapons.The nuclear force is powerfully attractive between nucleons at distances of about 1 femtometer (fm, or 1.0 × 10−15 metres) between their centers, but rapidly decreases to insignificance at distances beyond about 2.5 fm. At distances less than 0.7 fm, the nuclear force becomes repulsive. This repulsive component is responsible for the physical size of nuclei, since the nucleons can come no closer than the force allows. By comparison, the size of an atom, measured in angstroms (Å, or 1.0 × 10−10 m), is five orders of magnitude larger. The nuclear force is not simple, however, since it depends on the nucleon spins, has a tensor component, and may depend on the relative momentum of the nucleons.A quantitative description of the nuclear force relies on partially empirical equations that model the internucleon potential energies, or potentials. (Generally, forces within a system of particles can be more simply modeled by describing the system's potential energy; the negative gradient of a potential is equal to the vector force.) The constants for the equations are phenomenological, that is, determined by fitting the equations to experimental data. The internucleon potentials attempt to describe the properties of nucleon–nucleon interaction. Once determined, any given potential can be used in, e.g., the Schrödinger equation to determine the quantum mechanical properties of the nucleon system.The discovery of the neutron in 1932 revealed that atomic nuclei were made of protons and neutrons, held together by an attractive force. By 1935 the nuclear force was conceived to be transmitted by particles called mesons. This theoretical development included a description of the Yukawa potential, an early example of a nuclear potential. Mesons, predicted by theory, were discovered experimentally in 1947. By the 1970s, the quark model had been developed, which showed that the mesons and nucleons were composed of quarks and gluons. By this new model, the nuclear force, resulting from the exchange of mesons between neighboring nucleons, is a residual effect of the strong force.