Atomic Theory (2
... 4.) Describe how reactivity changes (how easily an element changes in a chemical reaction) as you go up/down/across the periodic table for both metals and non-metals. 5.) Define Period. 6.) How many periods are found on the periodic table? 7.) What period does the D-block start in? 8.) What group do ...
... 4.) Describe how reactivity changes (how easily an element changes in a chemical reaction) as you go up/down/across the periodic table for both metals and non-metals. 5.) Define Period. 6.) How many periods are found on the periodic table? 7.) What period does the D-block start in? 8.) What group do ...
THE DISCOVERY OF ATOMIC PARTICLES
... directed high-energy electrons at samples of pure elements. Electrons decelerate rapidly on impact and in so doing emit x-rays. The x-rays emitted are recorded photographically as a series of lines – their patterns varying with the atomic mass of the element. On the basis of mathematical analysis of ...
... directed high-energy electrons at samples of pure elements. Electrons decelerate rapidly on impact and in so doing emit x-rays. The x-rays emitted are recorded photographically as a series of lines – their patterns varying with the atomic mass of the element. On the basis of mathematical analysis of ...
1 - Hobbs Freshman High School
... following? (electrons only, neutrons and electrons, protons and electrons, protons and neutrons) 8. The positively charged particles found in an atom are called (electrons, neutrons, nuclei, protons) 9. Where is most of the mass of an atom located? (in the energy levels, in the electron cloud, insid ...
... following? (electrons only, neutrons and electrons, protons and electrons, protons and neutrons) 8. The positively charged particles found in an atom are called (electrons, neutrons, nuclei, protons) 9. Where is most of the mass of an atom located? (in the energy levels, in the electron cloud, insid ...
Atoms Ions Valence Electrons Isotopes
... ▪ ex: an atom with 12 electrons, 1s2 2s2 2p6 3s2 , has 2 valence electrons that exist at the 3rd energy level and 10 inner-shell electrons that exist at the 1st and 2nd energy ...
... ▪ ex: an atom with 12 electrons, 1s2 2s2 2p6 3s2 , has 2 valence electrons that exist at the 3rd energy level and 10 inner-shell electrons that exist at the 1st and 2nd energy ...
Unit 2 -- Atomic Structure, Periodic Table, and
... Semi-Metals (metalloids) • Divides metals from nonmetals • properties intermediate between metals and nonmetals • Ex: Si, Ge, ... • “stair case” on right Side of periodic table ...
... Semi-Metals (metalloids) • Divides metals from nonmetals • properties intermediate between metals and nonmetals • Ex: Si, Ge, ... • “stair case” on right Side of periodic table ...
CHEM 1405 CHAPTER 4
... present in the nucleus. The electrons revolve round the nucleus in specified orbits known as energy levels or energy shells. Bohr’s Theory of the Hydrogen atom The hydrogen atom has only one electron, which is present in the first shell. When energy is supplied to the ‘H’ atoms the electrons get exc ...
... present in the nucleus. The electrons revolve round the nucleus in specified orbits known as energy levels or energy shells. Bohr’s Theory of the Hydrogen atom The hydrogen atom has only one electron, which is present in the first shell. When energy is supplied to the ‘H’ atoms the electrons get exc ...
A or `Mass Number` - Uplift Pinnacle Prep
... The US mint estimates that of all the pennies currently in circulation 66.5% of them are “new” (post-1982) pennies and 33.5% are ‘old’ pennies. A ‘new’ penny weighs 2.5g and an old penny weighs 3.1 g. Use this information to determine the average mass of a penny. ...
... The US mint estimates that of all the pennies currently in circulation 66.5% of them are “new” (post-1982) pennies and 33.5% are ‘old’ pennies. A ‘new’ penny weighs 2.5g and an old penny weighs 3.1 g. Use this information to determine the average mass of a penny. ...
Atomic - Ms. Dawkins
... • Electrons are much smaller than protons (2000 times smaller). • Electrons move around the nucleus very quickly. Scientists have found that it is not possible to determine the exact position of any single electron in an atom because they are moving too fast. This is why we picture electrons as a cl ...
... • Electrons are much smaller than protons (2000 times smaller). • Electrons move around the nucleus very quickly. Scientists have found that it is not possible to determine the exact position of any single electron in an atom because they are moving too fast. This is why we picture electrons as a cl ...
Unit 4 Test REVIEW
... 30. Atomic mass is a relative scale based on which nuclide? 31. What is the mass number of an atom that has 20 protons, 22 neutrons and 20 electrons? 32. The word atom comes from the Greek word atomos meaning __________________. 33. In the synthesis of sulfur trifluoride, 33.0 g of sulfur combines w ...
... 30. Atomic mass is a relative scale based on which nuclide? 31. What is the mass number of an atom that has 20 protons, 22 neutrons and 20 electrons? 32. The word atom comes from the Greek word atomos meaning __________________. 33. In the synthesis of sulfur trifluoride, 33.0 g of sulfur combines w ...
Chemistry I Review - BarbaraElam-Rice
... 29) Describe the difference between cations and anions. How are they formed? ...
... 29) Describe the difference between cations and anions. How are they formed? ...
Chapter 1 Chemistry: The Study of Matter
... Compounds can be broken down by chemical methods When they are broken down, the components have completely different properties than the compound. ...
... Compounds can be broken down by chemical methods When they are broken down, the components have completely different properties than the compound. ...
CHAPTER 2 - Net Start Class
... o The results were astounding [poor Geiger and Marsden first suffered Rutherford’s wrath and were told to try again—this couldn’t be!]. Most of the particles did pass straight through, BUT many were deflected at LARGE angles and some even REFLECTED! Rutherford stated that was like “shooting a ...
... o The results were astounding [poor Geiger and Marsden first suffered Rutherford’s wrath and were told to try again—this couldn’t be!]. Most of the particles did pass straight through, BUT many were deflected at LARGE angles and some even REFLECTED! Rutherford stated that was like “shooting a ...
File
... 1. If an atom has an atomic number of 8, how many protons does the atom have? _______ protons. a. Which is also equal to the number of (circle all that apply) Protons Neutrons Electrons 2. What are the three sub-atomic particles and why are they identified as sub-atomic? ____________________________ ...
... 1. If an atom has an atomic number of 8, how many protons does the atom have? _______ protons. a. Which is also equal to the number of (circle all that apply) Protons Neutrons Electrons 2. What are the three sub-atomic particles and why are they identified as sub-atomic? ____________________________ ...
North Haven Public Schools Curriculum
... Thomson, Bohr, Rutherford, and Millikan. The position of an element in the periodic table is related to its atomic number. Conservation of Matter and Stoichiometry – The conservation of atoms in chemical reactions leads to the principle of conservations of matter and the ability to calculate the m ...
... Thomson, Bohr, Rutherford, and Millikan. The position of an element in the periodic table is related to its atomic number. Conservation of Matter and Stoichiometry – The conservation of atoms in chemical reactions leads to the principle of conservations of matter and the ability to calculate the m ...
Internal Structure of Atoms - Belle Vernon Area School District
... and electrons are present in copper which has an atomic number of 29 and a mass number of 64? ...
... and electrons are present in copper which has an atomic number of 29 and a mass number of 64? ...
ATOMS: THE BUILDING BLOCKS OF MATTER
... Atoms of different elements combine in In chemical reactions, atoms are LAW OF MULTIPLE PROPORTION Atoms of different elements combine in simple whole number ratios to form chemical compounds Not all aspects of Dalton’s atomic theory have proven to be correct Dalton’s atomic theory have been modifie ...
... Atoms of different elements combine in In chemical reactions, atoms are LAW OF MULTIPLE PROPORTION Atoms of different elements combine in simple whole number ratios to form chemical compounds Not all aspects of Dalton’s atomic theory have proven to be correct Dalton’s atomic theory have been modifie ...
Prentice Hall Chemistry Worksheets
... • Describe Democritus’s ideas about atoms • Explain Dalton’s atomic theory • Describe the size of an atom ...
... • Describe Democritus’s ideas about atoms • Explain Dalton’s atomic theory • Describe the size of an atom ...
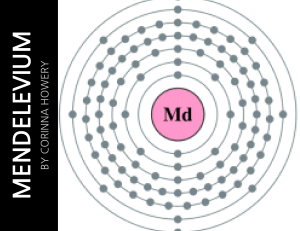
Mendelevium
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
... table so its atomic number is 101. There are 101 protons/electrons in the nucleus and 157 neutrons. It also has 2 valence electrons. Mendelevium has 7 shells. On the periodic table, mendelevium is in the group actinide and it is radioactive. Mendeleviums state of matter is radioactive. ...
Itty-Bitty Atoms
... science? Would you like to be a scientist? If so, what would you like to study? What do you think scientists of the future will study? 4. Answer the following questions: a. Who is Dmitry Mendeleyev and what did he do? b. What is chemistry? c. How big are atoms? 5. When a teacher calls out a symbol f ...
... science? Would you like to be a scientist? If so, what would you like to study? What do you think scientists of the future will study? 4. Answer the following questions: a. Who is Dmitry Mendeleyev and what did he do? b. What is chemistry? c. How big are atoms? 5. When a teacher calls out a symbol f ...
DEFINING THE ATOM - Southgate Schools
... 6. _______________________ 7. _______________________ 8. _______________________ ...
... 6. _______________________ 7. _______________________ 8. _______________________ ...
Structure of the Atom
... 3) Atoms combine in whole-number ratios to form compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
... 3) Atoms combine in whole-number ratios to form compounds 4) In chemical reactions, atoms are combined, separated, or rearranged – but never changed into atoms of another element. ...
Atoms, molecules and ions
... • The neutron has no charge and is slightly larger than the proton (1.67493 x 10-24 g for the neutron vs 1.67252 x 10-24 g for the proton) ...
... • The neutron has no charge and is slightly larger than the proton (1.67493 x 10-24 g for the neutron vs 1.67252 x 10-24 g for the proton) ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.