Please use your NUMERICAL RESPONSE SHEET to answer the
... Use the following diagram to answer then next 4 questions Zarley was studying the periodic table and looking for any patterns in the arrangement of the elements. ...
... Use the following diagram to answer then next 4 questions Zarley was studying the periodic table and looking for any patterns in the arrangement of the elements. ...
Atomic History WebQuest
... – (4) Atoms of the same element can unite in more than one ratio with another element to form more than one compound. Atoms can unite with other atoms in simple numerical ratios to form compounds (law of multiple proportions). ...
... – (4) Atoms of the same element can unite in more than one ratio with another element to form more than one compound. Atoms can unite with other atoms in simple numerical ratios to form compounds (law of multiple proportions). ...
Atoms, Molecules and Ions
... Comic book periodic table (http://www.uky.edu/Projects/Chemcomics/) ...
... Comic book periodic table (http://www.uky.edu/Projects/Chemcomics/) ...
Subatomic Particles
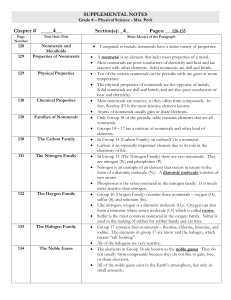
... Unit 2 / Matter Subatomic Particles Introduction: Noble gases, halogens, alkali metals, and alkaline earth metals are “families” of elements found in columns of the periodic table. These elements are related by sharing similar properties with the other elements in their family. Is there some common ...
... Unit 2 / Matter Subatomic Particles Introduction: Noble gases, halogens, alkali metals, and alkaline earth metals are “families” of elements found in columns of the periodic table. These elements are related by sharing similar properties with the other elements in their family. Is there some common ...
Name - TeacherWeb
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
Atoms: The Building Blocks of Matter
... Why are elements placed in a group (column)? Why are elements placed in a period (row?) ...
... Why are elements placed in a group (column)? Why are elements placed in a period (row?) ...
word-doc Practice for the final exam!
... In general, as you go across a period in the periodic table from left to right: (1) the atomic radius __________; (2) the electron affinity becomes __________ negative; and (3) the first ionization energy ___________. a. decreases, decreasingly, increases b. increases, increasingly, decreases c. inc ...
... In general, as you go across a period in the periodic table from left to right: (1) the atomic radius __________; (2) the electron affinity becomes __________ negative; and (3) the first ionization energy ___________. a. decreases, decreasingly, increases b. increases, increasingly, decreases c. inc ...
The Atom: Quick Note Guide
... Democritus Named the atom Dalton All matter is made up of small particles called atoms Atoms of a given element are identical in size and mass Atoms of different elements can physically/chemically combine to form compounds In reactions, atoms combine, separate, or rearrange Atoms cannot be divided, ...
... Democritus Named the atom Dalton All matter is made up of small particles called atoms Atoms of a given element are identical in size and mass Atoms of different elements can physically/chemically combine to form compounds In reactions, atoms combine, separate, or rearrange Atoms cannot be divided, ...
Review 2 - Solutions - Mayfield City Schools
... Know that light comes from or is emitted by electrons changing from higher to lower energy levels. Know that light is taken in or is absorbed by electrons changing from lower to higher energy levels. Know that different elements give off different spectrums or colors of light. Know the order of the ...
... Know that light comes from or is emitted by electrons changing from higher to lower energy levels. Know that light is taken in or is absorbed by electrons changing from lower to higher energy levels. Know that different elements give off different spectrums or colors of light. Know the order of the ...
HW Problems
... CHE1031 Lecture 2 HW Problems must be solved, or written out, in their entirety with all work shown on engineering graph paper. You must label each set in the upper left hand corner with your name, the date and the chapter. Problems must be identified by number and all work must be shown with answer ...
... CHE1031 Lecture 2 HW Problems must be solved, or written out, in their entirety with all work shown on engineering graph paper. You must label each set in the upper left hand corner with your name, the date and the chapter. Problems must be identified by number and all work must be shown with answer ...
and the atomic
... Carbon- 12 as a standard • carbon- 12 – ALL masses on the periodic table are based on their relationship to carbon-12 • the C-12 atom has been given the atomic weight of exactly 12.000000000 and is used as the basis upon which the atomic weight of other isotopes is determined ...
... Carbon- 12 as a standard • carbon- 12 – ALL masses on the periodic table are based on their relationship to carbon-12 • the C-12 atom has been given the atomic weight of exactly 12.000000000 and is used as the basis upon which the atomic weight of other isotopes is determined ...
Regents_Chem_Core_for_review
... IV.8 Atoms attain a stable valence electron configuration by bonding with other atoms. Noble gases have stable valence configurations and tend not to bond. (5.2b) IV.9 Physical properties of substances can be explained in terms of chemical bonds and intermolecular forces. These properties include co ...
... IV.8 Atoms attain a stable valence electron configuration by bonding with other atoms. Noble gases have stable valence configurations and tend not to bond. (5.2b) IV.9 Physical properties of substances can be explained in terms of chemical bonds and intermolecular forces. These properties include co ...
document
... An electron’s location at any time can not be pinpointed exactly, yet you have a good idea of where it should be. ...
... An electron’s location at any time can not be pinpointed exactly, yet you have a good idea of where it should be. ...
Elements
... – Electrons that move around the nucleus at an average distance of about 10-8 cm away. – Electrons and protons having equal and opposite charges while neutrons have no charge. – Protons and neutrons almost 2000 times more massive than electrons. ...
... – Electrons that move around the nucleus at an average distance of about 10-8 cm away. – Electrons and protons having equal and opposite charges while neutrons have no charge. – Protons and neutrons almost 2000 times more massive than electrons. ...
Periodic Properties of the Elements Effective Nuclear Charge, Zeff
... Remember that for many electron atoms, the energies of orbitals with the same n value increase in the order ns < np < nd < nf. This can be explained by the following: • In general, for a given n value: s electrons penetrate closer to the nucleus than p p electrons penetrate closer to the nucleus tha ...
... Remember that for many electron atoms, the energies of orbitals with the same n value increase in the order ns < np < nd < nf. This can be explained by the following: • In general, for a given n value: s electrons penetrate closer to the nucleus than p p electrons penetrate closer to the nucleus tha ...
Name: Period: ______ Date: Atom Models Elements are made up of
... 2. By looking at the atomic number, figure out how many protons it has 3. Draw those number of protons inside the nucleus Number of neutrons: 4. Then, subtract the number of protons from the atomic mass (look on the periodic table) to find the number of neutrons the atom contains 5. Draw those numbe ...
... 2. By looking at the atomic number, figure out how many protons it has 3. Draw those number of protons inside the nucleus Number of neutrons: 4. Then, subtract the number of protons from the atomic mass (look on the periodic table) to find the number of neutrons the atom contains 5. Draw those numbe ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.