TEK 8.5D: Chemical Formulas
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
Acids and Bases B.pps
... times smaller than protons and neutrons, so almost all the mass of an atom is concentrated in its nucleus. ...
... times smaller than protons and neutrons, so almost all the mass of an atom is concentrated in its nucleus. ...
chemistry i - surrattchemistry
... 31. Which substance would have London dispersion forces as the main type of intermolecular forces of attraction? a. H2O b. F2 d. HCl d. NaCl 32. Diamond, graphite, and silicon dioxide all exhibit which type of intermolecular force? a. metallic b. network covalent c. ionic d. hydrogen e. dipole-dipol ...
... 31. Which substance would have London dispersion forces as the main type of intermolecular forces of attraction? a. H2O b. F2 d. HCl d. NaCl 32. Diamond, graphite, and silicon dioxide all exhibit which type of intermolecular force? a. metallic b. network covalent c. ionic d. hydrogen e. dipole-dipol ...
ISOTOPES 3 SUBATOMIC PARTICLES Proton Located inside the
... Located outside of the nucleus in an “electron cloud” Involved in chemical bonding Negative charge Equal to the # of protons in a neutral atom How many electrons does Potassium have? How many electrons does Nitrogen have? o Neutron Located inside the nucleus of an atom No charge # ...
... Located outside of the nucleus in an “electron cloud” Involved in chemical bonding Negative charge Equal to the # of protons in a neutral atom How many electrons does Potassium have? How many electrons does Nitrogen have? o Neutron Located inside the nucleus of an atom No charge # ...
Development of the Atomic Theory
... Valence electrons are in outermost level. They give an atom its chemical properties/reactivity ...
... Valence electrons are in outermost level. They give an atom its chemical properties/reactivity ...
Game Face: Quarter Test Review Answers You`re Welcome, Please
... 41. Which idea of Thomson’s atomic model is different than Dalton’s? a. Thomson thought atoms were tiny and indivisible b. Thomson thought that atoms solid c. Thomson thought that atoms were divisible d. Thomson thought that atoms have a nucleus 42. Which of the following is accepted in our current ...
... 41. Which idea of Thomson’s atomic model is different than Dalton’s? a. Thomson thought atoms were tiny and indivisible b. Thomson thought that atoms solid c. Thomson thought that atoms were divisible d. Thomson thought that atoms have a nucleus 42. Which of the following is accepted in our current ...
atomos
... His model assumed that the electron exists at a precise distance from the nucleus, as long as an electron remains in one location no energy is given off, electrons have circular orbits (this is only correct in s orbits), and the angular momenta associated with allowed electron motion are integral mu ...
... His model assumed that the electron exists at a precise distance from the nucleus, as long as an electron remains in one location no energy is given off, electrons have circular orbits (this is only correct in s orbits), and the angular momenta associated with allowed electron motion are integral mu ...
Notes on Atomic Structure atoms
... numbers and types of atoms. Atoms are indivisible in chemical processes. That is, atoms are not created or destroyed in chemical reactions. A chemical reaction simply changes the way atoms are grouped together. ...
... numbers and types of atoms. Atoms are indivisible in chemical processes. That is, atoms are not created or destroyed in chemical reactions. A chemical reaction simply changes the way atoms are grouped together. ...
An Introduction to Matter
... – A chemical change is a change that does alter the identity of the matter. – A compound is a pure substance that can be decomposed by a chemical change into simpler substances – An element is a pure substance which cannot be broken down into anything simpler by either physical or chemical means. ...
... – A chemical change is a change that does alter the identity of the matter. – A compound is a pure substance that can be decomposed by a chemical change into simpler substances – An element is a pure substance which cannot be broken down into anything simpler by either physical or chemical means. ...
SOLUBILITY RULES FOR IONIC COMPOUNDS IN WATER
... 1. Identify the scientist responsible for each of the following: (a) first to propose that matter is composed of atoms (b) identified matter as being composed of either elements or compounds (c) showed that mass is conserved during chemical reactions (d) proposed that compounds always contain the sa ...
... 1. Identify the scientist responsible for each of the following: (a) first to propose that matter is composed of atoms (b) identified matter as being composed of either elements or compounds (c) showed that mass is conserved during chemical reactions (d) proposed that compounds always contain the sa ...
8th Grade Science: 1st Six Weeks At-A
... periods, to explain how properties are used to classify elements. SCI.8.2D Construct tables, using repeated trials and means, to organize data and identify patterns. SCI.8.2E Analyze data to formulate reasonable explanations, communicate valid conclusions supported by the data, and predict trends. S ...
... periods, to explain how properties are used to classify elements. SCI.8.2D Construct tables, using repeated trials and means, to organize data and identify patterns. SCI.8.2E Analyze data to formulate reasonable explanations, communicate valid conclusions supported by the data, and predict trends. S ...
Chapter 4 Powerpoint
... Atoms are neither created nor destroyed in any chemical reaction. A given compound always has the same relative numbers and kinds of atoms. ...
... Atoms are neither created nor destroyed in any chemical reaction. A given compound always has the same relative numbers and kinds of atoms. ...
J.J. Thomson and the Cathode Ray Tube 1897
... Niels Bohr (1885-1862) • Electrons have orbits about the nucleus (planetary theory) • Electrons could only exist at given energy levels • An energy level is where an electron is likely to be moving • Energy levels were like steps on a ladder – An electron can only be at any given step at any given ...
... Niels Bohr (1885-1862) • Electrons have orbits about the nucleus (planetary theory) • Electrons could only exist at given energy levels • An energy level is where an electron is likely to be moving • Energy levels were like steps on a ladder – An electron can only be at any given step at any given ...
Chapter 3 Chemical Foundations: Elements, Atoms, and Ions
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
... 1. Elements are made of tiny particles called atoms. 2. All atoms of a given element are identical. 3. The atoms of a given element are different from those of any other element. 4. Atoms of one element can combine with atoms of other elements to form compounds. A given compound always has the same ...
1H Atomic Theory Quiz Review
... 13. What evidence told scientists that electrons were located on specific energy levels, rather than being scattered randomly around the nucleus? ...
... 13. What evidence told scientists that electrons were located on specific energy levels, rather than being scattered randomly around the nucleus? ...
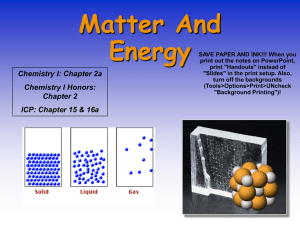
Matter and Energy
... only (2) compounds only (3) mixtures only (4) all of these 7. Which of the following is a type of mixture? (1) elements only(2) compounds only (3) solutions only (4) elements and Compounds 8. Which of the following is NOT composed of two or more types of atoms? (1) element (2) compound (3) solution ...
... only (2) compounds only (3) mixtures only (4) all of these 7. Which of the following is a type of mixture? (1) elements only(2) compounds only (3) solutions only (4) elements and Compounds 8. Which of the following is NOT composed of two or more types of atoms? (1) element (2) compound (3) solution ...
Atomic and Molecular Structure
... The number of electrons on the same energy level (period) are the VALENCE ELECTRONS. These are the electrons available for bonding. Usually it is the same as the group number (1-8A). e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. Remember- The n ...
... The number of electrons on the same energy level (period) are the VALENCE ELECTRONS. These are the electrons available for bonding. Usually it is the same as the group number (1-8A). e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. Remember- The n ...
Day 2 Guided Reading Chapter 4
... 3. T or F Dalton gathered evidence for the existence of atoms by measuring the masses of elements that reacted to form compounds. 4. What theory did Dalton propose to explain why the elements in a compound always join in the same way? 5. Which of the following sentences represent the main points of ...
... 3. T or F Dalton gathered evidence for the existence of atoms by measuring the masses of elements that reacted to form compounds. 4. What theory did Dalton propose to explain why the elements in a compound always join in the same way? 5. Which of the following sentences represent the main points of ...
Chemistry is a material science
... physical changes. In a chemical reaction new _________ are formed with a new and different set of physical and chemical properties. For example, when H2 and O2 react to ...
... physical changes. In a chemical reaction new _________ are formed with a new and different set of physical and chemical properties. For example, when H2 and O2 react to ...
Word - The Chemistry Book
... 1e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. The volume of the hydrogen nucleus is about one trillion times less than the volume of the hydrogen atom, yet the nucleus contains almost all the mass in the form of one proton. The diameter of an ...
... 1e. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. The volume of the hydrogen nucleus is about one trillion times less than the volume of the hydrogen atom, yet the nucleus contains almost all the mass in the form of one proton. The diameter of an ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.