Chapter 05
... Arranged elements according to increasing atomic number The arrangement today P.124 – long form Symbol, atomic number & mass Henry ...
... Arranged elements according to increasing atomic number The arrangement today P.124 – long form Symbol, atomic number & mass Henry ...
atomic structure
... a. Atom is the smallest particle of matter b. Atom is small sizes and round-shaped, indivisible, neither created nor destoyed. c. All atoms of given element are identical d. Atoms of different element can be joined to produce substance with fixed scale. e. Atoms combine to make compounds: a given co ...
... a. Atom is the smallest particle of matter b. Atom is small sizes and round-shaped, indivisible, neither created nor destoyed. c. All atoms of given element are identical d. Atoms of different element can be joined to produce substance with fixed scale. e. Atoms combine to make compounds: a given co ...
Synthesis of elements by helium and oxygen building blocks Bohr
... moment of hydrogen is fixed to be “1”. Atoms with odd mass number possess magnetic moment 1, atoms with even mass number have zero magnetic moment. Elements are fusion products of hydrogen, deuterium, helium and oxygen building blocks. Helium consists of four hydrogen atoms. The architecture (aufbau ...
... moment of hydrogen is fixed to be “1”. Atoms with odd mass number possess magnetic moment 1, atoms with even mass number have zero magnetic moment. Elements are fusion products of hydrogen, deuterium, helium and oxygen building blocks. Helium consists of four hydrogen atoms. The architecture (aufbau ...
Atomic Structure - Hannah E. Styron
... Potassium has an atomic mass of 39 and has 19 electrons. How many neutrons and protons does it have and what is it’s atomic number? ...
... Potassium has an atomic mass of 39 and has 19 electrons. How many neutrons and protons does it have and what is it’s atomic number? ...
Chapter 12
... Table 2.3 lists alphabetically the names of a number of common cations and anions. Transition metals can form more than one type of cation. Iron can form two cations: Fe2+, Fe3+. The Stock system is used to name most transition metals, by indicating charge on metal with ...
... Table 2.3 lists alphabetically the names of a number of common cations and anions. Transition metals can form more than one type of cation. Iron can form two cations: Fe2+, Fe3+. The Stock system is used to name most transition metals, by indicating charge on metal with ...
Honors Chemistry Chapter 4 Student Notes
... samples are vaporized, bombarded with electrons (in order to create + ions [positively charged particles due to a loss of 1 electron]), and placed in electrical and magnetic fields. Due to differences in mass ( # of neutrons) the paths of the molecules curve based on their individual mass. Heavier p ...
... samples are vaporized, bombarded with electrons (in order to create + ions [positively charged particles due to a loss of 1 electron]), and placed in electrical and magnetic fields. Due to differences in mass ( # of neutrons) the paths of the molecules curve based on their individual mass. Heavier p ...
Topic 2
... A greater than the mass of a neutron B the same as the mass of a proton C smaller than the mass of a proton D the same as the mass of a neutron (b) The atomic number of oxygen is 8. The mass number of an atom of oxygen is 17. Describe the number and type of particles in the nucleus of this atom. ...
... A greater than the mass of a neutron B the same as the mass of a proton C smaller than the mass of a proton D the same as the mass of a neutron (b) The atomic number of oxygen is 8. The mass number of an atom of oxygen is 17. Describe the number and type of particles in the nucleus of this atom. ...
Atoms and molecules
... Element and compounds are the only pure substances that can exist. If two or more kinds of molecules are present together they form a mixture. Most of the materials encountered are mixtures: air, earth, sea water, plants. One of the chemicals most important and difficulty jobs is to sort out the nat ...
... Element and compounds are the only pure substances that can exist. If two or more kinds of molecules are present together they form a mixture. Most of the materials encountered are mixtures: air, earth, sea water, plants. One of the chemicals most important and difficulty jobs is to sort out the nat ...
(3.3 × 10!4) + (2.52 × 10!2) = (3.3 × 10!4) × (2.52 × 10!2)
... A pure substance has well defined physical and chemical properties. Pure substances can be classified as elements or compounds. Compounds can be further reduced into two or more elements. Elements consist of only one type of atom. They cannot be decomposed or further simplified by ordinary means. ...
... A pure substance has well defined physical and chemical properties. Pure substances can be classified as elements or compounds. Compounds can be further reduced into two or more elements. Elements consist of only one type of atom. They cannot be decomposed or further simplified by ordinary means. ...
THE CHEMICAL BASIS OF LIFE
... 7 atoms having the same atomic number but a different atomic weight due to the number of neutrons 8 molecule that contains atoms of more than one element 9 substance or group of substances that tend to resist pH changes in a solution, thus stabilizing the solution’s relative acidity 10 weak bond tha ...
... 7 atoms having the same atomic number but a different atomic weight due to the number of neutrons 8 molecule that contains atoms of more than one element 9 substance or group of substances that tend to resist pH changes in a solution, thus stabilizing the solution’s relative acidity 10 weak bond tha ...
Development of Atomic Theory
... number = the sum of the protons and neutrons Number of neutrons in an element can vary (called isotopes: atoms of the same element that have different mass numbers) This means the number of _protons_ is the same, and the number of _neutrons_ is different. ...
... number = the sum of the protons and neutrons Number of neutrons in an element can vary (called isotopes: atoms of the same element that have different mass numbers) This means the number of _protons_ is the same, and the number of _neutrons_ is different. ...
Why are atoms of lead different to those of gold and why can we not
... His results showed that most of the gold foil is space as the radioactive particles went straight through the foil. This left little cirles where the gold atoms were. It was not until 1932 that we finally established the structure of the atom. Lets look at an atoms of helium. ...
... His results showed that most of the gold foil is space as the radioactive particles went straight through the foil. This left little cirles where the gold atoms were. It was not until 1932 that we finally established the structure of the atom. Lets look at an atoms of helium. ...
Electrons in atoms practice test File
... c. energy level b. speed d. electron cloud shape ____ 9. How many energy sublevels are in the second principal energy level? a. 1 c. 3 b. 2 d. 4 ____ 10. When an electron moves from a lower to a higher energy level, the electron ____. a. always doubles its energy b. absorbs a continuously variable a ...
... c. energy level b. speed d. electron cloud shape ____ 9. How many energy sublevels are in the second principal energy level? a. 1 c. 3 b. 2 d. 4 ____ 10. When an electron moves from a lower to a higher energy level, the electron ____. a. always doubles its energy b. absorbs a continuously variable a ...
According to Democritus, atoms were small
... particles (atoms). Although he did not know about their structure, he knew that the evidence pointed to something fundamental. He proposed the Atomic theory of matter based on his experimental observations. To Dalton : ...
... particles (atoms). Although he did not know about their structure, he knew that the evidence pointed to something fundamental. He proposed the Atomic theory of matter based on his experimental observations. To Dalton : ...
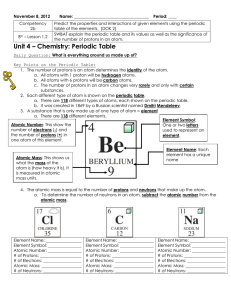
Intro to the Periodic Table
... c. How many protons are in one atom of this element? __________________ d. How many electrons are in one atom of this element? __________________ e. What is the atomic mass of this element? ______________________ f. How many neutrons are in one atom of this element? __________________ 8. Find the el ...
... c. How many protons are in one atom of this element? __________________ d. How many electrons are in one atom of this element? __________________ e. What is the atomic mass of this element? ______________________ f. How many neutrons are in one atom of this element? __________________ 8. Find the el ...
Atoms of a given element are identical in size, mass, and
... Cathode ray tubes pass electricity through a gas that is contained at a very low pressure. ...
... Cathode ray tubes pass electricity through a gas that is contained at a very low pressure. ...
Atoms, Molecules, and Ions Chapter 2
... The Discovery of Atomic Structure Cathode Rays and Electrons Consider the following experiment: •Oil drops are sprayed above a positively charged plate containing a small hole. •As the oil drops fall through the hole, they are given a negative charge. •Gravity forces the drops downward. The applied ...
... The Discovery of Atomic Structure Cathode Rays and Electrons Consider the following experiment: •Oil drops are sprayed above a positively charged plate containing a small hole. •As the oil drops fall through the hole, they are given a negative charge. •Gravity forces the drops downward. The applied ...
Document
... Valence shell: Outermost, highest energy shell of an atom. Valence electrons: An electron in an outermost shell of an atom. These electrons are loosely held, they are most important in determining an element’s properties. ...
... Valence shell: Outermost, highest energy shell of an atom. Valence electrons: An electron in an outermost shell of an atom. These electrons are loosely held, they are most important in determining an element’s properties. ...
General Chemistry
... Group 7 (halogens) have the most negative electron affinity because it have a room in it valence shell for an additional electron, Group 2 and Nobel gases have electron affinity near zero or positive, the above figure show how the Ionization energy increase through groups and periods. Electron affin ...
... Group 7 (halogens) have the most negative electron affinity because it have a room in it valence shell for an additional electron, Group 2 and Nobel gases have electron affinity near zero or positive, the above figure show how the Ionization energy increase through groups and periods. Electron affin ...
Periodic table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons in the nucleus), electron configurations, and recurring chemical properties. The table also shows four rectangular blocks: s-, p- d- and f-block. In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.The rows of the table are called periods; the columns are called groups. Six groups (columns) have names as well as numbers: for example, group 17 elements are the halogens; and group 18, the noble gases. The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized. The periodic table provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or reportedly synthesized, with elements 113, 115, 117, and 118 having yet to be confirmed. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that einsteinium and fermium once occurred in nature but currently do not. Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.