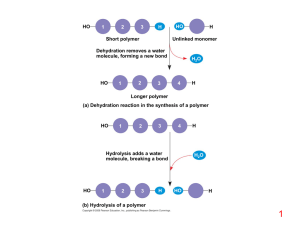
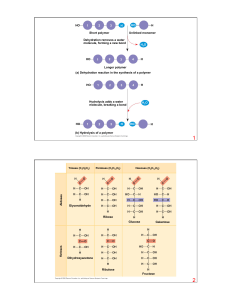
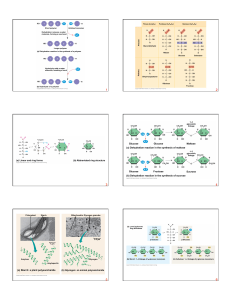
Macromolecules 9-3
... 1. At least one double 2. Plant/Fish sources 3. Liquid at room temperature 4. Considered best to consume 5. Two types of unsaturated fats a. Monounsaturated i. One double bond b. Polyunsaturated i. 2 or more bonds e. Phospholipids i. Remove one fatty acid chain from a regular fat ii. Replace the fat ...
... 1. At least one double 2. Plant/Fish sources 3. Liquid at room temperature 4. Considered best to consume 5. Two types of unsaturated fats a. Monounsaturated i. One double bond b. Polyunsaturated i. 2 or more bonds e. Phospholipids i. Remove one fatty acid chain from a regular fat ii. Replace the fat ...
Microbial Metabolism
... factors • Micronutrients: Metals and metalloids – Generally not necessary to add to medium – Deficiencies can arise when medium constituents are very pure ...
... factors • Micronutrients: Metals and metalloids – Generally not necessary to add to medium – Deficiencies can arise when medium constituents are very pure ...
ENGINEERING PROTEINS
... Ester –COOR, Acyl chloride –COCl, Amide -CONH2 Acid anhydride -(CO)2O The region in an enzyme into which the substrate binds before it reacts with it. An organic molecule that contains both an –NH2 group and a –COOH group Pairs of bases that fit neatly together thus placing groups in just the right ...
... Ester –COOR, Acyl chloride –COCl, Amide -CONH2 Acid anhydride -(CO)2O The region in an enzyme into which the substrate binds before it reacts with it. An organic molecule that contains both an –NH2 group and a –COOH group Pairs of bases that fit neatly together thus placing groups in just the right ...
File
... Hemoglobin in red blood cells acts as a buffer by preventing a drop in pH of the blood. It is a tertiary protein that bonds to iron, which gives it its red hue when carrying oxygen. ...
... Hemoglobin in red blood cells acts as a buffer by preventing a drop in pH of the blood. It is a tertiary protein that bonds to iron, which gives it its red hue when carrying oxygen. ...
Trace Metal Biogeochemistry 12.755
... The activity coefficient, gMn+, can be estimated by the Debye-Huckel correction or the Davies expression (modified Debye-Huckel) ...
... The activity coefficient, gMn+, can be estimated by the Debye-Huckel correction or the Davies expression (modified Debye-Huckel) ...
Monomer - Teacher Pages
... your body doesn’t have to work as hard to break it down; • Since polysaccharides are made up of many subunits, they need a lot of energy to be broken down and thus, you burn ...
... your body doesn’t have to work as hard to break it down; • Since polysaccharides are made up of many subunits, they need a lot of energy to be broken down and thus, you burn ...
defend your answer in 1
... true If a hydrophobic amino acid found in a protein residing in the cytosol is replaced with an amino acid with a charged side group, the protein will likely turn inside out. 2. (3 pts.) Examine the diagram shown below which was taken from your textbook. Which sentence or sentences is(are) true with ...
... true If a hydrophobic amino acid found in a protein residing in the cytosol is replaced with an amino acid with a charged side group, the protein will likely turn inside out. 2. (3 pts.) Examine the diagram shown below which was taken from your textbook. Which sentence or sentences is(are) true with ...
Chapter 2 Notes - Duplin County Schools
... Chemical reaction = changes one set of chemicals into another set of chemicals Chemical reactions have two parts: 1. Reactants 2. Products ...
... Chemical reaction = changes one set of chemicals into another set of chemicals Chemical reactions have two parts: 1. Reactants 2. Products ...
Slide 1 - helmricht
... Denaturation- the loss of the secondary, tertiary, and quaternary structures of a protein by chemical or physical agent that leaves the primary structure intact Enzymes lose their catalytic activity and other proteins can’t carry out their biological functions when denatured ...
... Denaturation- the loss of the secondary, tertiary, and quaternary structures of a protein by chemical or physical agent that leaves the primary structure intact Enzymes lose their catalytic activity and other proteins can’t carry out their biological functions when denatured ...
Unit 3 Review Sheet – Biochemistry
... 5. What are the characteristics of water that make it important to life? Polar, high heat capacity, resists temperature change, ability to bond and attract other molecules (cohesion and adhesion), ice is less dense than liquid water, universal solvent, most abundant compound in living things 6. What ...
... 5. What are the characteristics of water that make it important to life? Polar, high heat capacity, resists temperature change, ability to bond and attract other molecules (cohesion and adhesion), ice is less dense than liquid water, universal solvent, most abundant compound in living things 6. What ...
ELECTRON COUNTING IN TRANSITION METAL COMPLEXES
... charge on the structure. We don't have to do that here, because we already make the adjustment when we decide the "oxidation state" or charge on the metal. ...
... charge on the structure. We don't have to do that here, because we already make the adjustment when we decide the "oxidation state" or charge on the metal. ...
Ch.05The Structure and Function of Large Biological Molecules
... to carry oxygen is greatly reduced. ...
... to carry oxygen is greatly reduced. ...
Metal Sequestration (English version)
... ethylene diamino tetracarboxylic acid (EDTA) the term has become increasingly associated with the products having similar action. Sequestration is the suppression of a property or reaction of a metal without the removal of that metal from the system or phase by any process f precipitation, solvent e ...
... ethylene diamino tetracarboxylic acid (EDTA) the term has become increasingly associated with the products having similar action. Sequestration is the suppression of a property or reaction of a metal without the removal of that metal from the system or phase by any process f precipitation, solvent e ...
Ch.05The Structure and Function of Large Biological Molecules
... crystallize into a fiber; capacity to carry oxygen is greatly reduced. ...
... crystallize into a fiber; capacity to carry oxygen is greatly reduced. ...
Gas-Forming reactions Reactions that form a
... 4. The sum of the oxidation numbers is zero for an electrically neutral compound and equals the overall charge for an ionic species. HCl ...
... 4. The sum of the oxidation numbers is zero for an electrically neutral compound and equals the overall charge for an ionic species. HCl ...
MLAB 1315- Hematology Fall 2007 Keri Brophy
... A siderocyte is a red blood cell (anucleated) with excess iron Proper globin synthesis depends on genes. The precise order of amino acids in the globin chains is critical to the structure and function of hemoglobin. (More discussion to follow in ...
... A siderocyte is a red blood cell (anucleated) with excess iron Proper globin synthesis depends on genes. The precise order of amino acids in the globin chains is critical to the structure and function of hemoglobin. (More discussion to follow in ...
model answers
... 5. The M-P distance in (η5-C5H5)Co(PEt3)2 is 221.8 pm and the P-C distance is 184.6 pm. The corresponding distances in [(η5-C5H5)Co(PEt3)2]+ are 223 pm and 182.9 pm. Account for the changes in these distances as the former complex is oxidised. The oxidised complex is less able to donate electron den ...
... 5. The M-P distance in (η5-C5H5)Co(PEt3)2 is 221.8 pm and the P-C distance is 184.6 pm. The corresponding distances in [(η5-C5H5)Co(PEt3)2]+ are 223 pm and 182.9 pm. Account for the changes in these distances as the former complex is oxidised. The oxidised complex is less able to donate electron den ...
4. bonding - New Hartford Central Schools
... (I, II, III, IV, V, VI, VII, VIII, IX, X) FeO Iron (II) oxide ...
... (I, II, III, IV, V, VI, VII, VIII, IX, X) FeO Iron (II) oxide ...
Chemistry of Life
... Glucose: C6H12O6 forms ring structure in solution, major nutrient for cells, carbon skeletons serve as raw material for synthesis of other types of small organic molecules ...
... Glucose: C6H12O6 forms ring structure in solution, major nutrient for cells, carbon skeletons serve as raw material for synthesis of other types of small organic molecules ...
The Busch Catalyst
... Fe(Bcyclam) was a much better catalyst than Fe(TMC) Mechanistic investigation showed activation with Acetic Acid, but loss of dihydroxylation and only epoxidation Without Acetic Acid, dihydroxylation is favored Que suggests an FeV=O active catalyst for both ...
... Fe(Bcyclam) was a much better catalyst than Fe(TMC) Mechanistic investigation showed activation with Acetic Acid, but loss of dihydroxylation and only epoxidation Without Acetic Acid, dihydroxylation is favored Que suggests an FeV=O active catalyst for both ...
Absorption Spectra and Colours of Complexes
... series is essentially the same no matter what the metal ion is. Thus, water not only splits the dorbitals more than chloride for cobalt(II), but also for cobalt(III), iron(II), iron(III), nickel(II), platinum(IV), chromium(III), and so on. This series is as follows: I- < Br- < Cl- < SCN- < NO3- < F- ...
... series is essentially the same no matter what the metal ion is. Thus, water not only splits the dorbitals more than chloride for cobalt(II), but also for cobalt(III), iron(II), iron(III), nickel(II), platinum(IV), chromium(III), and so on. This series is as follows: I- < Br- < Cl- < SCN- < NO3- < F- ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.