Redox Reactions - KFUPM Faculty List
... Oxidation-reduction reactions (sometimes called redox reactions)) are reactions involvingg the transfer of one electron or more from one reactant to another. Redox reaction also involves the change in oxidation states for molecules. These reactions are very common in life: • Photosynthesis. (convers ...
... Oxidation-reduction reactions (sometimes called redox reactions)) are reactions involvingg the transfer of one electron or more from one reactant to another. Redox reaction also involves the change in oxidation states for molecules. These reactions are very common in life: • Photosynthesis. (convers ...
Unit 3 * Chapter 3 Biochemistry
... ____________(Covalent Bonding) – carbon only has 4 e-’s in its outer shell…..it would like to have 8. Therefore, carbon will easily form covalent bonds to fill its shell. ...
... ____________(Covalent Bonding) – carbon only has 4 e-’s in its outer shell…..it would like to have 8. Therefore, carbon will easily form covalent bonds to fill its shell. ...
Ch. 2 - Basic Chemistry
... (a) Include cholesterol, bile salts, vitamin D, and some hormones I. Cholesterol A. The basis for all steroids made in the body 3. Proteins ...
... (a) Include cholesterol, bile salts, vitamin D, and some hormones I. Cholesterol A. The basis for all steroids made in the body 3. Proteins ...
1 - UCSB CLAS
... b. Oxaloacetate 2. (Ch 24, #16) Which of the following compounds is more easily decarboxylated? O ...
... b. Oxaloacetate 2. (Ch 24, #16) Which of the following compounds is more easily decarboxylated? O ...
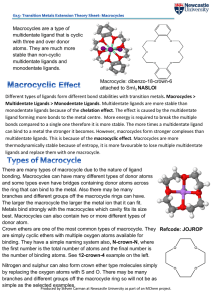
Different types of ligands form different bond stabilities with transition
... Macrocycles have many uses. A first major use of macrocycles was there use in dyes such as phalocyanine, a blue dye used in the textile and paper dying industry. Macrocycles also have use in medicine, their strong affinity for metals means they can be used in chelation therapy to treat heavy metal p ...
... Macrocycles have many uses. A first major use of macrocycles was there use in dyes such as phalocyanine, a blue dye used in the textile and paper dying industry. Macrocycles also have use in medicine, their strong affinity for metals means they can be used in chelation therapy to treat heavy metal p ...
Biomolecule Test Review 2015
... 9. What is the difference between saturated and unsaturated fatty acid? Which is better for you? Why? Saturated fatty acid- single bonds, straight and tightly packed. Solid at room temperature. (Bad for us!) Unsaturated fatty acid- double bonds bend the tails and it’s crooked (not straight). Liquid ...
... 9. What is the difference between saturated and unsaturated fatty acid? Which is better for you? Why? Saturated fatty acid- single bonds, straight and tightly packed. Solid at room temperature. (Bad for us!) Unsaturated fatty acid- double bonds bend the tails and it’s crooked (not straight). Liquid ...
Biomolecules Test Review -KEY
... 9. What is the difference between saturated and unsaturated fatty acid? Which is better for you? Why? Saturated fatty acid- single bonds, straight and tightly packed. Solid at room temperature. (Bad for us!) Unsaturated fatty acid- double bonds bend the tails and it’s crooked (not straight). Liquid ...
... 9. What is the difference between saturated and unsaturated fatty acid? Which is better for you? Why? Saturated fatty acid- single bonds, straight and tightly packed. Solid at room temperature. (Bad for us!) Unsaturated fatty acid- double bonds bend the tails and it’s crooked (not straight). Liquid ...
AP Chemistry 2013 Semester 1 Final Exam Review Problems
... atomic subshell energies and electron assignments; atomic electron configurations; electron configurations of ions; atomic properties and periodic trends; periodic trends and chemical properties. 12. An electron microscope employs a beam of electrons to obtain an image of an object. What energy must ...
... atomic subshell energies and electron assignments; atomic electron configurations; electron configurations of ions; atomic properties and periodic trends; periodic trends and chemical properties. 12. An electron microscope employs a beam of electrons to obtain an image of an object. What energy must ...
Shakedpresentation
... Role of P in human nutrition * Humans obtain P from eating plants, and use it to make bones, teeth, and shells. It is also an important constituent of cell membranes, DNA, RNA, and ATP. * On average, the human body contains 1 3/4 lb of P, with the overwhelming amount found in bones. The remainder i ...
... Role of P in human nutrition * Humans obtain P from eating plants, and use it to make bones, teeth, and shells. It is also an important constituent of cell membranes, DNA, RNA, and ATP. * On average, the human body contains 1 3/4 lb of P, with the overwhelming amount found in bones. The remainder i ...
NS 315 Unit 6: Proteins
... Protein- Case Study R.C. is a 30 year old male who is interested in losing weight and has heard the quickest way to do it is by a low carbohydrate, high protein diet. He realizes he also has to exercise, but since he cannot do cardio because he is always short of breath, he is going to lift weigh ...
... Protein- Case Study R.C. is a 30 year old male who is interested in losing weight and has heard the quickest way to do it is by a low carbohydrate, high protein diet. He realizes he also has to exercise, but since he cannot do cardio because he is always short of breath, he is going to lift weigh ...
Enterokinase (E4906)
... The FLAG protein expression system is based on the fusion of the 8 amino acid FLAG tag to the recombinant protein of choice. The rare 5 amino acid recognition sequence of the enterokinase cleavage site is incorporated into the FLAG tag. The FLAG sequence is: N-Asp-Tyr-Lys-Asp-Asp-Asp-Asp-Lys-C Clea ...
... The FLAG protein expression system is based on the fusion of the 8 amino acid FLAG tag to the recombinant protein of choice. The rare 5 amino acid recognition sequence of the enterokinase cleavage site is incorporated into the FLAG tag. The FLAG sequence is: N-Asp-Tyr-Lys-Asp-Asp-Asp-Asp-Lys-C Clea ...
Aminoacids
... – No electrons are exchanged as in charge effect but electrons are shared creating an attraction – Some amino acids can participate in multiple reactions acting as both donors and acceptors ...
... – No electrons are exchanged as in charge effect but electrons are shared creating an attraction – Some amino acids can participate in multiple reactions acting as both donors and acceptors ...
Chem 1411 Chapter 4
... Each Na atom loses one electron and each Cl atom gains one electron. Thus, Na undergoes oxidation and Cl2 undergoes reduction. ...
... Each Na atom loses one electron and each Cl atom gains one electron. Thus, Na undergoes oxidation and Cl2 undergoes reduction. ...
AP Notes Chapter 11
... Tetrahedral – [CoCl4]2- , [NiCl4]2[Ni(CO)4] , [Zn(NH3)4]2+ Square Planar – [Ni(CN)4]2- ...
... Tetrahedral – [CoCl4]2- , [NiCl4]2[Ni(CO)4] , [Zn(NH3)4]2+ Square Planar – [Ni(CN)4]2- ...
Ch 102 – Problem Set 7 Due: Thursday, May 26
... lowest energy singlet state is significantly higher in energy than the triplet state (22.5 kcal/mol) and is not easily accessible. Oxygenases are a class of enzymes that are able to bind and activate triplet oxygen leading to oxygenation of organic molecules. Cytochrome P450 is a well-studied type o ...
... lowest energy singlet state is significantly higher in energy than the triplet state (22.5 kcal/mol) and is not easily accessible. Oxygenases are a class of enzymes that are able to bind and activate triplet oxygen leading to oxygenation of organic molecules. Cytochrome P450 is a well-studied type o ...
Solutions to Problem Set #9
... is enough, but this is also a trivalent metal ion, which also serves to increase the crystal field splitting (relative to a divalent ion). [FeCl4]- will be high spin because the crystal field splitting for tetrahedral complexes is always small and they are therefore always high spin. It is worth not ...
... is enough, but this is also a trivalent metal ion, which also serves to increase the crystal field splitting (relative to a divalent ion). [FeCl4]- will be high spin because the crystal field splitting for tetrahedral complexes is always small and they are therefore always high spin. It is worth not ...
Modeling Chemical Evolution
... water could condense and trickle back into the first flask in a continuous cycle. At the end of one week of continuous operation, Miller and Urey observed that as much as 10–15% of the carbon within the system was now in the form of organic compounds. ...
... water could condense and trickle back into the first flask in a continuous cycle. At the end of one week of continuous operation, Miller and Urey observed that as much as 10–15% of the carbon within the system was now in the form of organic compounds. ...
The Chemical Basis of Life Atoms, Molecules, Ions, and Bonds
... • Thus, you will always see four lines connecting a carbon atom to other atoms, each line representing a pair of shared electrons (one electron from carbon and one from another atom). ...
... • Thus, you will always see four lines connecting a carbon atom to other atoms, each line representing a pair of shared electrons (one electron from carbon and one from another atom). ...
Plastics and Modern Materials - Smithycroft Secondary School
... 5. Some supermarkets no longer supply free polythene bags because they are non-biodegradable and can cause environmental problems. (a) What does non-biodegradable mean? ...
... 5. Some supermarkets no longer supply free polythene bags because they are non-biodegradable and can cause environmental problems. (a) What does non-biodegradable mean? ...
Metalloprotein

Metalloprotein is a generic term for a protein that contains a metal ion cofactor. A large number of all proteins are part of this category.