SOLUGEL Protein Gummies Leaflet
... collagen protein in each gummy Triple your gummies’ protein content with SOLUGEL®! The traditional gummy bear contains around 6g of protein per 100g, entirely from its gelatin content. With SOLUGEL®, it is now possible to create a gummy rich in collagen protein that looks and tastes like any other g ...
... collagen protein in each gummy Triple your gummies’ protein content with SOLUGEL®! The traditional gummy bear contains around 6g of protein per 100g, entirely from its gelatin content. With SOLUGEL®, it is now possible to create a gummy rich in collagen protein that looks and tastes like any other g ...
Ch. 5. Protein Purification and Characterization Techniques
... • Electrophoresis- charged particles migrate in electric field toward opposite charge • Proteins have different mobility: • Charge • Size • Shape ...
... • Electrophoresis- charged particles migrate in electric field toward opposite charge • Proteins have different mobility: • Charge • Size • Shape ...
Through the Looking Glass a New World of Proteins Enabled
... Recent advances in synthetic methods enable the routine synthesis of protein enantiomorphs, unnatural protein molecules made up entirely of D-amino acids. These D-proteins have a tertiary structure that is the mirror image of the backbone fold of their counterparts found in nature. Such mirror image ...
... Recent advances in synthetic methods enable the routine synthesis of protein enantiomorphs, unnatural protein molecules made up entirely of D-amino acids. These D-proteins have a tertiary structure that is the mirror image of the backbone fold of their counterparts found in nature. Such mirror image ...
Slide () - AccessEmergency Medicine
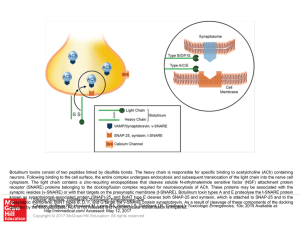
... Botulinum toxins consist of two peptides linked by disulfide bonds. The heavy chain is responsible for specific binding to acetylcholine (ACh) containing neurons. Following binding to the cell surface, the entire complex undergoes endocytosis and subsequent translocation of the light chain into the ...
... Botulinum toxins consist of two peptides linked by disulfide bonds. The heavy chain is responsible for specific binding to acetylcholine (ACh) containing neurons. Following binding to the cell surface, the entire complex undergoes endocytosis and subsequent translocation of the light chain into the ...
1.16891 / 1.16886 Fractogel® EMD COO
... to the tentacle technology. The Fractogel® beads have a high mechanical and chemical stability. Since the functional ion exchanger groups are bonded via linear polymer chains, the ionic groups are accessible for proteins. Fractogel® EMD COO- is a chromatographic support for the purification of basic ...
... to the tentacle technology. The Fractogel® beads have a high mechanical and chemical stability. Since the functional ion exchanger groups are bonded via linear polymer chains, the ionic groups are accessible for proteins. Fractogel® EMD COO- is a chromatographic support for the purification of basic ...
Abstract
... maps for many protein domains. Inferred contacts by mfDCA can be utilized as a reliable guide in high accuracy computational predictions of domain structure. Our results capture clear signals beyond intradomain residue contacts, for instance, interdomain interactions in macro molecular assemblies an ...
... maps for many protein domains. Inferred contacts by mfDCA can be utilized as a reliable guide in high accuracy computational predictions of domain structure. Our results capture clear signals beyond intradomain residue contacts, for instance, interdomain interactions in macro molecular assemblies an ...
Protein adsorption

Adsorption (not to be mistaken for absorption) is the accumulation and adhesion of molecules, atoms, ions, or larger particles to a surface, but without surface penetration occurring. The adsorption of larger biomolecules such as proteins is of high physiological relevance, and as such they adsorb with different mechanisms than their molecular or atomic analogs. Some of the major driving forces behind protein adsorption include: surface energy, intermolecular forces, hydrophobicity, and ionic or electrostatic interaction. By knowing how these factors affect protein adsorption, they can then be manipulated by machining, alloying, and other engineering techniques to select for the most optimal performance in biomedical or physiological applications.