EXPERIMENT 5: Oxidation of Alcohols: Solid
... relationship has led to the development of a convenient qualitative test for distinguishing primary and secondary alcohols (and aldehydes) from tertiary alcohols (and ketones). The qualitative test involves the addition of a solution of CrO3 in sulfuric acid (Jones' Reagent) to a solution of the com ...
... relationship has led to the development of a convenient qualitative test for distinguishing primary and secondary alcohols (and aldehydes) from tertiary alcohols (and ketones). The qualitative test involves the addition of a solution of CrO3 in sulfuric acid (Jones' Reagent) to a solution of the com ...
CHM412 June 2013 paper
... Within the H-bonding species, Carboxylic acids form dimers (as part d) mentions), that is, two sets of Hbonding to one other molecule, so their H-bonds are stronger than the hydrogen bonding in water. Withing the LDF categories, the species with the more open / straight chain structure and the more ...
... Within the H-bonding species, Carboxylic acids form dimers (as part d) mentions), that is, two sets of Hbonding to one other molecule, so their H-bonds are stronger than the hydrogen bonding in water. Withing the LDF categories, the species with the more open / straight chain structure and the more ...
Improvements & new technologies
... Advantages of heterogeneous process: •Lower initial costs •High conversion rate •Less chemicals are used •Lower consumption of methanol •Glycerine is purer Advantages of homogeneous process: •Biodiesel does not depend on glycerine price •Lower consumption of energy is needed ...
... Advantages of heterogeneous process: •Lower initial costs •High conversion rate •Less chemicals are used •Lower consumption of methanol •Glycerine is purer Advantages of homogeneous process: •Biodiesel does not depend on glycerine price •Lower consumption of energy is needed ...
Bronsted acidic ionic liquid as an efficient and reusable catalyst for
... chemical equilibrium is reached within 3 h at 80 °C (entry 1). Increase in the reaction time and temperature does not offer any significant advantage (entries 2–5). MAA was found to undergo cyclization at higher temperature to form cyclic esters and prolonged reaction time leading to the formation of ...
... chemical equilibrium is reached within 3 h at 80 °C (entry 1). Increase in the reaction time and temperature does not offer any significant advantage (entries 2–5). MAA was found to undergo cyclization at higher temperature to form cyclic esters and prolonged reaction time leading to the formation of ...
Alcohols - WordPress.com
... Alcohols are weak Brønsted bases Protonated by strong acids to yield oxonium ions, ...
... Alcohols are weak Brønsted bases Protonated by strong acids to yield oxonium ions, ...
Chem 400 Review Chem 350 JJ.S17
... Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabilize a conjugate base Alcohols preparation: via SN1, SN2, hydration, and Grignard (MgBr-[C…]) mechanisms Reduction: H2 with Pt/Pd, NaBH4 and LiAlH4 (more reactive, can be used for esters). Need quen ...
... Electron withdrawing substituents stabilize conjugate bases while electron donating ones destabilize a conjugate base Alcohols preparation: via SN1, SN2, hydration, and Grignard (MgBr-[C…]) mechanisms Reduction: H2 with Pt/Pd, NaBH4 and LiAlH4 (more reactive, can be used for esters). Need quen ...
Organic Chem Functional Groups
... known as the trans isomer. (trans : from latin meaning "across" - as in transatlantic). In the other, the two chlorine atoms are locked on the same side of the double bond. This is know as the cis isomer. (cis : from latin meaning "on this side" cis & trans isomers differ from each other in their ph ...
... known as the trans isomer. (trans : from latin meaning "across" - as in transatlantic). In the other, the two chlorine atoms are locked on the same side of the double bond. This is know as the cis isomer. (cis : from latin meaning "on this side" cis & trans isomers differ from each other in their ph ...
CHEMISTRY 263
... Chapter 21 – Phenols (read for overview sections 21.1 – 21.3 only) Chapter 22 – Carbohydrates (read sections 22.1 – 22.4 and sections 22.12 – 22.13) ...
... Chapter 21 – Phenols (read for overview sections 21.1 – 21.3 only) Chapter 22 – Carbohydrates (read sections 22.1 – 22.4 and sections 22.12 – 22.13) ...
CHE 312 Answers in BOLD RED EXAM 1 KEY (Ch. 16
... DRAW THE STEPS IN THIS MECHANISM. Be sure to include all major steps and show the structures of the main intermediates that are formed along the way as the following ester is saponified. ...
... DRAW THE STEPS IN THIS MECHANISM. Be sure to include all major steps and show the structures of the main intermediates that are formed along the way as the following ester is saponified. ...
3.5 revision guide alcohols
... Reagent: potassium dichromate (VI) solution and dilute sulphuric acid. Conditions: (use a limited amount of dichromate) warm gently and distil out the aldehyde as it forms: ...
... Reagent: potassium dichromate (VI) solution and dilute sulphuric acid. Conditions: (use a limited amount of dichromate) warm gently and distil out the aldehyde as it forms: ...
Chapter 5
... ◦ Thiols have lower boiling points and are less soluble in water and other polar solvents than alcohols of similar ...
... ◦ Thiols have lower boiling points and are less soluble in water and other polar solvents than alcohols of similar ...
Oxidation of alcohol to carboxylic acid under mild acidic condition
... INTRODUCTION Corey’s lactone was fist synthesized by E.J.Corey from cyclopentadiene[1] and it is key starting material for synthesis of prostaglandins [2] and prostaglandin possess a diverse range of biological activities including the treatment of glaucoma and ocular hypertension[3], chronic consti ...
... INTRODUCTION Corey’s lactone was fist synthesized by E.J.Corey from cyclopentadiene[1] and it is key starting material for synthesis of prostaglandins [2] and prostaglandin possess a diverse range of biological activities including the treatment of glaucoma and ocular hypertension[3], chronic consti ...
OrganicChem10 RxPaths SOLUTIONS (2014)
... Chemists in a variety of industries need to design reactions to make certain organic compounds. Many organic compounds are derived from crude oil products – alkanes or alkenes. The individual reactions you have looked can be linked together to make a particular product from a particular reactant, vi ...
... Chemists in a variety of industries need to design reactions to make certain organic compounds. Many organic compounds are derived from crude oil products – alkanes or alkenes. The individual reactions you have looked can be linked together to make a particular product from a particular reactant, vi ...
Practical and selective aerobic oxidation of alcohols to
... easily removed from the product stream, thus affording an easy workup; and 6. Safe: does not involve hazardous reaction conditions or reagents. ...
... easily removed from the product stream, thus affording an easy workup; and 6. Safe: does not involve hazardous reaction conditions or reagents. ...
CHEM 203 Topics Discussed on Nov. 25 Toxic and carcinogenic
... IMPORTANT: PCC is used ONLY in anhydrous (=water-free) media, while Jones rgt. is an aqueous solution. This seemingly minor difference has a major influence on the course of the reaction of primary alcohols with the two reagents. The Jones reagent: oxidation of primary alcohols to carboxylic acids a ...
... IMPORTANT: PCC is used ONLY in anhydrous (=water-free) media, while Jones rgt. is an aqueous solution. This seemingly minor difference has a major influence on the course of the reaction of primary alcohols with the two reagents. The Jones reagent: oxidation of primary alcohols to carboxylic acids a ...
18 Important and sometimes forgotten) organic transformations
... •Tertiary alcohols are prone to elimination (Chugaev reaction) •Thionoformates may be used to derivatise and deoxygenate tertiary alcohols without competing elimination ...
... •Tertiary alcohols are prone to elimination (Chugaev reaction) •Thionoformates may be used to derivatise and deoxygenate tertiary alcohols without competing elimination ...
212Final`97
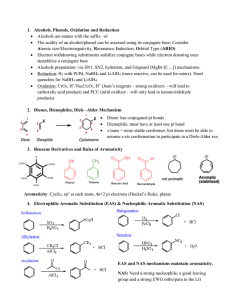
... 4. (12) State whether further electrophilic substitution of each of the following molecules would occur in the ortho / para or meta positions. It is not necessary to explain why. O O H a) b) c) d) S N Br N NH2 CH3 ...
... 4. (12) State whether further electrophilic substitution of each of the following molecules would occur in the ortho / para or meta positions. It is not necessary to explain why. O O H a) b) c) d) S N Br N NH2 CH3 ...
Kinetics and mechanism of oxidation of alcohols
... work has been supported by Hammett reaction constants; the modest negative q values obtained imply that electronreleasing substituents facilitate the oxidation and suggest the removal of hydride ion to be in the rate-limiting step. The values of reaction constants q and q? obtained in this work, alt ...
... work has been supported by Hammett reaction constants; the modest negative q values obtained imply that electronreleasing substituents facilitate the oxidation and suggest the removal of hydride ion to be in the rate-limiting step. The values of reaction constants q and q? obtained in this work, alt ...
The Shell Higher Olefins Process (SHOP)
... This yields about 10 – 15 wt.% of the desired C11 – C14 linear internal alkenes per pass, which are the separated by distillation and converted into detergent alcohols (via a hydroformylation process) or into detergent alkylates (see next slide). ...
... This yields about 10 – 15 wt.% of the desired C11 – C14 linear internal alkenes per pass, which are the separated by distillation and converted into detergent alcohols (via a hydroformylation process) or into detergent alkylates (see next slide). ...
Exam 3 Answer Key
... 12. (7.5) For the following questions match each definition to a term from the list below. ...
... 12. (7.5) For the following questions match each definition to a term from the list below. ...
Answer Key for Final Exam
... 11. Consider all the data shown below, and determine the structure of the unknown molecule. Assign every peak in the 1H NMR and 13C NMR to atoms in the molecule. Also identify key peak(s) in the IR spectrum and MS spectrum (20 points). Molecular formula: C11H14O2 ...
... 11. Consider all the data shown below, and determine the structure of the unknown molecule. Assign every peak in the 1H NMR and 13C NMR to atoms in the molecule. Also identify key peak(s) in the IR spectrum and MS spectrum (20 points). Molecular formula: C11H14O2 ...
Chapter 9. Addition Reactions of Alkenes
... Provide the principal organic reaction product for each of the following reactions. Include stereochemistry of the product when appropriate. If you believe there will be no reaction given the starting materials and the reaction condition, indicate so by writing "NR" for "no reaction". a. ...
... Provide the principal organic reaction product for each of the following reactions. Include stereochemistry of the product when appropriate. If you believe there will be no reaction given the starting materials and the reaction condition, indicate so by writing "NR" for "no reaction". a. ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.