C 1 hapter
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
Ligand Exchange Chromatography
... discriminated by the action of a chiral selector only. The latter has to be involved in interaction with the enantiomers, resulting in the formation of adducts which, being diastereomeric species, may differ in their stability. These adducts in LEC are ternary complexes composed of the molecule of t ...
... discriminated by the action of a chiral selector only. The latter has to be involved in interaction with the enantiomers, resulting in the formation of adducts which, being diastereomeric species, may differ in their stability. These adducts in LEC are ternary complexes composed of the molecule of t ...
Stability constants of some rare-earth-metal chelates
... The use of extinctiometric measurements to determine stability constants is possible if there is a change in absorption of the solution upon complexing. This method has been found to be applicable to the copper nitrate system (29,30) and to the uranyl monochloroacetate system (31)» ...
... The use of extinctiometric measurements to determine stability constants is possible if there is a change in absorption of the solution upon complexing. This method has been found to be applicable to the copper nitrate system (29,30) and to the uranyl monochloroacetate system (31)» ...
The Trans Effect. Implications in Enantioselective
... NH3 Cl z ❑ Groups A and NH3 both occupy part of the 6py and 6pz orbitals. ❑ L is orthogonal to the 6pz orbital, but it overlaps well with the 6py orbital. ❑ Stabilization of the transition state is achieved by increased donation ligand (L) into 6py orbital in ≠. ❑ Calculations for trigonal bipyamida ...
... NH3 Cl z ❑ Groups A and NH3 both occupy part of the 6py and 6pz orbitals. ❑ L is orthogonal to the 6pz orbital, but it overlaps well with the 6py orbital. ❑ Stabilization of the transition state is achieved by increased donation ligand (L) into 6py orbital in ≠. ❑ Calculations for trigonal bipyamida ...
Improved Synthesis, Separation, Transition Metal Coordination and
... P{1H} spectra of meso-Ni2Cl4(et,ph-P4) with 1-hexene in acetone-d6/D2O recorded at 15°C (light blue), 10°C (dark blue), 25°C (black), 50°C (orange), 80°C (purple), and 100°C (red). For higher temperatures the NMR tube was tube pressurized to 90 psi with O2 ......................................... ...
... P{1H} spectra of meso-Ni2Cl4(et,ph-P4) with 1-hexene in acetone-d6/D2O recorded at 15°C (light blue), 10°C (dark blue), 25°C (black), 50°C (orange), 80°C (purple), and 100°C (red). For higher temperatures the NMR tube was tube pressurized to 90 psi with O2 ......................................... ...
PDF (Chapter 4)
... ygen binding and release are summarized first. The nonbiological chemistry of iron and copper in the presence of dioxygen is described briefly to elucidate the key role that the protein plays in supporting oxygen transport by preventing irreversible oxidation of the binding site or of its ligands. T ...
... ygen binding and release are summarized first. The nonbiological chemistry of iron and copper in the presence of dioxygen is described briefly to elucidate the key role that the protein plays in supporting oxygen transport by preventing irreversible oxidation of the binding site or of its ligands. T ...
Rational design of single-site heterogeneous catalysts: towards high
... However, in the epoxidation of these terpenes, the use of materials with an ordered array of mesopores is not strictly necessary in order to have an efficient heterogeneous epoxidation catalyst. Provided that the pore diameter of the solid is large enough to accommodate the reactant molecules and tha ...
... However, in the epoxidation of these terpenes, the use of materials with an ordered array of mesopores is not strictly necessary in order to have an efficient heterogeneous epoxidation catalyst. Provided that the pore diameter of the solid is large enough to accommodate the reactant molecules and tha ...
1 The Chemistry of Gold - Wiley-VCH
... takes place between d10s2 anions but surprisingly yields a similar distance to the aurophilic attraction in d10 cations. The theoretical nature of this effect is not clear. Both the dispersion effects and the net bonding, resulting from partial oxidation from the top of the 6s band, could play a rol ...
... takes place between d10s2 anions but surprisingly yields a similar distance to the aurophilic attraction in d10 cations. The theoretical nature of this effect is not clear. Both the dispersion effects and the net bonding, resulting from partial oxidation from the top of the 6s band, could play a rol ...
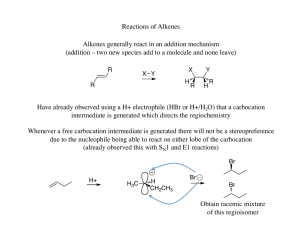
Chapter 11
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
Photochemical and Photophysical Properties of Tetranuclear and
... The excitation spectrum of 1 displays different maxima depending on whether the LE or HE emission maximum is the monitoring ~avelength.~O Interestingly, the ”A‘, for the so-called HE band occurs at lower energy than does that for the LE emission (Table I); thus the Stokes shift46for the latter (1.64 ...
... The excitation spectrum of 1 displays different maxima depending on whether the LE or HE emission maximum is the monitoring ~avelength.~O Interestingly, the ”A‘, for the so-called HE band occurs at lower energy than does that for the LE emission (Table I); thus the Stokes shift46for the latter (1.64 ...
Bonding Capabilities of Transition Metal
... of A is negative, and z , with the phase as defined in 7, mixes of orbitals, the degenerate e level, is nearly degenerate with into z2 with a minus sign. The result, as before, is hybridization the b2 level. The direction of the splitting of the octahedral away from the remaining apical ligand. t2g ...
... of A is negative, and z , with the phase as defined in 7, mixes of orbitals, the degenerate e level, is nearly degenerate with into z2 with a minus sign. The result, as before, is hybridization the b2 level. The direction of the splitting of the octahedral away from the remaining apical ligand. t2g ...
CH 2 - ResearchGate
... NaNO2/HCl (Semi-pinacolone rearrangement) Role of anti-pariplanar relationship ...
... NaNO2/HCl (Semi-pinacolone rearrangement) Role of anti-pariplanar relationship ...
Taking advantage of luminescent lanthanide ions
... application. Trivalent cerium (not shown on Fig. 1) emits a broad band spectrum in the range 370–410 nm due to the interconfigurational allowed 5d–4f transition and is not documented in this review, as is luminescence from the divalent and tetravalent ions (CeIV, SmII, EuII, TbIV, YbII, for instance ...
... application. Trivalent cerium (not shown on Fig. 1) emits a broad band spectrum in the range 370–410 nm due to the interconfigurational allowed 5d–4f transition and is not documented in this review, as is luminescence from the divalent and tetravalent ions (CeIV, SmII, EuII, TbIV, YbII, for instance ...
fundamental concept of transition and inner
... First ionization energy of 5d elements is higher than those of 3d and 4d elements due to greater effective nuclear charge that is experienced over weak shielding of the nucleus by 4f electrons. (g) Acidic character of oxides increases with increase in oxidation number (h) Elements from Cr to Cu have ...
... First ionization energy of 5d elements is higher than those of 3d and 4d elements due to greater effective nuclear charge that is experienced over weak shielding of the nucleus by 4f electrons. (g) Acidic character of oxides increases with increase in oxidation number (h) Elements from Cr to Cu have ...
examination of catalysts containing palladium in oxidation of
... of oxidation in air, mainly in the presence of platinum and sometimes of palladium catalysts. BuseR [1] prepared D-glyconic acid from D-glucose in the course of oxidation in air in the presence of palladium catalyst on calcium carhonate carrier. OKNI [2] and KOROTKY [3] applied Pt catalyst for this ...
... of oxidation in air, mainly in the presence of platinum and sometimes of palladium catalysts. BuseR [1] prepared D-glyconic acid from D-glucose in the course of oxidation in air in the presence of palladium catalyst on calcium carhonate carrier. OKNI [2] and KOROTKY [3] applied Pt catalyst for this ...
Investigation of the steric and electronic properties of 3
... love and support over the past couple of decades, I don’t think I’d be half the person that I am today. The past three years here while obtaining this degree have helped me to realize all of that. To my folks, I couldn’t have asked for a better upbringing; you taught me as much as you could and then ...
... love and support over the past couple of decades, I don’t think I’d be half the person that I am today. The past three years here while obtaining this degree have helped me to realize all of that. To my folks, I couldn’t have asked for a better upbringing; you taught me as much as you could and then ...
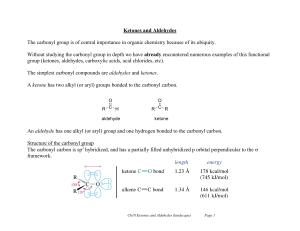
Ketones and Aldehydes
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
... Ketones and Aldehydes The carbonyl group is of central importance in organic chemistry because of its ubiquity. Without studying the carbonyl group in depth we have already encountered numerous examples of this functional group (ketones, aldehydes, carboxylic acids, acid chlorides, etc). The simples ...
Recent advances in homogeneous nickel catalysis
... (Fig. 1) is needed. Nickel is a relatively electropositive late transition metal. Therefore, oxidative addition5, which results in loss of electron density around nickel, tends to occur quite readily (though, conversely, reductive elimination is correspondingly more difficult)6. This facile oxidativ ...
... (Fig. 1) is needed. Nickel is a relatively electropositive late transition metal. Therefore, oxidative addition5, which results in loss of electron density around nickel, tends to occur quite readily (though, conversely, reductive elimination is correspondingly more difficult)6. This facile oxidativ ...
Homogeneous iron complexes for the conversion of dinitrogen into
... The binding of dinitrogen to a metal centre is believed to be crucial for the functionalisation of dinitrogen as it weakens the strong NRN bond. The interaction between a metal centre and dinitrogen involves both s and p components as shown in Fig. 2. The dinitrogen is able to bind as a Lewis base a ...
... The binding of dinitrogen to a metal centre is believed to be crucial for the functionalisation of dinitrogen as it weakens the strong NRN bond. The interaction between a metal centre and dinitrogen involves both s and p components as shown in Fig. 2. The dinitrogen is able to bind as a Lewis base a ...
Retrosynthetic Analysis of Nabumetone
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
... Grignard reactions that produce secondary alcohols, will require oxidation to the corresponding ketone to arrive at the desired product. Starting with the bond between the ring and the side chain, we are given two pairs of synthons as shown in Scheme 2. The synthetic equivalent to the aromatic anion ...
Metal carbonyl

Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. Metal carbonyls are useful in organic synthesis and as catalysts or catalyst precursors in homogeneous catalysis, such as hydroformylation and Reppe chemistry. In the Mond process, nickel carbonyl is used to produce pure nickel. In organometallic chemistry, metal carbonyls serve as precursors for the preparation of other organometalic complexes.Metal carbonyls are toxic by skin contact, inhalation or ingestion, in part because of their ability to carbonylate hemoglobin to give carboxyhemoglobin, which prevents the binding of O2.