Synthesis of Molybdenum Alkylidene Complexes That Contain the 2,6-Dimesitylphenylimido Ligand
... n the past several years Mo and W imido alkylidene complexes that have the formula M(NR)(CHR0 )(OR00 )(Pyr), where Pyr is a pyrrolide or a substituted pyrrolide and OR00 is an aryloxide, have been prepared and explored as olefin metathesis initiators; examples are 1, 2, and 3.1,2 These MonoAlkoxidePy ...
... n the past several years Mo and W imido alkylidene complexes that have the formula M(NR)(CHR0 )(OR00 )(Pyr), where Pyr is a pyrrolide or a substituted pyrrolide and OR00 is an aryloxide, have been prepared and explored as olefin metathesis initiators; examples are 1, 2, and 3.1,2 These MonoAlkoxidePy ...
Coordination Chemistry Reviews Metal catalysts for the vinyl
... Bicyclo[2.2.1]hept-2-ene, better known by its trivial name norbornene (NB, 1) and its derivatives can be polymerized in three different ways (Fig. 1). Each route leads to its own polymer type and can be differentiated through the choice of catalyst. The best-known polymerization of norbornene is the ...
... Bicyclo[2.2.1]hept-2-ene, better known by its trivial name norbornene (NB, 1) and its derivatives can be polymerized in three different ways (Fig. 1). Each route leads to its own polymer type and can be differentiated through the choice of catalyst. The best-known polymerization of norbornene is the ...
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
AMIDES AND RELATED FUNCTIONAL GROUPS
... (alkanes, alkenes, alkynes and aromatic compounds), amides typically are regarded as compounds with low water solubility. They are significantly less water soluble than comparable acids or alcohols due to: 1). their non-ionic character (see acid-base section below), 2). the presence of nonpolar hydr ...
... (alkanes, alkenes, alkynes and aromatic compounds), amides typically are regarded as compounds with low water solubility. They are significantly less water soluble than comparable acids or alcohols due to: 1). their non-ionic character (see acid-base section below), 2). the presence of nonpolar hydr ...
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... – 3-ethyl-2-butanone (what’s wrong with this name?) ...
... – 3-ethyl-2-butanone (what’s wrong with this name?) ...
Consecutive Stability Constants in Solution
... ! For ligands at the left of the series, weakening of the trans Pt–X bond may be most important. ! The trans-directing ligand polarizes the metal ion, inducing a slight repulsion for the leaving ligand in the trans position. ...
... ! For ligands at the left of the series, weakening of the trans Pt–X bond may be most important. ! The trans-directing ligand polarizes the metal ion, inducing a slight repulsion for the leaving ligand in the trans position. ...
Investigating the Dearomative Rearrangement of Biaryl Phosphine- Ligated Pd(II) Complexes
... is quite unusual. Concerted aryl migratory insertion processes from Pd(II) have been proposed as a potential pathway in a large number of C−H arylation reactions,20 but little is known experimentally about the viability of this process, and the direct observation of the product of the insertion of a ...
... is quite unusual. Concerted aryl migratory insertion processes from Pd(II) have been proposed as a potential pathway in a large number of C−H arylation reactions,20 but little is known experimentally about the viability of this process, and the direct observation of the product of the insertion of a ...
Class Notes
... 5. The formation of Grignard Reagents is completely general for all R-Halides: • 3º, 2º, and 1º alkyl halides all work well • Aryl and Vinyl halides as well as alkyl halides work well • RCl, RBr, and RI all work well • For class, we will normally use bromides, due to synthetic accessibility 6. View ...
... 5. The formation of Grignard Reagents is completely general for all R-Halides: • 3º, 2º, and 1º alkyl halides all work well • Aryl and Vinyl halides as well as alkyl halides work well • RCl, RBr, and RI all work well • For class, we will normally use bromides, due to synthetic accessibility 6. View ...
Cobalt-Nickel Separation in Hydrometallurgy: a Review*
... solvent extraction plants at Falconbridge Nikkelverk in Norway, by Eramet in France and was used by Sumitomo at Niihama in Japan [17]. Excellent separation factors in excess of 4000 are achieved by this means. For cation exchangers only the alkyl phosphoric, phosphonic and phosphinic acids show sele ...
... solvent extraction plants at Falconbridge Nikkelverk in Norway, by Eramet in France and was used by Sumitomo at Niihama in Japan [17]. Excellent separation factors in excess of 4000 are achieved by this means. For cation exchangers only the alkyl phosphoric, phosphonic and phosphinic acids show sele ...
PDF (Chapter7)
... platinum, and nickel chemistry.14 In all of these cases, a boron-carbon bond is also cleaved. Previous examples, however, have all shown the boron-methylene link to be the preferential site of reaction. The present example demonstrates that the boron-aryl link is also susceptible to being broken. Th ...
... platinum, and nickel chemistry.14 In all of these cases, a boron-carbon bond is also cleaved. Previous examples, however, have all shown the boron-methylene link to be the preferential site of reaction. The present example demonstrates that the boron-aryl link is also susceptible to being broken. Th ...
Nitrene Transfer Reactions Mediated by Transition
... Figure 2.16: 1H NMR spectra of TpMe,Me-supported complexes 3Fe, 3Co and 3Ni ...........89 Figure 2.17: 1H NMR spectra of TpmPh-supported complexes 2Fe, 2Co and 2Ni .............90 Figure 2.18: 1H NMR spectra of TpPh,Me-supported complexes 4Fe, 4Co and 4Ni ............91 Figure 2.19: Graphical repres ...
... Figure 2.16: 1H NMR spectra of TpMe,Me-supported complexes 3Fe, 3Co and 3Ni ...........89 Figure 2.17: 1H NMR spectra of TpmPh-supported complexes 2Fe, 2Co and 2Ni .............90 Figure 2.18: 1H NMR spectra of TpPh,Me-supported complexes 4Fe, 4Co and 4Ni ............91 Figure 2.19: Graphical repres ...
Molecular Encapsulation - Colin Nuckolls
... emergent properties through supramolecular chemistry. Stephen Craig received his undergraduate degree in chemistry at Duke University, Durham (1991) and obtained an M.Phil. degree from Cambridge (1992) and a Ph.D. from Stanford University (1997). After two years as a Research Chemist in DuPont Centr ...
... emergent properties through supramolecular chemistry. Stephen Craig received his undergraduate degree in chemistry at Duke University, Durham (1991) and obtained an M.Phil. degree from Cambridge (1992) and a Ph.D. from Stanford University (1997). After two years as a Research Chemist in DuPont Centr ...
From metal complexes to fullerene arrays: exploring the exciting
... it is not surprising that supramolecular photochemistry has progressively enlarged the spectrum of interest towards light-powered molecular machines,33 molecular switches 5 and sensors,34 and molecular electronics,35 thus reaching the crossroad between chemistry, biology, and information technology. ...
... it is not surprising that supramolecular photochemistry has progressively enlarged the spectrum of interest towards light-powered molecular machines,33 molecular switches 5 and sensors,34 and molecular electronics,35 thus reaching the crossroad between chemistry, biology, and information technology. ...
Preparation and structural characterization of group 1 metal
... as dimethoxyethane (DME), THF, Et2O, toluene, or pentane produced cleanly the dilithium complex as indicated by 31P{1H} NMR spectroscopy. Upon lithiation, the 31P chemical shift typically changes from 50 ppm to ca. 32 ppm regardless of the solvent employed. Coordinating solvents such as THF and Et2O ...
... as dimethoxyethane (DME), THF, Et2O, toluene, or pentane produced cleanly the dilithium complex as indicated by 31P{1H} NMR spectroscopy. Upon lithiation, the 31P chemical shift typically changes from 50 ppm to ca. 32 ppm regardless of the solvent employed. Coordinating solvents such as THF and Et2O ...
Full-Text PDF
... for epoxidation reactions is well illustrated by some past reviews on the matter. In particular, iron-porphyrins have been the subject of extensive reviews, due to their biological relevance as part of cytochrome P450 [14,15], with their use as catalysts in epoxidation reactions being highlighted on ...
... for epoxidation reactions is well illustrated by some past reviews on the matter. In particular, iron-porphyrins have been the subject of extensive reviews, due to their biological relevance as part of cytochrome P450 [14,15], with their use as catalysts in epoxidation reactions being highlighted on ...
5.New functional coordination compounds
... Narrow sense: Coordination compounds are formed by ligand and central atom according to certain composition and space configurations. Broad sense: The compound forming by atom A and atom B ...
... Narrow sense: Coordination compounds are formed by ligand and central atom according to certain composition and space configurations. Broad sense: The compound forming by atom A and atom B ...
Metal-Catalyzed Epoxidations of Alkenes with Hydrogen
... does not undergo redox chemistry during catalytic reactions; it remains in the +VII oxidation state. MTO activates H2O2 through an equilibrium formation of the η2-peroxo species 8 and 9. The structure of 9 was confirmed crystallographically, and the methyl resonances of 8 and 9 were detected by both ...
... does not undergo redox chemistry during catalytic reactions; it remains in the +VII oxidation state. MTO activates H2O2 through an equilibrium formation of the η2-peroxo species 8 and 9. The structure of 9 was confirmed crystallographically, and the methyl resonances of 8 and 9 were detected by both ...
Complexes for use in metathesis reactions
... Ganter, Chiral organometallic half-sandwich complexes with de?ned metal con?guration. Chem Soc Rev. 32(3): 130-8 (2003). Garber, S. B., et al., Ef?cient and recyclable monomeric and dendritic Ru-based metathesis catalysts. J. Am. Chem. Soc. 122, 8168-8179 ...
... Ganter, Chiral organometallic half-sandwich complexes with de?ned metal con?guration. Chem Soc Rev. 32(3): 130-8 (2003). Garber, S. B., et al., Ef?cient and recyclable monomeric and dendritic Ru-based metathesis catalysts. J. Am. Chem. Soc. 122, 8168-8179 ...
Evolution of strategies to prepare synthetic mimics of carboxylate
... after ~10 min. The UV-visible (UV-vis), Mössbauer, resonance Raman, and EXAFS spectra of the transient intermediate closely match those of oxyHr (Scheme 1, top), strongly suggesting that 11 contains a (hydroperoxo)(µ-oxo)diiron(III) unit. Unlike Hr, however, oxygenation of 10 is irreversible and le ...
... after ~10 min. The UV-visible (UV-vis), Mössbauer, resonance Raman, and EXAFS spectra of the transient intermediate closely match those of oxyHr (Scheme 1, top), strongly suggesting that 11 contains a (hydroperoxo)(µ-oxo)diiron(III) unit. Unlike Hr, however, oxygenation of 10 is irreversible and le ...
Complexometric Reactions and Titrations
... A ligand is called a monodentate if it donates a single pair of electrons (like :NH3) while a bidentate ligand (like ethylenediamine, :NH2CH2CH2H2N:) donates two pairs of electrons. Ethylenediaminetetraacetic acid (EDTA) is a hexadentate ligand. The ligand can be as simple as ammonia which forms a ...
... A ligand is called a monodentate if it donates a single pair of electrons (like :NH3) while a bidentate ligand (like ethylenediamine, :NH2CH2CH2H2N:) donates two pairs of electrons. Ethylenediaminetetraacetic acid (EDTA) is a hexadentate ligand. The ligand can be as simple as ammonia which forms a ...
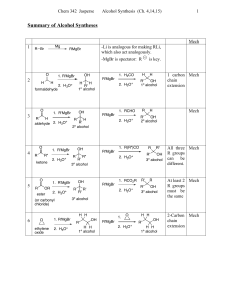
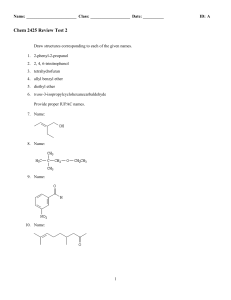
Chem 2425-Test 2 Review
... aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed to carboxylic acids using aqueous base. Unfortunately, when a cyanohydrin is treated with aqueous base the original carbonyl compound is isolated. ...
... aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed to carboxylic acids using aqueous base. Unfortunately, when a cyanohydrin is treated with aqueous base the original carbonyl compound is isolated. ...
raman spectroscopy of transition metal complexes: molecular
... other compilations.[3,4] This preference is mainly due to widely available, sensitive, easy-to-use IR and FTIR spectrometers, concisely summarized by Harris and Bertolucci in their book on symmetry and spectroscopy published in 1978:[5] ‘‘At present, instrumentation of IR spectroscopy is generally m ...
... other compilations.[3,4] This preference is mainly due to widely available, sensitive, easy-to-use IR and FTIR spectrometers, concisely summarized by Harris and Bertolucci in their book on symmetry and spectroscopy published in 1978:[5] ‘‘At present, instrumentation of IR spectroscopy is generally m ...
The highly preorganized ligands 1,10-Phenanthroline
... attachment to gadolinium, leaving one available coordination site for a water molecule This is of importance because the ability to shift the MRI signal is a function of the number of water molecules directly attached to gadolinium.8 An ideal contrast agent would not have as many points of attachmen ...
... attachment to gadolinium, leaving one available coordination site for a water molecule This is of importance because the ability to shift the MRI signal is a function of the number of water molecules directly attached to gadolinium.8 An ideal contrast agent would not have as many points of attachmen ...
Metal carbonyl

Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. Metal carbonyls are useful in organic synthesis and as catalysts or catalyst precursors in homogeneous catalysis, such as hydroformylation and Reppe chemistry. In the Mond process, nickel carbonyl is used to produce pure nickel. In organometallic chemistry, metal carbonyls serve as precursors for the preparation of other organometalic complexes.Metal carbonyls are toxic by skin contact, inhalation or ingestion, in part because of their ability to carbonylate hemoglobin to give carboxyhemoglobin, which prevents the binding of O2.