Chapter 7 Chemical Reactions
... There are millions of compounds that will produce endless chemical reactions, therefore not all chemical reactions can be carried out in the laboratory A system is used to classify chemical reactions, which allows chemist to recognize patterns and predict the products of reactions One of these ...
... There are millions of compounds that will produce endless chemical reactions, therefore not all chemical reactions can be carried out in the laboratory A system is used to classify chemical reactions, which allows chemist to recognize patterns and predict the products of reactions One of these ...
Chapter 10
... • The new amine can then also act as a nucleophile. • Thus the final reaction mixture will consist of varying ratios of RNH2, R2NH, R3N, and R4N+Cl–. • These ratios are difficult to control and, therefore, amines are avoided as nucleophiles in nucleophilic substitutions reactions. ...
... • The new amine can then also act as a nucleophile. • Thus the final reaction mixture will consist of varying ratios of RNH2, R2NH, R3N, and R4N+Cl–. • These ratios are difficult to control and, therefore, amines are avoided as nucleophiles in nucleophilic substitutions reactions. ...
CHAPTER 1 Synthesis of amides using Lewis acid catalyst: Iodine
... to first activate the carboxylic acid, a process that usually takes place by converting the –OH of the acid into a good leaving group prior to treatment with the amine (Scheme 1.19). O R ...
... to first activate the carboxylic acid, a process that usually takes place by converting the –OH of the acid into a good leaving group prior to treatment with the amine (Scheme 1.19). O R ...
Alcohols phenols ethers
... (7) Action of Grignard reagents: All the three types of the alcohols can be ...
... (7) Action of Grignard reagents: All the three types of the alcohols can be ...
AP Chemistry Summer Assignment
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
AP Chemistry Summer Assignment
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
AP Chemistry Summer Assignment
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
... fine students, and with plenty of motivation and hard work you should find AP Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and gra ...
Homogeneous and Heterogeneous Catalysis
... rate of a reaction by lowering the activation energy required to reach the transition state. Unlike reactants, a catalyst is not consumed as part of the reaction process. Catalysts can be divided into two types depending on the reaction phase that they occupy: homogeneous and heterogeneous. Homogene ...
... rate of a reaction by lowering the activation energy required to reach the transition state. Unlike reactants, a catalyst is not consumed as part of the reaction process. Catalysts can be divided into two types depending on the reaction phase that they occupy: homogeneous and heterogeneous. Homogene ...
Chapter 6
... A student carries out an experiment to standardize a sodium hydroxide solution. To do this, the student weighs out 1.3009 g sample of potassium hydrogen phthalate (KHC8H4O4 or KHP–molar mass 204.22 g/mol). The student dissolves the KHP in distilled water, adds phenolphthalein as an indicator, and ti ...
... A student carries out an experiment to standardize a sodium hydroxide solution. To do this, the student weighs out 1.3009 g sample of potassium hydrogen phthalate (KHC8H4O4 or KHP–molar mass 204.22 g/mol). The student dissolves the KHP in distilled water, adds phenolphthalein as an indicator, and ti ...
A Mild and Convenient Conversion of Ketones to the Corresponding
... hydrogen evolution.') The clear solution was stirred for 30 rnin and then the tosylhydrazoneof 6-oxo-15-hexadecenoicacid (0.62, 1.42 mmol) was added. The reduction was allowed to proceed for 60 mids and then NaOAc.3Hz0 (0.46 g, 3.4 mmol) was added and the solution allowed to warm to room temperature ...
... hydrogen evolution.') The clear solution was stirred for 30 rnin and then the tosylhydrazoneof 6-oxo-15-hexadecenoicacid (0.62, 1.42 mmol) was added. The reduction was allowed to proceed for 60 mids and then NaOAc.3Hz0 (0.46 g, 3.4 mmol) was added and the solution allowed to warm to room temperature ...
File - chemistryattweed
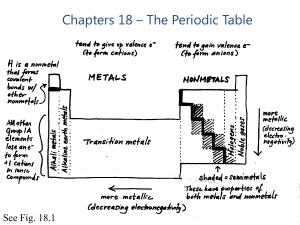
... oxides and non-metal oxides show a pattern in properties. Metal oxides are usually basic and non-metal oxides usually acidic. The extent of the acidity or basicity of an oxide can often be predicted from the element's position in the Periodic Table. ...
... oxides and non-metal oxides show a pattern in properties. Metal oxides are usually basic and non-metal oxides usually acidic. The extent of the acidity or basicity of an oxide can often be predicted from the element's position in the Periodic Table. ...
PPT
... • The carbonyl group is moderately polar, but it doesn’t have any hydrogen atoms attached, so it cannot hydrogen bond between molecules. ...
... • The carbonyl group is moderately polar, but it doesn’t have any hydrogen atoms attached, so it cannot hydrogen bond between molecules. ...
Hein and Arena - faculty at Chemeketa
... The concentrations of A, B, C, and D represent the equilibrium concentrations. The brackets around [A], [B], [C], and [D] represent concentrations in Molarity. The products are written on the top of the fraction & the reactants on the bottom. The coefficients to balance the equation a, b, c, and d a ...
... The concentrations of A, B, C, and D represent the equilibrium concentrations. The brackets around [A], [B], [C], and [D] represent concentrations in Molarity. The products are written on the top of the fraction & the reactants on the bottom. The coefficients to balance the equation a, b, c, and d a ...
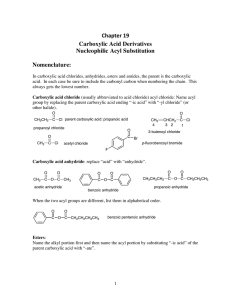
Chapter 19 Carboxylic Acid Derivatives Nucleophilic Acyl
... Since a nucleophile can attack either carbonyl, symmetrical anhydrides are usually used so as to give one product. (1) Preparation of Esters (a) We can use neutral alcohols. With the neutral alcohols we usually use acid catalysis to activate the carbonyl carbon of the anhydride to nucleophilic attac ...
... Since a nucleophile can attack either carbonyl, symmetrical anhydrides are usually used so as to give one product. (1) Preparation of Esters (a) We can use neutral alcohols. With the neutral alcohols we usually use acid catalysis to activate the carbonyl carbon of the anhydride to nucleophilic attac ...
AP Chemistry Summer Assignment
... fine students, and with plenty of motivation and hard work you should find AP/IB Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and ...
... fine students, and with plenty of motivation and hard work you should find AP/IB Chemistry a successful and rewarding experience. Finally, I recommend that you spread out the summer assignment. Please do not try to complete it all in the final week of the summer. Chemistry takes time to process and ...
Chapters 18 – The Periodic Table
... H2 (g); H2 molecules dissociate at the metal surface and H atoms occupy holes in the crystal structure (potential use as a portable fuel) ...
... H2 (g); H2 molecules dissociate at the metal surface and H atoms occupy holes in the crystal structure (potential use as a portable fuel) ...
Drawing Organic Structures Functional Groups
... • Name = alkyl group name • Number = point of attachment to parent chain • Two substituents on the same C get the same number 4. Write the name as a single word • Substituents before parent name (include #) • Separate # and word with hyphen • Separate two numbers with a comma • List substituents in ...
... • Name = alkyl group name • Number = point of attachment to parent chain • Two substituents on the same C get the same number 4. Write the name as a single word • Substituents before parent name (include #) • Separate # and word with hyphen • Separate two numbers with a comma • List substituents in ...
Alcohols, Penols, and Thiols
... pKa’s of Selected Alcohols & Phenols • 2,2,2-trifluoroethanol is a much stronger acid than is ...
... pKa’s of Selected Alcohols & Phenols • 2,2,2-trifluoroethanol is a much stronger acid than is ...
homework assignment 2 - the Petersen Home Page
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
CHM-373 American Women in Science and Society
... Synthesis of Aldehydes and Ketones • Hydration of Alkynes • Involves a keto-enol tautomerization • Mixture of ketones seen with internal alkynes ...
... Synthesis of Aldehydes and Ketones • Hydration of Alkynes • Involves a keto-enol tautomerization • Mixture of ketones seen with internal alkynes ...
4888 Journal of the American Chemical Society 1OO:lS 1 July 19
... iodoso compound) which can lead to olefins by a syn elimination process. It is known that amine oxides,2 sulfoxide^,^ and selenoxides4 undergo thermal pericyclic eliminations of hydroxylamine, sulfenic acid (RSOH), and selenenic acid (RSeOH), respectively, to form olefins. Alkyl iodide oxides (iodos ...
... iodoso compound) which can lead to olefins by a syn elimination process. It is known that amine oxides,2 sulfoxide^,^ and selenoxides4 undergo thermal pericyclic eliminations of hydroxylamine, sulfenic acid (RSOH), and selenenic acid (RSeOH), respectively, to form olefins. Alkyl iodide oxides (iodos ...
Arenes - Science Skool!
... A little dinitrobenzene may also be formed by the further attack of NO2+ on nitrobenzene (which is now more suspectible to attack due to the electron inductive effect of the NO2+ group). This extra group will go the the 3 position to give the disubstituted product 1,3-dinitrobenzene. To actually pro ...
... A little dinitrobenzene may also be formed by the further attack of NO2+ on nitrobenzene (which is now more suspectible to attack due to the electron inductive effect of the NO2+ group). This extra group will go the the 3 position to give the disubstituted product 1,3-dinitrobenzene. To actually pro ...
solutions - chem.msu.su
... at the first step, and formation of the acyl chloride with its subsequent transformation into the amide C as the second one. This is followed by deprotection resulting in D, containing amide and amino groups. Then D is treated with an acid providing the corresponding salt. Finally, formaldehyde is a ...
... at the first step, and formation of the acyl chloride with its subsequent transformation into the amide C as the second one. This is followed by deprotection resulting in D, containing amide and amino groups. Then D is treated with an acid providing the corresponding salt. Finally, formaldehyde is a ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.