Loeblein chemistry clicker questions2013
... 1. There are 2 balloons in a room. They are identical in size and material. One balloon is filled with air and the other balloon is filled with Helium. How does the pressure of the air balloon compare to the pressure of the Helium balloon. The pressure in the air balloon is ...
... 1. There are 2 balloons in a room. They are identical in size and material. One balloon is filled with air and the other balloon is filled with Helium. How does the pressure of the air balloon compare to the pressure of the Helium balloon. The pressure in the air balloon is ...
Carefully detach the last page. It is the Data Sheet.
... This exam is being written by several thousand students. Please be sure that you follow the instructions below. We'll send you a report on your performance. Top performers are eligible for a prize. The names of the top 200 students will be published in the September issue of Chem 13 News. ...
... This exam is being written by several thousand students. Please be sure that you follow the instructions below. We'll send you a report on your performance. Top performers are eligible for a prize. The names of the top 200 students will be published in the September issue of Chem 13 News. ...
Advanced Higher Chemistry Resource Guide
... pair/bonding pair. These different strengths of electron pair repulsion account for slight deviations from expected bond angles in molecules such as NH3 and H2O. Transition metals The d-block transition metals are metals with an incomplete d subshell in at least one of their ions. The filling of the ...
... pair/bonding pair. These different strengths of electron pair repulsion account for slight deviations from expected bond angles in molecules such as NH3 and H2O. Transition metals The d-block transition metals are metals with an incomplete d subshell in at least one of their ions. The filling of the ...
Document
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
Acid-Base
... A buffer solution contains 0.40 mole of formic acid, HCOOH, and 0.60 mole of sodium formate, HCOONa, in 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1. ...
... A buffer solution contains 0.40 mole of formic acid, HCOOH, and 0.60 mole of sodium formate, HCOONa, in 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1. ...
Gas Chromatography-Mass Spectrometry (GCMS) of Amino Acids
... This lab is devoted to biological molecules. The keys to biologically active molecules are the functional groups in the molecules. Functional groups define the class of molecules. For example, every amino acid has both a carboxyl and an amino group. However, functional groups also add polarity to th ...
... This lab is devoted to biological molecules. The keys to biologically active molecules are the functional groups in the molecules. Functional groups define the class of molecules. For example, every amino acid has both a carboxyl and an amino group. However, functional groups also add polarity to th ...
Mock Examination (2016/2017) CHEMISTRY PAPER 1 SECTION B
... write in the margins. Answers written in the margins will not be marked. ...
... write in the margins. Answers written in the margins will not be marked. ...
Document
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
... • When a mixture of stereoisomers is possible from a dehydrohalogenation, the major product is the more stable stereoisomer. • A reaction is stereoselective when it forms predominantly or exclusively one stereoisomer when two or more are possible. • The E2 reaction is stereoselective because one ste ...
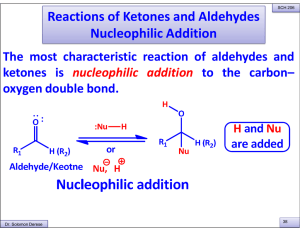
Reactions of Ketones and Aldehydes Nucleophilic Addition
... There is a complication, however. The carbonyl group in the starting alkyne will neither tolerate the strongly basic conditions required for anion formation nor survive in a solution containing carbanions. Acetylide ions add to carbonyl groups . The strategy that is routinely followed is to protect ...
... There is a complication, however. The carbonyl group in the starting alkyne will neither tolerate the strongly basic conditions required for anion formation nor survive in a solution containing carbanions. Acetylide ions add to carbonyl groups . The strategy that is routinely followed is to protect ...
Use the following answers for questions 10
... 26. According to the rate law for the reaction, an increase in the concentration of hydronium ion has what effect on this reaction? (A) The rate of reaction increases. (B) The rate of reaction decreases. (C) The value of the equilibrium constant increases. (D) The value of the equilibrium constant d ...
... 26. According to the rate law for the reaction, an increase in the concentration of hydronium ion has what effect on this reaction? (A) The rate of reaction increases. (B) The rate of reaction decreases. (C) The value of the equilibrium constant increases. (D) The value of the equilibrium constant d ...
Ch 4 Student.pptx
... • Balanced chemical equations provide the exact relationships between the amount of reactants and products. • 2 C8H18 (l) + 25 O2 (g) → 16 CO2 (g) + 18 H2O (g) • For example 2 molecules of octane (gasoline) react with 25 molecules of oxygen to produce 16 molecules of carbon dioxide gas and 18 mol ...
... • Balanced chemical equations provide the exact relationships between the amount of reactants and products. • 2 C8H18 (l) + 25 O2 (g) → 16 CO2 (g) + 18 H2O (g) • For example 2 molecules of octane (gasoline) react with 25 molecules of oxygen to produce 16 molecules of carbon dioxide gas and 18 mol ...
Unit 3 Exam Level Questions
... Which of the following graphs shows how the rates of the forward and reverse reactions change when carbon monoxide and steam are mixed? ...
... Which of the following graphs shows how the rates of the forward and reverse reactions change when carbon monoxide and steam are mixed? ...
CIS Exam Questions
... Which of the following graphs shows how the rates of the forward and reverse reactions change when carbon monoxide and steam are mixed? ...
... Which of the following graphs shows how the rates of the forward and reverse reactions change when carbon monoxide and steam are mixed? ...
chemical reactions
... Elements listed higher will displace any elements listed below them. For example Na will displace any elements listed below it from one of its compounds. 2 Na (s) + MgCl2 (aq) 2 NaCl (aq) + Mg (s) Na (s) + AgCl (aq) NaCl (aq) + Ag (s) ...
... Elements listed higher will displace any elements listed below them. For example Na will displace any elements listed below it from one of its compounds. 2 Na (s) + MgCl2 (aq) 2 NaCl (aq) + Mg (s) Na (s) + AgCl (aq) NaCl (aq) + Ag (s) ...
Full-Text PDF
... All of the starting materials used in this work are commercially available or were prepared according to published procedures [24-27]. All of the products are known compounds and were identified by comparison of their physical and spectral data with those of authentic samples. Reported yields refer ...
... All of the starting materials used in this work are commercially available or were prepared according to published procedures [24-27]. All of the products are known compounds and were identified by comparison of their physical and spectral data with those of authentic samples. Reported yields refer ...
1. A pharmacy analyst supervises the state of a refractometer. For its
... 6. Diethyl ether relates to simple ethers. Prior to its identification by using the boiling temperature an analytical chemist must ensure that there are no: A Peroxides B Reducing substances C Alcohols D Non-volatile residue E Carboxylic acids Before determining the boiling temperature first determ ...
... 6. Diethyl ether relates to simple ethers. Prior to its identification by using the boiling temperature an analytical chemist must ensure that there are no: A Peroxides B Reducing substances C Alcohols D Non-volatile residue E Carboxylic acids Before determining the boiling temperature first determ ...
The Advanced Placement Examination in Chemistry Acid–Base
... A buffer solution contains 0.40 mole of formic acid, HCOOH, and 0.60 mole of sodium formate, HCOONa, in 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1. ...
... A buffer solution contains 0.40 mole of formic acid, HCOOH, and 0.60 mole of sodium formate, HCOONa, in 1.00 litre of solution. The ionization constant, Ka, of formic acid is 1.810–4. (a) Calculate the pH of this solution. (b) If 100. millilitres of this buffer solution is diluted to a volume of 1. ...
Chapter 1 Introduction
... performance was also investigated. A hot filtration experiment coupled with a blank test revealed that oxidation proceeded mostly on nickel-salen sites in LDH-[nickel-salen]. A reaction mechanism is proposed based on the experimental results[31] Layered double hydroxides (LDH) with brucite like stru ...
... performance was also investigated. A hot filtration experiment coupled with a blank test revealed that oxidation proceeded mostly on nickel-salen sites in LDH-[nickel-salen]. A reaction mechanism is proposed based on the experimental results[31] Layered double hydroxides (LDH) with brucite like stru ...
Chapter 7 Review
... For the reaction CO(g) + 2 H2(g) <---> CH3OH(g) + heat; [CO(g)] = 0.025 mol/L, [H2(g) ] = 0.050 mol/L and [CH3OH(g)] = 0.0063 mol/L a) b) ...
... For the reaction CO(g) + 2 H2(g) <---> CH3OH(g) + heat; [CO(g)] = 0.025 mol/L, [H2(g) ] = 0.050 mol/L and [CH3OH(g)] = 0.0063 mol/L a) b) ...
towards the synthesis of functionalised macrocyclic receptors
... polyethers. This classic reaction involves the SN2 displacement of an alkyl halide with an alkoxide anion21 when a diol and dihalide are reacted together in presence of a base. Recently it was reported that the halide leaving group could be replaced by a much more reactive tosylate or mesylate leavi ...
... polyethers. This classic reaction involves the SN2 displacement of an alkyl halide with an alkoxide anion21 when a diol and dihalide are reacted together in presence of a base. Recently it was reported that the halide leaving group could be replaced by a much more reactive tosylate or mesylate leavi ...
The decomposition of hydrogen peroxide to form water and oxygen
... The normal boiling point of N2H4 is 114 oC, whereas the normal boiling point of C2H6 is -89 oC. Explain, in terms of the intermolecular forces present in each liquid, which the boiling point of N2H4 is so much higher than that of C2H6. ...
... The normal boiling point of N2H4 is 114 oC, whereas the normal boiling point of C2H6 is -89 oC. Explain, in terms of the intermolecular forces present in each liquid, which the boiling point of N2H4 is so much higher than that of C2H6. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.