IChO 2012
... Boron-nitrogen chemistry has attracted significant attention in part because a B–N unit is isoelectronic with C–C. Furthermore, the radius of carbon and its electronegativity are roughly the average of those properties for B and N. One of the simplest boron-nitrogen compounds is H3N–BH3, the ammonia ...
... Boron-nitrogen chemistry has attracted significant attention in part because a B–N unit is isoelectronic with C–C. Furthermore, the radius of carbon and its electronegativity are roughly the average of those properties for B and N. One of the simplest boron-nitrogen compounds is H3N–BH3, the ammonia ...
Common Student Misconceptions
... • These substances exist as a mixture of ions and un-ionized molecules in solution. • The predominant form of the solute is the un-ionized molecule. • Example: acetic acid, HC2H3O2. HC2H3O2(aq) ⇋ H+(aq) + C2H3O2–(aq) • The double arrow means that the reaction is significant in both directions. • It ...
... • These substances exist as a mixture of ions and un-ionized molecules in solution. • The predominant form of the solute is the un-ionized molecule. • Example: acetic acid, HC2H3O2. HC2H3O2(aq) ⇋ H+(aq) + C2H3O2–(aq) • The double arrow means that the reaction is significant in both directions. • It ...
Organic Chemistry
... Because of the relative stability of alkyl radical intermediates, selectivity in free radical halogenation favors tertiary over secondary over primary carbon radicals. Bromination, though, is more selective than chlorination, because the proton extraction step is more endothermic in bromination than ...
... Because of the relative stability of alkyl radical intermediates, selectivity in free radical halogenation favors tertiary over secondary over primary carbon radicals. Bromination, though, is more selective than chlorination, because the proton extraction step is more endothermic in bromination than ...
Solution Chemistry and the Hydrosphere
... © 2012 by W. W. Norton & Company © 2012 by W. W. Norton & Company ...
... © 2012 by W. W. Norton & Company © 2012 by W. W. Norton & Company ...
Organic Molecules
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
... structures containing thousands of atoms! Although carbon is present in all organic compounds, other elements such as hydrogen (H), oxygen (O), nitrogen (N), sulfur (S) and phosphorus (P) are also common in these molecules. Until the early nineteenth century, chemists had managed to make many simple ...
CHEMICAL REACTIONS
... · One type of redox reactions are single replacement reactions. In these reactions an active metal replace a less active metal. · Activity series is a listing of metallic elements in descending order of reactivity. Hydrogen is also included in the series since it behaves similar to metals. · ...
... · One type of redox reactions are single replacement reactions. In these reactions an active metal replace a less active metal. · Activity series is a listing of metallic elements in descending order of reactivity. Hydrogen is also included in the series since it behaves similar to metals. · ...
Energy Matters - Perth Grammar
... The number of H+ (aq) ions in the beaker decreased. The pH of the solution decreased. The number of SO42−(aq) ions in the beaker decreased. Water molecules formed during the reaction. A precipitate formed during the reaction. The final solution contained equal numbers of H+(aq) and OH− (aq) ions. ...
... The number of H+ (aq) ions in the beaker decreased. The pH of the solution decreased. The number of SO42−(aq) ions in the beaker decreased. Water molecules formed during the reaction. A precipitate formed during the reaction. The final solution contained equal numbers of H+(aq) and OH− (aq) ions. ...
Solution
... Hydrolysis-polymerization reactions can proceed until finally the resulting polymers become too large to remain soluble, i.e., they achieve colloidal dimensions. When this stage is reached, we say we have precipitated a solid. In this hypothetical system consisting of only one type of metal ion in w ...
... Hydrolysis-polymerization reactions can proceed until finally the resulting polymers become too large to remain soluble, i.e., they achieve colloidal dimensions. When this stage is reached, we say we have precipitated a solid. In this hypothetical system consisting of only one type of metal ion in w ...
File - wilson science WEBSITE
... If a system at equilibrium is disturbed by adding more NO to the system, which of the following will occur? a. the equilibrium [Cl2] will decrease and K will decrease b. the equilibrium [Cl2] will decrease, and the K will remain the same c. the equilibrium [Cl2] will increase, and the K will increas ...
... If a system at equilibrium is disturbed by adding more NO to the system, which of the following will occur? a. the equilibrium [Cl2] will decrease and K will decrease b. the equilibrium [Cl2] will decrease, and the K will remain the same c. the equilibrium [Cl2] will increase, and the K will increas ...
Document
... While dehydrations have favorable entropy changes, the position of equilibrium favors reactants. Operation of the Principle of Le Châtelier in Alcohol Dehydrations • removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alke ...
... While dehydrations have favorable entropy changes, the position of equilibrium favors reactants. Operation of the Principle of Le Châtelier in Alcohol Dehydrations • removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alke ...
AP Chemistry
... 0.345 g of impure BeC2O4 is dissolved in water and titrated to equivalence with 17.8 mL of 0.0150 M KMnO4(aq) according to the equation: 16 H+ + 2 MnO4- + 5 C2O42- 2 Mn2+ + 10 CO2 + 8 H2O (1) How many moles of KMnO4 are used? ...
... 0.345 g of impure BeC2O4 is dissolved in water and titrated to equivalence with 17.8 mL of 0.0150 M KMnO4(aq) according to the equation: 16 H+ + 2 MnO4- + 5 C2O42- 2 Mn2+ + 10 CO2 + 8 H2O (1) How many moles of KMnO4 are used? ...
Acid-Base Equilibria - Riverside Local Schools
... acids and bases by their characteristic properties. Acids have a sour taste (for example, citric acid in lemon juice) and cause certain dyes to change color (for example, litmus turns red on contact with acids). Indeed, the word acid comes from the Latin word acidus, meaning sour or tart. Bases, in ...
... acids and bases by their characteristic properties. Acids have a sour taste (for example, citric acid in lemon juice) and cause certain dyes to change color (for example, litmus turns red on contact with acids). Indeed, the word acid comes from the Latin word acidus, meaning sour or tart. Bases, in ...
Study Guide Chapter 4 Alcohols and Alkyl Halides
... The hydroxyl group is trans to the isopropyl group and cis to the methyl group. All three substituents need not always be equatorial; instead, one or two of them may be axial. Since neomenthol is the second most stable stereoisomer, we choose the structure with one axial substituent. Furthermore, we ...
... The hydroxyl group is trans to the isopropyl group and cis to the methyl group. All three substituents need not always be equatorial; instead, one or two of them may be axial. Since neomenthol is the second most stable stereoisomer, we choose the structure with one axial substituent. Furthermore, we ...
Deuterium fractionation of methylamine through atomic grain
... Deuterium fractionation of methylamine through atomic grain-surface reactions at low temperatures: implication for the possible D/H ratio in molecular clouds Y. Oba, T. Chigai, N. Watanabe, and A. Kouchi Institute of Low Temperature Science, Hokkaido University, Japan Interstellar methylamine (CH3NH ...
... Deuterium fractionation of methylamine through atomic grain-surface reactions at low temperatures: implication for the possible D/H ratio in molecular clouds Y. Oba, T. Chigai, N. Watanabe, and A. Kouchi Institute of Low Temperature Science, Hokkaido University, Japan Interstellar methylamine (CH3NH ...
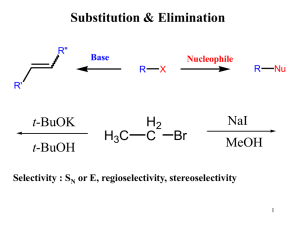
10 Haloalkanes and Haloarenes
... substitution reaction product instead of addition reaction product. This is because at higher temperature, the addition reaction is reversible and the substitution reaction is irreversible. The hydrogen atom of allylic carbon is replaced with the halogen atom to form allylic halides and the reaction ...
... substitution reaction product instead of addition reaction product. This is because at higher temperature, the addition reaction is reversible and the substitution reaction is irreversible. The hydrogen atom of allylic carbon is replaced with the halogen atom to form allylic halides and the reaction ...
NATIONAL HIGH SCHOOL CHEMISTRY EXAMINATION (1995
... 19. Pure solid ammonium carbamate (NH4 CO2 NH 2 ) is put into a vessel which already contained ammonia at a partial pressure of 10.1 kPa. The solid ammonium carbamate is allowed to dissociate according to the equation: NH 4 CO2 NH 2 (s) → 2 NH 3 (g) + CO2 (g) ...
... 19. Pure solid ammonium carbamate (NH4 CO2 NH 2 ) is put into a vessel which already contained ammonia at a partial pressure of 10.1 kPa. The solid ammonium carbamate is allowed to dissociate according to the equation: NH 4 CO2 NH 2 (s) → 2 NH 3 (g) + CO2 (g) ...
Le Chatelier`s Principle Notes
... With the rate of the moving stairs and your walking evenly matched, you appear to be at a standstill. But what happens if the escalator begins moving just a little faster? If you want to maintain the same position you had, at some specific point between the bottom and the top of the stairs, you'll a ...
... With the rate of the moving stairs and your walking evenly matched, you appear to be at a standstill. But what happens if the escalator begins moving just a little faster? If you want to maintain the same position you had, at some specific point between the bottom and the top of the stairs, you'll a ...
Chapter 9 Stoichiometry
... produced based on the limiting reactant. If everything in the reaction went according to plan, and all of the reactant(s) reacted, this is how much product should be made. This is NOT the same as the actual yield- amount that is produced based on an experiment Error occurs, so actual yield is ...
... produced based on the limiting reactant. If everything in the reaction went according to plan, and all of the reactant(s) reacted, this is how much product should be made. This is NOT the same as the actual yield- amount that is produced based on an experiment Error occurs, so actual yield is ...
1994 Released Exam
... (B) basic becauseCa(OH)2 is a weak and insoluble base (C) neutral if the concentrationis kept below 0.1 molar (D) acidic becauseof the hydrolysisof the Ca” ions (E) acidic becausethe acid HOC1 is formed ...
... (B) basic becauseCa(OH)2 is a weak and insoluble base (C) neutral if the concentrationis kept below 0.1 molar (D) acidic becauseof the hydrolysisof the Ca” ions (E) acidic becausethe acid HOC1 is formed ...
AP Chemistry
... conservation of mass, which states that the total mass of the products of a chemical reaction is the same as the total mass of the reactants. Likewise, the same numbers of atoms of each type are present before and after a chemical reaction. A balanced chemical equation shows equal numbers of atoms o ...
... conservation of mass, which states that the total mass of the products of a chemical reaction is the same as the total mass of the reactants. Likewise, the same numbers of atoms of each type are present before and after a chemical reaction. A balanced chemical equation shows equal numbers of atoms o ...
Kinetics Simulations of the Neutralizing Capacity of Silicate Minerals
... (Lawrence and Wang, 1997). Experimental tests also show that mafic silicates can provide an appreciable amount of acid neutralizing capacity that can affect interpretations of standard acid base accounting determinations (Desborough et al., 1998; Jambor et al., 2000; Paktunc, 1999). The primary obje ...
... (Lawrence and Wang, 1997). Experimental tests also show that mafic silicates can provide an appreciable amount of acid neutralizing capacity that can affect interpretations of standard acid base accounting determinations (Desborough et al., 1998; Jambor et al., 2000; Paktunc, 1999). The primary obje ...
I have put this in the format of the 1984 exam
... If the equilibrium constant for the reaction above is 3.7 x 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, G°, for this reaction? (A) E° is positive and G° is negative. (B) E° is negative and G° is positive. (C) E° and G° are both ...
... If the equilibrium constant for the reaction above is 3.7 x 1015, which of the following correctly describes the standard voltage, E°, and the standard free energy change, G°, for this reaction? (A) E° is positive and G° is negative. (B) E° is negative and G° is positive. (C) E° and G° are both ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.