Chapter 4 Aqueous Reactions and Solution Stoichiometry
... Analyze: We are given several chemical formulas and asked to classify each substance as a strong electrolyte, weak electrolyte, or nonelectrolyte. Plan: The approach we take is outlined in Table 4.3. We can predict whether a substance is ionic or molecular, based on its composition. As we saw in Sec ...
... Analyze: We are given several chemical formulas and asked to classify each substance as a strong electrolyte, weak electrolyte, or nonelectrolyte. Plan: The approach we take is outlined in Table 4.3. We can predict whether a substance is ionic or molecular, based on its composition. As we saw in Sec ...
Glossary - Chemistry (Intro)
... Noble Gas: Nonmetallic elements in group 8A; He, Ne, Ar, Kr, Xe, and Rn. Transition Metal.: Elements that have incompletely filled d subshells or readily give rise to cations that have incompletely filled d subshells; i.e.: it belongs to the central part of the periodic table, between Groups II and ...
... Noble Gas: Nonmetallic elements in group 8A; He, Ne, Ar, Kr, Xe, and Rn. Transition Metal.: Elements that have incompletely filled d subshells or readily give rise to cations that have incompletely filled d subshells; i.e.: it belongs to the central part of the periodic table, between Groups II and ...
Dr. Spencer`s PPT
... Nonelectrolytes are not dissociated into ions in solution Extent of dissolution does not dictate strong or weak electrolyte solution (i.e., HC2H3O2 is very soluble but is a weak electrolyte while Ba(OH)2 is only slightly soluble is a strong electrolyte) ...
... Nonelectrolytes are not dissociated into ions in solution Extent of dissolution does not dictate strong or weak electrolyte solution (i.e., HC2H3O2 is very soluble but is a weak electrolyte while Ba(OH)2 is only slightly soluble is a strong electrolyte) ...
3.0 Properties of Phosgene
... iron and produce a fire. Detection of these impurity generated reactions can be noticed by a rapid rise in the temperature of the feed gas after the carbon monoxide and chlorine mixing point. The use of high mixing temperature automatic shutdown is a useful method to help eliminate this type of fail ...
... iron and produce a fire. Detection of these impurity generated reactions can be noticed by a rapid rise in the temperature of the feed gas after the carbon monoxide and chlorine mixing point. The use of high mixing temperature automatic shutdown is a useful method to help eliminate this type of fail ...
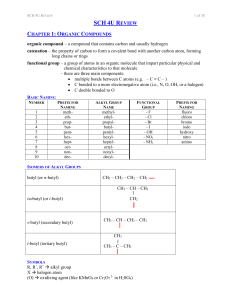
SCH 4U REVIEW Notes
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
answers to part a of the national high school
... of Science Learning Outcomes, see http://www.cmec.ca/science/framework/Pages/english/1112%20chemistry.html. The people involved in preparing the NHSCE very much appreciate all the comments and feedback that we get from teachers. We have tried to incorporate some of these comments in with the solutio ...
... of Science Learning Outcomes, see http://www.cmec.ca/science/framework/Pages/english/1112%20chemistry.html. The people involved in preparing the NHSCE very much appreciate all the comments and feedback that we get from teachers. We have tried to incorporate some of these comments in with the solutio ...
5 SURFACE CHEMISTRY CATEGORY
... 4.Define osmotic pressure. How is it that measurement of osmotic pressures is more widely used for determining molar masses of macromolecules than the rise in boiling point or fall in freezing point of their solutions? 5. Calculate the amount of KCl which must be added to 1 kg of water so that its f ...
... 4.Define osmotic pressure. How is it that measurement of osmotic pressures is more widely used for determining molar masses of macromolecules than the rise in boiling point or fall in freezing point of their solutions? 5. Calculate the amount of KCl which must be added to 1 kg of water so that its f ...
Chemical Reactions and Solution Stoichiometry
... Notice that in Interactive Figure 4.2.1 the water molecules orient themselves so that the oxygen atoms are near the Na+ cations and the hydrogen atoms are near the Cl− anions. This is due to the polar nature of water, a result of uneven electron distribution in water molecules. ( Flashforward to Se ...
... Notice that in Interactive Figure 4.2.1 the water molecules orient themselves so that the oxygen atoms are near the Na+ cations and the hydrogen atoms are near the Cl− anions. This is due to the polar nature of water, a result of uneven electron distribution in water molecules. ( Flashforward to Se ...
Problem 28. TUNNELING IN CHEMISTRY
... applied to macrosystems. To illustrate this idea, E. Schrödinger proposed the following mental experiment. Consider the Geiger counter which detects the entering electrons. The counter is connected to a device which breaks the glass with the poison when the particle enters the counter. Near the glas ...
... applied to macrosystems. To illustrate this idea, E. Schrödinger proposed the following mental experiment. Consider the Geiger counter which detects the entering electrons. The counter is connected to a device which breaks the glass with the poison when the particle enters the counter. Near the glas ...
A Crash Course In Organic Chemistry
... Addition of a halogen can reduce or eliminate flammability, and can also increase potency. Depending on the halogen, some of these compounds can cause arrhythmias and/or renal or hepatic toxicity. Compounds containing only bromine are generally not useful. Compounds containing only chlorine are subj ...
... Addition of a halogen can reduce or eliminate flammability, and can also increase potency. Depending on the halogen, some of these compounds can cause arrhythmias and/or renal or hepatic toxicity. Compounds containing only bromine are generally not useful. Compounds containing only chlorine are subj ...
File - cpprashanths Chemistry
... Q29.Discuss oxides of nitrogen.Give structure and calculate the oxidation number of nitrogen in each of them.Arrange the oxides in order of increasing acid strength. Nitrogen forms a wide range of oxided .The oxidation number of nitrogen in these oxides varies from +1 to +5.The oxides are 1)N2O din ...
... Q29.Discuss oxides of nitrogen.Give structure and calculate the oxidation number of nitrogen in each of them.Arrange the oxides in order of increasing acid strength. Nitrogen forms a wide range of oxided .The oxidation number of nitrogen in these oxides varies from +1 to +5.The oxides are 1)N2O din ...
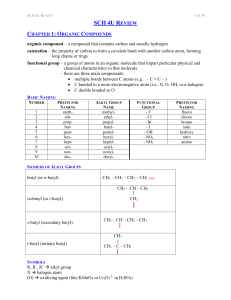
12 U Chem Review
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
sch4ureview
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
... catenation – the property of carbon to form a covalent bond with another carbon atom, forming long chains or rings functional group – a group of atoms in an organic molecule that impart particular physical and chemical characteristics to that molecule – there are three main components: multiple bo ...
Physical and Chemical equilibrium
... When there is a change in the state of occurrence of matter, then a physical transformation is said to have occurred. The equilibrium concepts are also applicable to physical state transformations of matter. (i) Solid-liquid equilibria Here, the solid and the liquid forms of a substance co exist at ...
... When there is a change in the state of occurrence of matter, then a physical transformation is said to have occurred. The equilibrium concepts are also applicable to physical state transformations of matter. (i) Solid-liquid equilibria Here, the solid and the liquid forms of a substance co exist at ...
4.2- Reaction Stoichiometry Reaction Stoichiometry
... Mass Relationships in Chemical Equations(Mass –to – Mass conversion ) Estimate the mass of CO2 produced in 2006 by the combustion of 3.5 × 1015 g petroleum(octane C8H18). Solution: The equation for the reaction is: 2 C8H18(l) + 25 O2(g) 16 CO2(g) + 18 H2O(g) ...
... Mass Relationships in Chemical Equations(Mass –to – Mass conversion ) Estimate the mass of CO2 produced in 2006 by the combustion of 3.5 × 1015 g petroleum(octane C8H18). Solution: The equation for the reaction is: 2 C8H18(l) + 25 O2(g) 16 CO2(g) + 18 H2O(g) ...
Physical Chemistry Problems. ©Mike Lyons 2009
... problems on the material delivered in lectures. Formulating and solving problems is a key skill needed for success in this branch of Chemistry, which has the reputation of being difficult because of its intrinsic mathematical nature. This booklet has been produced to provide Trinity College JF Chemi ...
... problems on the material delivered in lectures. Formulating and solving problems is a key skill needed for success in this branch of Chemistry, which has the reputation of being difficult because of its intrinsic mathematical nature. This booklet has been produced to provide Trinity College JF Chemi ...
Ch 17 Equilibrium Notes
... n = sum of coefficients of gaseous products minus sum of coefficients of gaseous reactants. Calculate Kp for the following reaction: PCl3(g) + Cl2(g) ↔ PCl3(g) where Kc= 1.67(500 K) Do Follow up Problem 17.5 Pg.735. Solving Equilibrium Problems 1. Balance the equation. 2.Write the equilibrium expre ...
... n = sum of coefficients of gaseous products minus sum of coefficients of gaseous reactants. Calculate Kp for the following reaction: PCl3(g) + Cl2(g) ↔ PCl3(g) where Kc= 1.67(500 K) Do Follow up Problem 17.5 Pg.735. Solving Equilibrium Problems 1. Balance the equation. 2.Write the equilibrium expre ...
Chapter 4
... the molarity desired for a particular solution. Dilution with water does not alter the numbers of moles of solute present. Moles of solute before dilution = moles of solute after dilution M1V1 = M2V2 Return to TOC ...
... the molarity desired for a particular solution. Dilution with water does not alter the numbers of moles of solute present. Moles of solute before dilution = moles of solute after dilution M1V1 = M2V2 Return to TOC ...
File - Chem with Appleby
... reactions are proceeding _____________ ___________________________ Once equilibrium is achieved, the _______________ of each reactant and product remains ________________. ...
... reactions are proceeding _____________ ___________________________ Once equilibrium is achieved, the _______________ of each reactant and product remains ________________. ...
This article was published in an Elsevier journal. The attached copy
... an easy phase separation. In the subsequent step, Section 3 including reactions (9) and (10), the separation of HI from L − 2, the heavier iodine/iodide–water phase, is the most critical scenario of the cycle [4] and believed to be the most expensive and energy-consuming step [5]. After establishing ...
... an easy phase separation. In the subsequent step, Section 3 including reactions (9) and (10), the separation of HI from L − 2, the heavier iodine/iodide–water phase, is the most critical scenario of the cycle [4] and believed to be the most expensive and energy-consuming step [5]. After establishing ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.